Scalable Copper-Catalyzed Synthesis of Oxaallylamine Derivatives for Advanced Pharmaceutical Manufacturing
Scalable Copper-Catalyzed Synthesis of Oxaallylamine Derivatives for Advanced Pharmaceutical Manufacturing
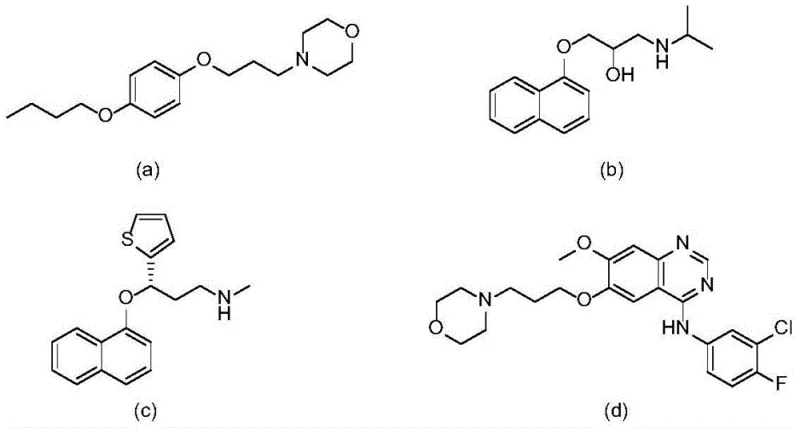
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable processes. A significant breakthrough in this domain is detailed in patent CN112521289A, which discloses a novel preparation method for oxaallylamine compounds. These compounds serve as critical building blocks in the synthesis of biologically active molecules, including well-known pharmaceuticals such as Pramocaine hydrochloride, Propranolol, Duloxetine, and Gefitinib. The structural diversity and biological relevance of these target molecules underscore the importance of developing robust synthetic routes for their precursors. As illustrated in the reference structures, the ability to construct complex amine frameworks efficiently is paramount for modern drug discovery and development pipelines.
This patent introduces a transformative strategy that leverages copper salt catalysis to facilitate the hydroamination of allene ethers. Unlike conventional methods that often suffer from limited substrate scope or harsh reaction conditions, this new approach operates under mild thermal conditions, typically between 40°C and 60°C. The methodology not only simplifies the operational workflow but also enhances the overall atom utilization rate, aligning perfectly with the principles of green chemistry. For R&D directors and process chemists, this represents a viable pathway to access high-purity intermediates with reduced impurity profiles, thereby streamlining downstream purification efforts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allylamine and oxaallylamine compounds has relied heavily on transition metal-catalyzed reactions such as direct allylic amination of allyl alcohols or vinylation of amines. While these strategies have established a foundation in organic synthesis, they are fraught with significant limitations when applied to complex molecule construction. Traditional methods often exhibit poor reaction selectivity, leading to the formation of unwanted by-products that complicate purification and reduce overall yield. Furthermore, many existing protocols require expensive catalysts, stringent anhydrous conditions, or elevated temperatures that can degrade sensitive functional groups present in advanced intermediates. The limited applicability of substrates in older methods restricts the chemical space that medicinal chemists can explore, often necessitating lengthy multi-step sequences to achieve the desired molecular architecture.
The Novel Approach
In stark contrast, the method disclosed in CN112521289A offers a streamlined, one-step synthesis of oxaallylamine compounds directly from allene ethers and amines. The core innovation lies in the use of a copper salt catalyst, specifically copper acetate, in conjunction with a zinc chloride additive. This catalytic system activates the allene ether efficiently, allowing for a smooth hydroamination reaction that proceeds with high regioselectivity. The general reaction scheme demonstrates the versatility of this approach, accommodating a wide array of substituents on both the ether and amine components. By utilizing simple and readily available starting materials, this novel route drastically reduces the complexity of the supply chain and lowers the barrier to entry for producing these valuable intermediates.

Mechanistic Insights into Copper-Catalyzed Hydroamination
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis. The reaction initiates with the activation of the allene ether by the copper catalyst, forming a reactive copper-amine intermediate. This species then undergoes a coordinated insertion into the allene system, generating a key alkenyl copper intermediate. Subsequently, the free amine acts as a nucleophile, attacking the activated complex to form the final carbon-nitrogen bond. This concerted mechanism ensures that the double bond geometry is preserved or controlled, resulting in the specific oxaallylamine structure. The presence of the zinc chloride additive plays a crucial role in stabilizing the transition states and enhancing the electrophilicity of the substrate, thereby accelerating the reaction rate without compromising selectivity.
