Advanced One-Pot Synthesis of Biaryl Compounds for Scalable Pharmaceutical Intermediate Manufacturing
Advanced One-Pot Synthesis of Biaryl Compounds for Scalable Pharmaceutical Intermediate Manufacturing
The rapid evolution of modern medicinal chemistry demands increasingly efficient and robust methodologies for constructing complex molecular architectures, particularly the ubiquitous biaryl motif found in countless active pharmaceutical ingredients. A groundbreaking approach detailed in Chinese Patent CN113402350A introduces a novel preparation method for biaryl compounds that fundamentally shifts the paradigm from traditional cross-coupling strategies. This technology leverages the direct reductive cross-coupling of aryl quaternary ammonium salts with aryl bromides, utilizing a unique activation system comprising magnesium chips, lithium chloride, and a palladium catalyst. By operating under mild conditions ranging from 25 to 60 degrees Celsius in a one-pot configuration, this invention addresses critical pain points regarding substrate stability and operational safety. For R&D directors and process chemists seeking reliable pathways to high-purity intermediates, this patent offers a compelling solution that merges academic innovation with industrial practicality, ensuring that the synthesis of these valuable building blocks is both economically viable and technically superior.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biaryl scaffolds has relied heavily on the use of pre-formed organometallic nucleophiles, such as Grignard reagents, organolithiums, or organozinc species, which must be generated prior to the coupling event. These traditional methodologies impose severe logistical and safety burdens on chemical manufacturing facilities, primarily because these reagents are notoriously sensitive to atmospheric moisture and oxygen, often requiring cryogenic temperatures and strictly anhydrous environments to prevent decomposition. Furthermore, the preparation of these nucleophiles frequently involves hazardous exothermic reactions and the use of pyrophoric materials, which significantly escalates the risk profile during commercial scale-up. The necessity for specialized equipment to handle air-sensitive intermediates not only inflates capital expenditure but also complicates supply chain logistics, as these reagents often have limited shelf lives and cannot be easily stored or transported. Consequently, the reliance on such fragile intermediates creates a bottleneck in the production of pharmaceutical intermediates, where consistency and safety are paramount, driving up the overall cost of goods and extending lead times for critical drug substances.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in Patent CN113402350A utilizes stable and readily available aryl quaternary ammonium salts as electrophilic partners, effectively bypassing the need for pre-generated organometallic species. This innovative strategy employs a direct cross-coupling mechanism where the aryl quaternary ammonium salt reacts directly with an aryl bromide in the presence of a magnesium/lithium chloride activation system and a palladium catalyst. The reaction proceeds efficiently in tetrahydrofuran at ambient to moderately elevated temperatures, demonstrating exceptional functional group tolerance that allows for the preservation of sensitive moieties such as esters, nitriles, and halogens. 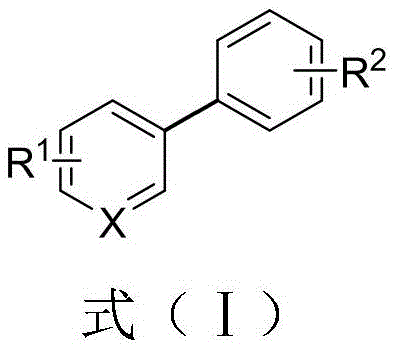 By eliminating the preliminary step of organometallic formation, this approach drastically simplifies the synthetic workflow, reducing the number of unit operations and minimizing waste generation. For procurement managers, this translates to a significant reduction in raw material complexity, as stable solid salts replace hazardous liquid reagents, thereby enhancing the overall reliability and safety of the supply chain for high-value chemical intermediates.
By eliminating the preliminary step of organometallic formation, this approach drastically simplifies the synthetic workflow, reducing the number of unit operations and minimizing waste generation. For procurement managers, this translates to a significant reduction in raw material complexity, as stable solid salts replace hazardous liquid reagents, thereby enhancing the overall reliability and safety of the supply chain for high-value chemical intermediates.
