Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Global Pharmaceutical Supply Chains
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Intermediates for Global Pharmaceutical Supply Chains
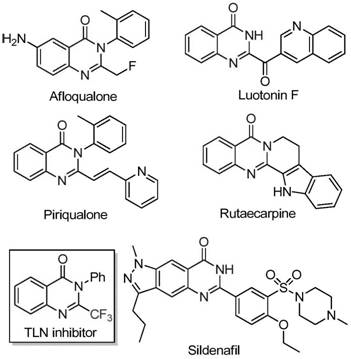
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for a wide array of bioactive molecules exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these structures is known to drastically enhance metabolic stability, lipophilicity, and bioavailability, making them highly prized assets in modern drug discovery pipelines. This report analyzes the technical merits and commercial implications of this iron-catalyzed methodology for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. While chemically valid, these traditional pathways suffer from significant drawbacks that hinder their viability for large-scale commercial manufacturing. The reaction conditions are often severe, requiring harsh reagents that pose safety risks and environmental challenges. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, can be prohibitively expensive and difficult to source in bulk quantities. Perhaps most critically for process chemists, these legacy methods frequently exhibit narrow substrate scope and inconsistent yields, leading to unpredictable production timelines and increased waste generation, which directly impacts the bottom line for procurement teams.
The Novel Approach
In stark contrast to these legacy issues, the methodology outlined in the referenced patent introduces a transformative approach utilizing readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials. This route leverages a cheap, earth-abundant iron catalyst to drive the cyclization, representing a major departure from precious metal-dependent processes. The innovation lies not just in the reagents but in the operational simplicity; the reaction proceeds efficiently under relatively mild thermal gradients and utilizes common organic solvents. By shifting the synthetic logic to utilize isatin, a commodity chemical, the process inherently lowers the barrier to entry for production. This new pathway offers broad functional group tolerance, allowing for the facile synthesis of diverse analogues without the need for extensive protecting group strategies, thereby streamlining the overall synthetic sequence and enhancing the economic feasibility of producing these high-value intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
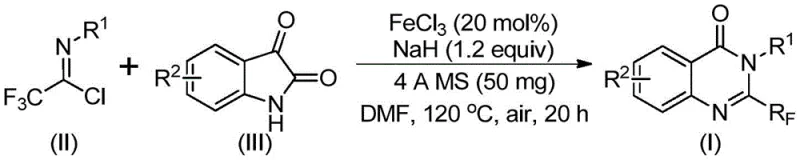
The core of this technological advancement is the iron-catalyzed decarbonylation and cyclization mechanism. The reaction initiates with the interaction between the trifluoroethylimidoyl chloride and the isatin derivative in the presence of a base, typically sodium hydride. This initial step likely facilitates the formation of a carbon-nitrogen bond, generating a trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst plays a pivotal role in promoting the decarbonylation of this intermediate. This step is crucial as it drives the thermodynamic equilibrium towards the formation of the stable quinazolinone ring system. The use of 4A molecular sieves in the reaction mixture further aids the process by sequestering moisture, ensuring that the sensitive imidoyl chloride and the reactive sodium hydride function optimally without hydrolysis side reactions.
