Scalable Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharma Supply Chains
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112480015B introduces a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a structural motif prevalent in numerous bioactive molecules. This technology addresses long-standing challenges in heterocyclic chemistry by leveraging a palladium-catalyzed carbonylation cascade that operates under relatively mild conditions compared to traditional high-pressure methods. The significance of this advancement cannot be overstated for a reliable pharmaceutical intermediate supplier, as it opens new avenues for accessing complex fluorinated architectures with high atom economy. Quinazolinones are renowned for their diverse pharmacological profiles, ranging from antifungal and antibacterial activities to potent anticancer properties, making them indispensable in modern drug discovery pipelines. By integrating this novel methodology into our production capabilities, we can offer clients a streamlined pathway to these high-value intermediates, ensuring both chemical excellence and supply chain resilience.
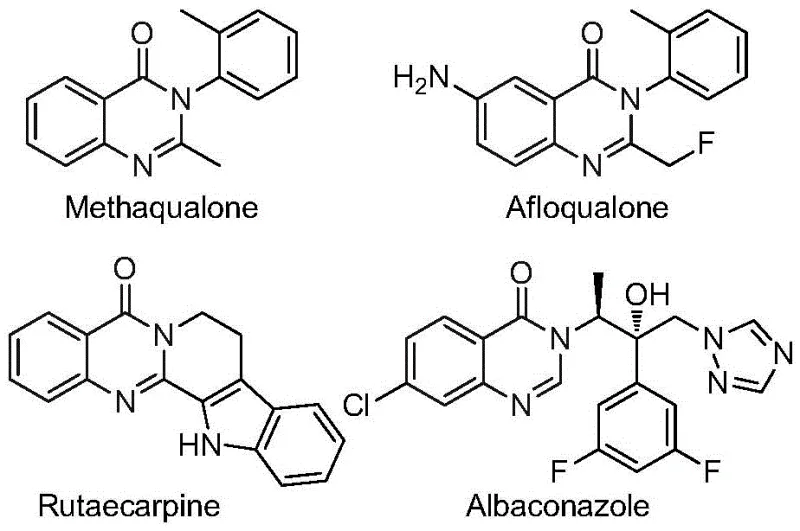
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been fraught with synthetic inefficiencies that hinder large-scale manufacturing and cost-effective procurement. Traditional protocols often rely on the use of hazardous high-pressure carbon monoxide gas, which necessitates specialized autoclave equipment and rigorous safety protocols, thereby inflating capital expenditure and operational risks. Furthermore, many established routes require expensive and less accessible starting materials, such as 2-bromoformylaniline or pre-activated acid anhydrides, which significantly drive up the raw material costs for the final active pharmaceutical ingredient. Other methods involving ruthenium or platinum catalysts often suffer from narrow substrate scope and low turnover numbers, leading to poor yields and difficult purification processes that generate substantial chemical waste. These limitations create bottlenecks in cost reduction in API manufacturing, as the cumulative effect of expensive reagents, harsh conditions, and complex workups erodes profit margins and extends lead times for critical drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112480015B represents a paradigm shift towards greener and more economical synthesis by utilizing cheap and readily available nitro compounds as the nitrogen source. This innovative approach employs molybdenum hexacarbonyl as a solid, safe surrogate for carbon monoxide gas, effectively eliminating the need for high-pressure infrastructure while maintaining high reaction efficiency. The one-pot nature of the reaction allows for the direct coupling of trifluoroethylimidoyl chloride with nitro compounds in the presence of a palladium catalyst, streamlining the process into a single operational step. This simplification not only reduces the number of unit operations required but also minimizes solvent consumption and waste generation, aligning perfectly with modern principles of sustainable chemistry. For procurement teams, this translates to a more stable supply of precursors and a drastic simplification of the manufacturing workflow, ultimately enhancing the overall viability of producing high-purity OLED material or pharmaceutical intermediates based on this scaffold.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its intricate catalytic cycle, which seamlessly integrates nitro reduction, amidation, and cyclization into a cohesive sequence. The reaction initiates with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which simultaneously serves as the carbon monoxide source upon thermal decomposition. Subsequently, the generated amine undergoes a base-promoted intermolecular coupling with trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate in situ. The palladium catalyst then enters the cycle by oxidative addition into the carbon-iodine bond of the imidoyl chloride derivative, forming a reactive divalent palladium species. This is followed by the insertion of the carbon monoxide released from the molybdenum complex into the carbon-palladium bond, generating an acyl-palladium intermediate that is poised for cyclization.