Mechanistic Insights into Palladium-Catalyzed Reductive Cross-Coupling
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst and the magnesium-lithium chloride activation system, which facilitates the in situ generation of reactive species without the pitfalls of traditional Grignard formation. The mechanism likely involves the oxidative addition of the aryl bromide to the palladium center, followed by a transmetallation step mediated by the activated magnesium species generated from the Mg/LiCl mixture. The presence of N,N,N',N'-tetramethylethylenediamine (TMEDA) acts as a crucial ligand that stabilizes the magnesium species and enhances its solubility in the organic phase, ensuring a homogeneous and efficient reaction environment. This synergistic catalytic cycle allows for the cleavage of the strong carbon-nitrogen bond in the quaternary ammonium salt, a transformation that is typically challenging under standard conditions. 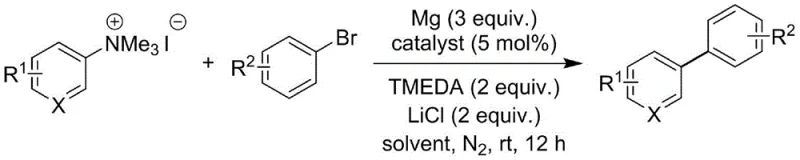 Furthermore, the system exhibits remarkable chemoselectivity, tolerating a wide array of functional groups including methoxy, trifluoromethyl, cyano, and ester groups, which are often incompatible with harsher organometallic conditions. This high level of selectivity ensures that the final biaryl products maintain high purity profiles, minimizing the formation of side products and simplifying downstream purification processes, which is a critical consideration for R&D teams focused on impurity control and regulatory compliance.
Furthermore, the system exhibits remarkable chemoselectivity, tolerating a wide array of functional groups including methoxy, trifluoromethyl, cyano, and ester groups, which are often incompatible with harsher organometallic conditions. This high level of selectivity ensures that the final biaryl products maintain high purity profiles, minimizing the formation of side products and simplifying downstream purification processes, which is a critical consideration for R&D teams focused on impurity control and regulatory compliance.
From an impurity control perspective, the mild nature of this reaction conditions prevents the degradation of sensitive functional groups that often leads to complex impurity profiles in traditional methods. The use of aryl bromides and quaternary salts, which are chemically robust, minimizes the risk of hydrolysis or oxidation side reactions that are common when handling highly reactive organolithium or Grignard reagents. Additionally, the specific optimization of the catalyst loading at 5 mol% and the precise molar ratios of reactants ensure that the reaction proceeds to completion with minimal residual starting materials. The patent data indicates that alternative catalysts such as iron or nickel chlorides result in negligible yields, underscoring the specificity of the palladium system in driving this transformation. This mechanistic precision allows for the consistent production of high-purity biaryl compounds, reducing the burden on analytical teams to identify and quantify trace impurities, and ultimately accelerating the timeline for process validation and technology transfer in pharmaceutical manufacturing.
How to Synthesize Biaryl Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating diverse biaryl structures, leveraging the stability of quaternary ammonium salts to streamline the workflow. The process begins with the activation of magnesium turnings using lithium chloride under reduced pressure, a critical step that ensures the metal surface is highly reactive for the subsequent coupling event. Following this activation, the reaction is conducted in a Schlenk tube under an inert nitrogen atmosphere, where ultra-dry tetrahydrofuran serves as the solvent to maintain the integrity of the reactive intermediates. The detailed procedural steps involve precise stoichiometric control, utilizing a 1:2 molar ratio of the aryl quaternary ammonium salt to the aryl bromide to drive the equilibrium towards the desired product.
- Activate magnesium chips by heating with lithium chloride under reduced pressure at 310-330°C for 2-4 minutes, then cool to room temperature.
- In a Schlenk tube under nitrogen, combine the activated magnesium mixture with ultra-dry THF, aryl quaternary ammonium salt, 5 mol% palladium catalyst, aryl bromide, and TMEDA, stirring at 25-60°C for 6-12 hours.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers transformative advantages by fundamentally altering the cost structure and risk profile of biaryl intermediate production. The primary economic driver is the substitution of expensive and hazardous pre-formed organometallic reagents with inexpensive, shelf-stable aryl bromides and quaternary ammonium salts, which are commodity chemicals available from multiple global suppliers. This shift eliminates the need for specialized storage infrastructure required for pyrophoric materials and reduces the dependency on single-source vendors for sensitive reagents, thereby enhancing supply chain resilience. Moreover, the simplified one-pot procedure reduces the total processing time and labor costs associated with multi-step sequences, leading to substantial operational efficiencies. By avoiding the generation of large volumes of hazardous waste associated with quenching excess organometallics, the process also aligns with increasingly stringent environmental regulations, potentially lowering waste disposal costs and improving the sustainability metrics of the manufacturing site.