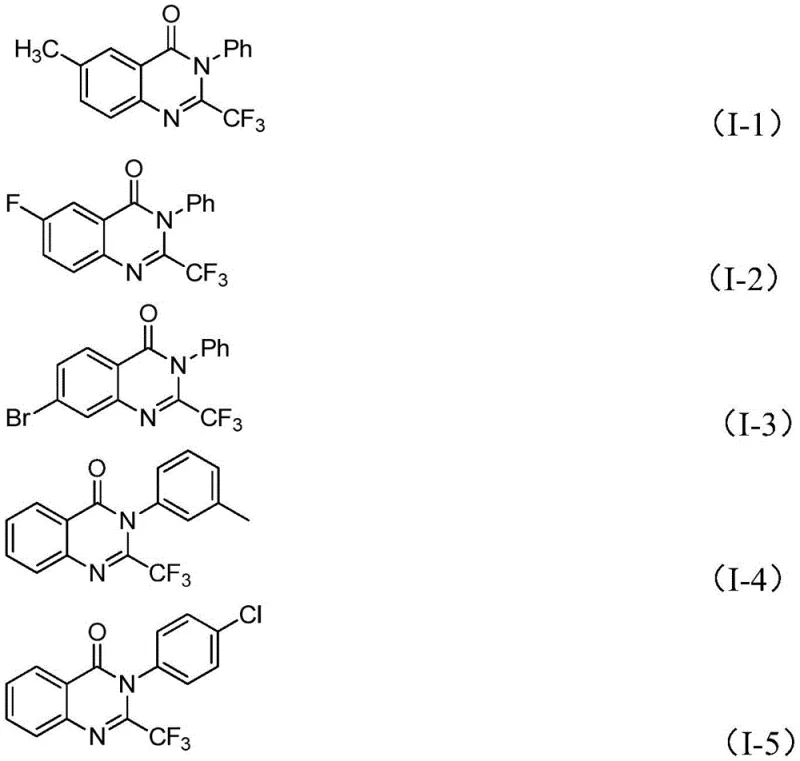
Beyond the primary mechanism, the robustness of this catalytic system is evidenced by its exceptional substrate scope. The reaction tolerates a wide variety of electronic and steric environments on both the imidoyl chloride and the isatin components. Electron-donating groups such as methyl and methoxy, as well as electron-withdrawing groups like halogens and nitro groups, are all compatible with the reaction conditions. This versatility allows medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies. The ability to synthesize complex, substituted quinazolinones in high yields without compromising purity is a testament to the selectivity of the iron catalyst. This mechanistic efficiency translates directly to reduced downstream purification burdens, a key factor in maintaining high throughput in a commercial setting.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it highly suitable for technology transfer from lab to plant. The process involves a straightforward one-pot procedure where reagents are combined in a polar aprotic solvent, typically DMF, which effectively dissolves both the organic substrates and the inorganic catalyst. The reaction profile involves a two-stage heating protocol, starting at a lower temperature to allow for initial adduct formation before ramping up to drive the cyclization to completion. For detailed standard operating procedures and specific stoichiometric ratios optimized for different scales, please refer to the standardized synthesis guide below.
- Combine ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an organic solvent such as DMF.
- Stir the mixture at 40°C for 8-10 hours to initiate the reaction, then increase temperature to 120°C.
- Maintain reaction at 120°C for 18-20 hours under air, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology offers compelling strategic advantages that extend beyond simple chemistry. The shift from precious metal catalysts to iron chloride fundamentally alters the cost structure of the manufacturing process. Iron is orders of magnitude cheaper than palladium or rhodium, and its use eliminates the need for costly and time-consuming heavy metal scavenging steps that are mandatory for pharmaceutical grade products. This reduction in processing complexity leads to substantial cost savings in terms of both raw materials and operational overhead. Furthermore, the reliance on commodity chemicals like isatin ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or single-source reagents that often plague the fine chemical industry.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of abundant starting materials drastically reduces the bill of materials. Additionally, the simplified workup procedure, which avoids complex metal removal protocols, lowers the consumption of specialized resins and solvents, contributing to a leaner and more cost-efficient production model that enhances overall margin potential.
- Enhanced Supply Chain Reliability: By utilizing widely available building blocks such as substituted isatins and aromatic amines, the manufacturing process becomes less vulnerable to supply disruptions. The robustness of the reaction conditions also means that production can be maintained across different geographic locations without significant re-optimization, ensuring consistent supply continuity for downstream API manufacturers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The reaction demonstrates excellent scalability, having been validated from gram-scale experiments to potential multi-kilogram production runs. The use of iron, a non-toxic metal, aligns with green chemistry principles and simplifies waste disposal compliance. The high atom economy and reduced solvent usage compared to traditional multi-step syntheses further minimize the environmental footprint, facilitating easier regulatory approval and sustainability reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this technology into their supply chains.
Q: What are the advantages of using FeCl3 over precious metal catalysts?
A: Using ferric chloride eliminates the need for expensive palladium or rhodium catalysts, significantly reducing raw material costs and simplifying heavy metal removal processes required for pharmaceutical compliance.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded from gram-level laboratory synthesis to industrial scale applications due to the use of robust, commercially available starting materials like isatin.
Q: What is the functional group tolerance of this reaction?
A: The method exhibits excellent functional group tolerance, successfully accommodating various substituents including halogens, alkyl groups, and methoxy groups on both the aryl ring and the isatin substrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive pharmaceutical landscape. Our team of expert process chemists has extensively evaluated the iron-catalyzed cyclization method described in patent CN111675662B and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest international standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your timeline and reduce your overall project costs.
