Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Pharma
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Pharma
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. Quinazolinone compounds, as vital nitrogen-containing fused ring systems, are ubiquitous in natural products and pharmaceutical agents exhibiting potent anti-cancer, anticonvulsant, and anti-inflammatory activities. As illustrated in the structural diversity of bioactive molecules below, the quinazolinone core serves as a privileged scaffold for developing next-generation therapeutics. The patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in existing synthetic routes.
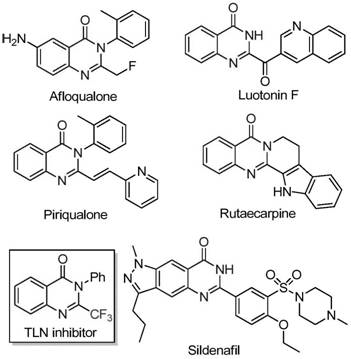
This technological breakthrough offers a robust pathway for generating high-value pharmaceutical intermediates with superior purity profiles. By leveraging a novel iron-catalyzed cyclization strategy, the process circumvents the reliance on costly fluorinating reagents and severe reaction conditions that have historically plagued this chemical space. For R&D directors and procurement specialists alike, this methodology presents a compelling opportunity to optimize supply chains for complex heterocyclic building blocks. The ability to access these structurally intricate motifs through a streamlined, cost-effective protocol is essential for accelerating drug discovery pipelines and ensuring the commercial viability of late-stage clinical candidates targeting various oncological and neurological indications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has been constrained by significant economic and operational inefficiencies. Literature precedents predominantly describe cyclization reactions utilizing synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate in conjunction with substrates like anthranilamide or isatoic anhydride. These conventional pathways are frequently hampered by severe reaction conditions that demand rigorous temperature control and specialized equipment, leading to escalated operational expenditures. Furthermore, the substrate scope in traditional methods is often narrow, limiting the structural diversity accessible to medicinal chemists during lead optimization phases. The reliance on expensive starting materials not only inflates the cost of goods sold but also introduces supply chain vulnerabilities, as the availability of specialized fluorinated reagents can be inconsistent. Additionally, these older protocols often suffer from mediocre yields and generate substantial chemical waste, posing challenges for environmental compliance and sustainable manufacturing practices in regulated industries.
The Novel Approach
In stark contrast to legacy techniques, the disclosed invention utilizes readily available trifluoroethylimidoyl chloride and isatin as primary starting materials, catalyzed by inexpensive ferric chloride. This paradigm shift enables a highly efficient series of cyclization reactions that proceed under relatively mild conditions compared to prior art. The general reaction scheme depicted below highlights the elegance of this transformation, where simple mixing of reagents in a polar aprotic solvent leads to the desired heterocyclic core. The use of iron as a catalyst is particularly advantageous from a commercial perspective, as it eliminates the need for precious metals like palladium or rhodium, drastically reducing raw material costs. Moreover, the reaction demonstrates exceptional functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkyl groups, and methoxy moieties without compromising yield. This versatility empowers chemists to rapidly generate libraries of analogues for structure-activity relationship studies while maintaining a lean and agile production workflow.
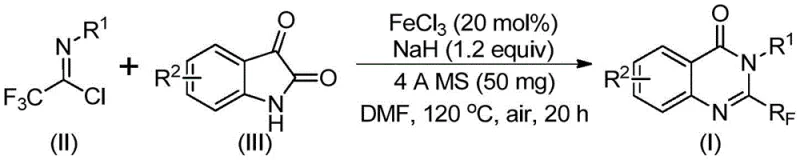
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base-promoted bond formation and transition metal catalysis. Initially, an alkali-promoted carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and the isatin substrate, generating a trifluoroacetamidine intermediate. This step is crucial for setting up the molecular architecture required for the subsequent ring closure. Following this initial condensation, the ferric chloride catalyst facilitates a decarbonylation and cyclization sequence that drives the isomerization towards the thermodynamically stable quinazolinone product. The presence of 4A molecular sieves in the reaction mixture plays a pivotal role by sequestering moisture, thereby preventing hydrolysis of the sensitive imidoyl chloride and shifting the equilibrium towards product formation. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the stoichiometric ratio of sodium hydride to catalyst, to maximize conversion efficiency and minimize the formation of side products.
