Optimizing Cefotiam Hexetil Hydrochloride Production: A High-Yield, Low-Impurity Strategy for Global Antibiotic Manufacturers
The global demand for second-generation cephalosporins continues to drive innovation in the synthesis of key oral antibiotic intermediates. Patent CN101948476A discloses a robust and highly efficient method for preparing and purifying cefotiam hexetil hydrochloride, a critical prodrug used in the formulation of oral cephalosporin antibiotics. This technology addresses long-standing challenges in the industry, specifically the issues of low yield, high isomer content, and undesirable color development during the esterification of cefotiam. By introducing a novel purification strategy that replaces traditional solid-phase decolorization with liquid-phase reductive washing, this process offers a pathway to significantly higher purity and operational efficiency. For pharmaceutical manufacturers seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this synthetic route is essential for securing a stable supply chain of high-quality antibiotic precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefotiam hexetil has been plagued by inefficiencies that hinder large-scale commercial viability. Early literature, such as methods described in The Journal of Antibiotics (1986), reported yields as low as 20% or suffered from excessive formation of the Delta-2 isomer, often exceeding the 2% limit set by pharmacopoeial standards. Subsequent improvements, such as those found in patent CN101619069, managed to control isomer content but still struggled with yields around 60% and significant product loss during the purification stages. A major bottleneck in these conventional processes is the reliance on activated carbon for decolorization. The introduction of solid adsorbents like activated carbon necessitates additional filtration steps, which not only complicates the workflow but also leads to substantial mechanical loss of the valuable product trapped within the filter cake. Furthermore, the instability of the intermediate often results in pink or faint yellow discoloration, indicating oxidative degradation that compromises the aesthetic and chemical quality of the final active pharmaceutical ingredient.
The Novel Approach
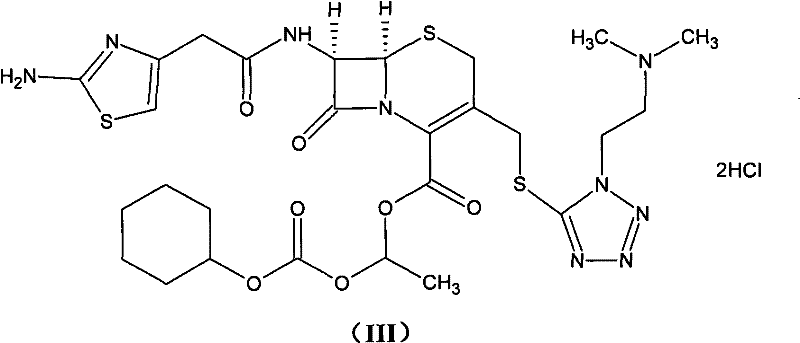
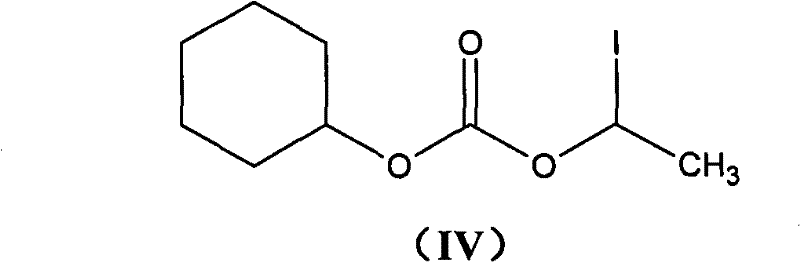
The methodology outlined in CN101948476A represents a paradigm shift in how cefotiam hexetil hydrochloride is manufactured, focusing on chemical intervention rather than physical adsorption to manage impurities. Instead of relying on activated carbon, this novel approach integrates water-soluble reducing agents, such as sodium thiosulfate, sodium sulfite, or sodium bisulfite, directly into the extraction and washing phases. This chemical strategy effectively scavenges oxidative species responsible for color development without introducing solid particulates that require removal. The process begins with the formation of a stable cefotiam salt using bicarbonates, followed by a controlled esterification with 1-iodoethyl cyclohexyl carbonate. By maintaining strict temperature controls during the reaction and utilizing a mixed solvent system for crystallization, the method achieves yields exceeding 95% and purity levels greater than 98.5%. This transition from physical filtration to chemical reduction drastically simplifies the downstream processing, making it an ideal solution for cost reduction in antibiotic manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Bicarbonate-Mediated Esterification and Reductive Workup
The core of this synthetic advancement lies in the precise management of the cefotiam nucleophile and the electrophilic carbonate intermediate. The process initiates by converting cefotiam into its corresponding salt (Formula V) using acid carbonates like potassium bicarbonate (KHCO3) or sodium bicarbonate (NaHCO3) in a water-acetone mixture. This salt formation enhances the nucleophilicity of the carboxylic acid group while maintaining the stability of the beta-lactam ring, which is sensitive to harsh basic conditions. The subsequent esterification involves the reaction of this salt with 1-iodoethyl cyclohexyl carbonate (Formula IV), a highly reactive alkylating agent. The reaction is conducted in polar aprotic solvents such as DMF or DMA at low temperatures ranging from -15°C to 10°C. This thermal control is critical; it minimizes the kinetic energy available for side reactions, thereby suppressing the formation of the unwanted Delta-2 isomer and preserving the stereochemical integrity of the cephalosporin nucleus.