From an impurity control perspective, this mechanism offers distinct advantages. The high specificity of the copper-catalyzed cycle minimizes side reactions such as polymerization of the allene or over-alkylation of the amine. The mild reaction temperature range of 40-60°C further suppresses thermal degradation pathways that are common in high-energy processes. Consequently, the crude reaction mixture contains fewer impurities, which simplifies the subsequent isolation steps. For quality control teams, this means that achieving the stringent purity specifications required for pharmaceutical intermediates becomes a more predictable and manageable task, reducing the risk of batch failures during scale-up.
How to Synthesize Oxaallylamine Compounds Efficiently
The practical execution of this synthesis is designed for ease of operation in both laboratory and pilot plant settings. The procedure involves dissolving the copper catalyst and additive in a common organic solvent such as dichloromethane or tetrahydrofuran, followed by the addition of the amine and allene ether substrates. The reaction mixture is then stirred at moderate temperatures for a duration of 5 to 10 hours. Workup involves standard aqueous extraction and drying, followed by purification via column chromatography or crystallization. The detailed standardized synthesis steps are outlined below to ensure reproducibility and safety.
- Prepare the reaction system by adding copper acetate catalyst and zinc chloride additive into a reactor containing an organic solvent such as dichloromethane.
- Introduce the allene ether substrate and the amine reactant into the mixture, ensuring precise molar ratios for optimal conversion efficiency.
- Stir the reaction at mild temperatures between 40-60°C for 5-10 hours, followed by extraction and purification via thin layer chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits. The shift towards this copper-catalyzed method addresses several pain points associated with traditional manufacturing, particularly regarding raw material availability and process safety. By relying on commodity chemicals like allene ethers and simple amines, the dependency on exotic or scarce reagents is eliminated. This stability in raw material sourcing translates directly into enhanced supply chain reliability, ensuring that production schedules are not disrupted by vendor shortages or price volatility in the specialty chemical market.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive noble metal catalysts and the reduction of energy consumption. Operating at mild temperatures significantly lowers utility costs compared to high-temperature reflux processes. Furthermore, the high atom economy of the reaction means that a greater proportion of the starting mass is converted into the desired product, reducing waste disposal costs. The simplified purification process also contributes to substantial cost savings by shortening the overall production cycle time and minimizing solvent usage.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in substrate structure allows for a more flexible manufacturing strategy. Facilities can produce a diverse portfolio of oxaallylamine derivatives using the same core equipment and protocol, reducing the need for specialized reactors. This flexibility ensures that the supply of critical intermediates remains continuous even when demand shifts between different drug candidates. The use of stable, non-hazardous reagents also simplifies logistics and storage requirements, further securing the supply chain against regulatory or transportation bottlenecks.
- Scalability and Environmental Compliance: From an environmental standpoint, this process aligns with increasingly strict global regulations on chemical manufacturing. The high selectivity reduces the generation of hazardous by-products, easing the burden on waste treatment facilities. The ability to scale this reaction from gram to kilogram quantities without loss of efficiency demonstrates its readiness for commercial production. This scalability ensures that as a drug candidate moves from clinical trials to market, the manufacturing process can grow seamlessly to meet demand without requiring a complete process redevelopment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxaallylamine synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of what partners can expect when integrating this chemistry into their operations.
Q: What are the primary advantages of this copper-catalyzed method over traditional allylic amination?
A: This novel approach utilizes readily available allene ethers and operates under significantly milder conditions (40-60°C) compared to harsh traditional methods, offering superior atom economy and broader substrate tolerance without requiring expensive transition metals like palladium.
Q: Can this synthesis method be scaled for industrial production of drug intermediates?
A: Yes, the process demonstrates excellent scalability potential due to its use of common organic solvents, simple workup procedures involving standard extraction, and high yields across diverse substrates, making it suitable for commercial scale-up.
Q: What types of amine substrates are compatible with this hydroamination reaction?
A: The method exhibits remarkable functional group tolerance, successfully reacting with a wide range of amines including aromatic amines, aliphatic amines, cyclic amines like morpholine, and even bulky structures like carbazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaallylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the copper-catalyzed hydroamination described in CN112521289A. Our team of expert process chemists is dedicated to translating such academic and patent breakthroughs into robust, commercial-scale manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of oxaallylamine intermediate delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