From an impurity control perspective, the robustness of this iron-catalyzed system ensures a clean reaction profile, which is paramount for pharmaceutical applications. The high selectivity of the cyclization minimizes the generation of regioisomers or over-fluorinated byproducts that are difficult to separate. The reaction conditions, specifically the two-stage heating protocol starting at 40°C and ramping to 120°C, are optimized to balance reaction kinetics with thermal stability. This controlled thermal profile prevents the degradation of sensitive functional groups on the aromatic rings, ensuring that the final product retains the intended substitution pattern. For quality assurance teams, this means that the crude product requires less intensive purification, streamlining the downstream processing and reducing the overall cycle time from synthesis to final isolation. The ability to predictably manage impurity profiles through mechanistic understanding is a key differentiator for manufacturers aiming to meet stringent regulatory standards.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The procedure involves charging a reaction vessel with the catalyst, base, and molecular sieves before introducing the organic substrates in a solvent like DMF. The reaction is allowed to proceed through a defined temperature gradient, ensuring complete conversion before workup. Detailed standardized synthesis steps see the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C and continue reacting for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers transformative benefits regarding cost structure and logistical reliability. The shift away from exotic fluorinating agents to commodity chemicals like isatin and aromatic amines fundamentally alters the cost basis of production. By utilizing earth-abundant iron catalysts instead of precious metals, the process eliminates the need for expensive metal scavenging steps and reduces the risk of heavy metal contamination in the final API. This simplification of the purification train translates directly into lower manufacturing costs and faster batch turnover times. Furthermore, the use of common solvents and standard reaction vessels means that this chemistry can be implemented in existing multipurpose facilities without requiring significant capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic impact of replacing precious metal catalysts with ferric chloride cannot be overstated in the context of large-scale production. Iron is orders of magnitude cheaper than palladium or platinum, and its removal from the final product is straightforward, avoiding the complex and costly resin treatments often required for noble metals. Additionally, the high atom economy of the cyclization reaction ensures that a greater proportion of the starting mass ends up in the final product, reducing waste disposal costs. The elimination of harsh reagents also lowers the burden on safety systems and personal protective equipment, contributing to a safer and more cost-effective operational environment. These cumulative savings allow for a more competitive pricing strategy when supplying these intermediates to downstream pharmaceutical partners.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term drug development projects, and this method leverages raw materials that are widely available in the global chemical market. Isatin and substituted anilines are produced in massive quantities for various industries, ensuring a steady supply even during market fluctuations. Unlike specialized fluorinated synthons that may have limited suppliers and long lead times, the precursors for this reaction are commoditized. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in reagent quality, providing a buffer against supply chain inconsistencies and ensuring consistent delivery schedules to clients.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration at the gram level with clear potential for kilogram and tonne-scale expansion. The reaction does not require extreme pressures or cryogenic temperatures, making it inherently safer and easier to scale in standard stainless steel reactors. From an environmental standpoint, the reduction in hazardous waste generation aligns with green chemistry principles and increasingly strict regulatory frameworks. The simplified workup procedure, involving filtration and standard chromatography, reduces the volume of solvent waste compared to multi-step traditional syntheses. This environmental efficiency not only reduces disposal costs but also enhances the sustainability profile of the manufacturing site, a factor that is becoming increasingly important for corporate social responsibility reporting and client audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their own development workflows.
Q: What are the advantages of this iron-catalyzed method over traditional synthesis?
A: Traditional methods often rely on expensive trifluoroacetic anhydride or ethyl trifluoroacetate and suffer from harsh conditions and low yields. This novel method uses cheap, readily available isatin and imidoyl chlorides with inexpensive iron catalysts, offering higher yields and better functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to simple operation, cheap raw materials, and convenient post-treatment procedures.
Q: What is the role of the 4A molecular sieves in this reaction?
A: The 4A molecular sieves act as a drying agent and likely assist in driving the equilibrium of the condensation reaction forward by removing water, thereby improving the overall conversion efficiency of the cyclization process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demands whether you are in early discovery or late-stage clinical trials. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry with precision, delivering materials that accelerate your timeline to market.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this iron-catalyzed method. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to build a resilient and cost-efficient supply chain for your next generation of quinazolinone-based therapeutics.