Following the esterification, the workup procedure employs a sophisticated liquid-liquid extraction protocol designed to eliminate color bodies without product loss. The reaction mixture is treated with an organic solvent and an aqueous solution containing a reducing agent. The reducing agent acts as a sacrificial electron donor, neutralizing trace oxidants or quinone-like impurities that would otherwise polymerize to form colored complexes. This is followed by an acid extraction to isolate the cefotiam hexetil hydrochloride into the aqueous phase, separating it from non-basic organic impurities. The aqueous layer is then basified to regenerate the free base, which is extracted back into an organic solvent. Finally, the product is converted back to the hydrochloride salt and crystallized from a mixed solvent system, typically involving acetone, isopropanol, or acetonitrile. This multi-step extraction and crystallization sequence ensures that the final product is not only chemically pure but also visually white, meeting the stringent aesthetic requirements of modern pharmaceutical standards.
How to Synthesize Cefotiam Hexetil Hydrochloride Efficiently
The synthesis of this high-value intermediate requires careful attention to solvent selection, temperature gradients, and the timing of reagent addition to maximize yield and minimize isomerization. The patented route provides a clear roadmap for transitioning from laboratory scale to commercial production, emphasizing the use of readily available reagents and standard unit operations. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers aiming to implement this technology. For detailed standardized synthesis steps, please refer to the guide below.
- Prepare cefotiam salt by reacting cefotiam with acid bicarbonate (KHCO3 or NaHCO3) in a water-acetone mixed solvent system.
- Perform esterification by reacting the cefotiam salt with 1-iodoethyl cyclohexyl carbonate in DMF or DMA at low temperatures (-15 to 10°C).
- Execute a reductive workup using water-soluble reducing agents (e.g., sodium bisulfite) during extraction to prevent color development, followed by acid-base extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refined synthesis route offers tangible strategic benefits that extend beyond simple technical metrics. The elimination of activated carbon filtration removes a significant bottleneck in the production line, reducing the requirement for specialized filtration equipment and the associated downtime for filter changes and cleaning. This streamlining of the process flow directly translates to shorter batch cycle times, allowing manufacturing facilities to increase throughput without expanding their physical footprint. Moreover, the use of common, commodity-grade reagents such as bicarbonates and thiosulfates ensures that the supply chain remains resilient against fluctuations in the availability of exotic catalysts or specialized purification media. This reliability is crucial for maintaining continuous production schedules in the highly regulated antibiotic market.
- Cost Reduction in Manufacturing: The most significant economic advantage of this method is the dramatic improvement in overall process yield. By avoiding the mechanical losses associated with activated carbon treatment and optimizing the crystallization recovery, the process achieves yields significantly higher than historical benchmarks. This efficiency means that less raw material is required to produce the same amount of finished goods, directly lowering the cost of goods sold (COGS). Additionally, the simplified workup reduces the consumption of auxiliary materials and solvents, further contributing to substantial cost savings. The ability to produce a white, high-purity product without extensive reprocessing also minimizes the risk of batch rejection, protecting the manufacturer from the financial impact of failed quality control tests.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances the predictability of supply deliveries. Because the process relies on stable intermediates and avoids sensitive catalytic steps that might require inert atmospheres or ultra-dry conditions, it is less prone to unexpected deviations or batch failures. The use of water-soluble reducing agents effectively manages the stability of the product during workup, ensuring consistent quality across different production runs. This consistency allows supply chain planners to forecast inventory levels with greater confidence, reducing the need for excessive safety stock. Furthermore, the scalability of the method ensures that suppliers can respond rapidly to surges in demand for oral cephalosporins, securing the continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner production profile. The replacement of solid waste (spent activated carbon) with aqueous waste streams containing biodegradable sulfites simplifies waste management and disposal protocols. The process utilizes standard organic solvents that can be efficiently recovered and recycled through distillation, aligning with green chemistry principles and reducing the facility's environmental footprint. The high purity of the final product also reduces the burden on downstream purification steps in the formulation of the final drug product. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for companies looking to modernize their manufacturing capabilities while adhering to increasingly strict global regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of cefotiam hexetil hydrochloride using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader pharmaceutical portfolios.
Q: How does this method control the Delta-2 isomer impurity?
A: The method utilizes specific low-temperature esterification conditions (-10 to -5°C) and precise solvent ratios during crystallization, which effectively suppresses the formation of the Delta-2 isomer, keeping total impurities below 2%.
Q: Why are water-soluble reducing agents preferred over activated carbon?
A: Traditional methods use activated carbon to remove color, which introduces solid filtration steps and significant product adsorption losses. Water-soluble reducing agents like sodium bisulfite chemically eliminate color-causing oxidants in the liquid phase, simplifying separation and improving yield.
Q: What is the expected purity of the final hydrochloride salt?
A: According to the patent data, this optimized process consistently produces cefotiam hexetil hydrochloride with a purity exceeding 98.5%, often reaching up to 99% in optimized embodiments, surpassing Japanese Pharmacopoeia standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Hexetil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of cephalosporin synthesis demands a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global pharmaceutical giants. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cefotiam hexetil hydrochloride meets or exceeds international pharmacopoeial standards. Our facility is equipped to handle the specific solvent systems and low-temperature reactions required by this patent, delivering a product that is both chemically superior and visually pristine.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By leveraging our optimized processes, we can help you reduce lead time for high-purity pharmaceutical intermediates while minimizing overall manufacturing costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to secure a sustainable and efficient supply chain for your next-generation antibiotic formulations.