- Cost Reduction in Manufacturing: The elimination of pre-formed organometallic reagents removes a significant cost center related to both the purchase of specialized reagents and the safety measures required to handle them. Since the reaction utilizes cheap magnesium chips and lithium chloride as activators instead of expensive organozinc or organoboron species, the raw material cost per kilogram of product is drastically lowered. Furthermore, the high yield and selectivity reported in the patent minimize the loss of valuable starting materials, ensuring that the atom economy of the process is optimized for commercial viability. This efficiency translates directly into a lower cost of goods sold, allowing manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates without compromising on quality or margin.
- Enhanced Supply Chain Reliability: The reliance on stable solid reagents like aryl quaternary ammonium iodides and aryl bromides ensures a consistent and uninterrupted supply of raw materials, as these compounds are not subject to the degradation issues that plague liquid organometallics. This stability allows for bulk purchasing and long-term storage, buffering the production schedule against market fluctuations or delivery delays. Additionally, the robustness of the reaction conditions means that the process is less susceptible to variations in ambient humidity or temperature, reducing the likelihood of batch failures and ensuring a steady output of material. For supply chain heads, this reliability is crucial for maintaining just-in-time inventory levels and meeting the rigorous delivery schedules demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction temperatures of 25 to 60 degrees Celsius and the use of common solvents like tetrahydrofuran make this process inherently scalable from gram to ton quantities without requiring exotic engineering controls. The absence of pyrophoric reagents significantly lowers the fire and explosion hazards associated with large-scale chemical manufacturing, simplifying the permitting process and reducing insurance premiums. From an environmental standpoint, the reduction in hazardous waste generation and the use of recyclable solvents contribute to a greener manufacturing footprint, aligning with corporate sustainability goals. This scalability ensures that the technology can seamlessly transition from pilot plant studies to full commercial production, supporting the growing demand for complex biaryl structures in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biaryl synthesis technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are designed to clarify the operational parameters and strategic benefits for stakeholders evaluating this method for their own manufacturing pipelines. Understanding the specific advantages of this catalyst system and substrate scope is essential for making informed decisions about process adoption and resource allocation.
Q: What are the advantages of using aryl quaternary ammonium salts over traditional organometallic reagents?
A: Unlike traditional organometallic reagents like Grignard or organolithium compounds which are highly sensitive to air and moisture and require strict anhydrous conditions, aryl quaternary ammonium salts are stable, easy to handle, and commercially available. This patent demonstrates that they can be directly coupled with aryl halides without pre-activation, significantly simplifying the operational complexity and improving safety profiles in industrial settings.
Q: Which catalyst system provides the highest yield for this biaryl synthesis?
A: According to the optimization data in the patent, bis(triphenylphosphine)palladium dichloride serves as the superior catalyst compared to iron, cobalt, or nickel alternatives. When combined with magnesium chips, lithium chloride, and TMEDA in tetrahydrofuran at 25°C, this system achieves optimal yields (up to 90%) while maintaining excellent functional group tolerance for substrates containing esters, halogens, and ethers.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to its mild reaction conditions (25-60°C) and the use of stable, non-pyrophoric reagents. The elimination of sensitive organometallic pre-preparation steps reduces the risk profile associated with large-scale manufacturing, making it an ideal candidate for the commercial scale-up of complex biaryl intermediates required in API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biaryl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation pharmaceuticals and agrochemicals. Our team of expert process chemists has extensively evaluated the technology disclosed in Patent CN113402350A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative method to life. We are committed to delivering high-purity biaryl compounds that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed couplings, ensuring that the benefits of this mild and efficient process are fully realized in a GMP-compliant environment.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this stable reagent system for your supply chain. We encourage you to contact us today to obtain specific COA data for our biaryl portfolio and to receive comprehensive route feasibility assessments that demonstrate our capability to support your long-term manufacturing goals with reliability and excellence.