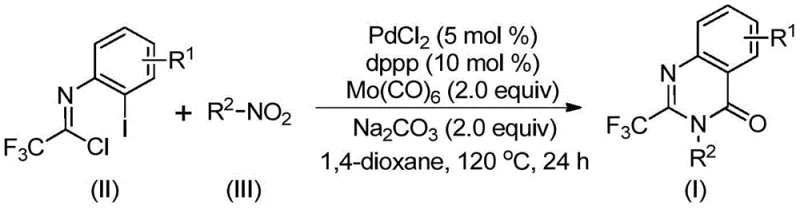
Following the carbonyl insertion, the intramolecular nucleophilic attack by the nitrogen atom onto the acyl-palladium center facilitates the formation of a seven-membered cyclic palladium intermediate. This key step is promoted by the presence of a base, such as sodium carbonate, which helps to neutralize acidic byproducts and drive the equilibrium forward. The catalytic cycle concludes with a reductive elimination step that releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium(0) species for further turnover. Understanding this mechanism is crucial for R&D directors focused on impurity control, as it highlights the importance of precise stoichiometry and temperature control to prevent side reactions such as homocoupling or incomplete cyclization. The robustness of this catalytic system ensures high selectivity and yield, even with sterically demanding substrates, making it a superior choice for the commercial scale-up of complex polymer additives or fine chemicals.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Executing this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the specific molar ratios of palladium chloride, dppp ligand, and sodium carbonate as outlined in the patent examples. The use of anhydrous 1,4-dioxane as the solvent is preferred to ensure optimal solubility of all components and to facilitate the smooth progression of the carbonylation cascade at elevated temperatures. Detailed standardized synthetic steps for this specific transformation are provided in the guide below, ensuring reproducibility across different laboratory scales.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and the nitro compound substrate in 1,4-dioxane solvent within a Schlenk tube.
- Heat the reaction mixture to 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion via the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product using column chromatography to isolate the high-purity quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that directly address the pain points of modern chemical supply chains. By shifting away from hazardous gaseous reagents and expensive pre-functionalized substrates, manufacturers can achieve significant cost savings in raw material acquisition and handling. The reliance on commodity chemicals like nitro compounds and simple imidoyl chlorides ensures a stable and diversified supply base, reducing the risk of shortages that often plague specialty chemical markets. Furthermore, the operational simplicity of the one-pot procedure reduces the demand for highly specialized labor and complex equipment maintenance, contributing to a leaner and more agile production model.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide equipment removes a major capital expense barrier, allowing for production in standard glass-lined reactors which are more widely available and cheaper to maintain. Additionally, the use of earth-abundant nitro compounds as starting materials instead of scarce brominated precursors drastically lowers the bill of materials, enabling substantial cost savings that can be passed down the value chain to the end customer without compromising on quality standards.
- Enhanced Supply Chain Reliability: Since the key starting materials are bulk commodities with multiple global suppliers, the risk of supply disruption is minimized compared to routes relying on proprietary or single-source reagents. The robustness of the reaction conditions, tolerating a wide range of functional groups, means that slight variations in raw material quality can be accommodated without batch failure, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates minimal waste due to its high atom efficiency and the use of a solid CO source, simplifying effluent treatment and lowering environmental compliance costs. The ability to scale this reaction from milligram to kilogram quantities without re-optimizing critical parameters demonstrates its readiness for industrial adoption, providing a seamless transition from process development to full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method utilizes cheap and readily available nitro compounds and trifluoroethylimidoyl chloride, avoiding the need for high-pressure carbon monoxide gas or expensive pre-activated substrates like 2-bromoformylaniline, thereby significantly simplifying the operational complexity and safety requirements.
Q: What is the substrate scope for this palladium-catalyzed reaction?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents on the aromatic ring including halogens (F, Cl, Br), alkyl groups (methyl), and electron-withdrawing groups (trifluoromethyl), as well as diverse amine components like phenyl, naphthyl, and cyclohexyl groups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and beyond, utilizing standard organic solvents like dioxane and common catalysts, which facilitates the commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of synthetic innovation to deliver superior value to our global clientele. Our technical team has thoroughly analyzed the potential of this palladium-catalyzed route and is fully equipped to translate these laboratory findings into robust industrial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle complex organometallic chemistry safely, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to explore how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality chemical solutions efficiently and reliably.
