Optimizing Cefotiam Hexetil Hydrochloride Production: A Technical Breakthrough in Oral Cephalosporin Manufacturing
The pharmaceutical industry constantly seeks robust manufacturing routes for critical oral antibiotics, and the refining process detailed in patent CN102127093A represents a significant advancement in the production of Cefotiam hexetil hydrochloride. This compound serves as a vital prodrug, rapidly hydrolyzing into the active antibiotic cefotiam upon ingestion, offering broad-spectrum efficacy against Gram-positive and Gram-negative bacteria including resistant strains. The disclosed technology addresses long-standing inefficiencies in industrial synthesis, specifically targeting the high costs and complex purification protocols associated with traditional methods. By shifting from expensive iodine-based reagents to more accessible bromine-based alternatives and streamlining the isolation of the hydrochloride salt, this process offers a compelling value proposition for large-scale API manufacturing. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates that meet stringent international pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Cefotiam hexetil hydrochloride has been plagued by several critical bottlenecks that hinder cost-effective mass production. Traditional methodologies typically rely on the use of 1-iodoethyl cyclohexyl carbonate as the esterifying agent, a reagent that is not only prohibitively expensive due to the cost of iodine but also challenging to synthesize with high consistency. Furthermore, conventional processes often suffer from low overall yields, with bibliographic data suggesting total recovery rates hovering around merely 20%, which drastically inflates the cost of goods sold. The post-reaction treatment in these legacy methods is equally problematic, frequently necessitating laborious purification techniques such as column chromatography and multiple acid-base extractions to remove byproducts. These complex downstream processing steps not only extend production lead times but also introduce significant risks of product degradation and contamination, making them ill-suited for the rigorous demands of modern GMP-compliant facilities.
The Novel Approach
In stark contrast, the innovative process outlined in the patent introduces a streamlined three-step sequence that fundamentally alters the economic and operational landscape of production. The core breakthrough lies in the substitution of the costly iodo-reagent with 1-bromoethyl cyclohexyl carbonate, a structurally similar yet far more economical alkylating agent. This strategic switch allows for the direct esterification of cefotiam salts without the need for intermediate purification of the ester product, effectively collapsing multiple processing stages into a single efficient operation. The subsequent salting-out procedure utilizes dry hydrogen chloride gas in optimized organic solvents like ethyl acetate, facilitating the direct precipitation of the target hydrochloride salt. This approach eliminates the need for chromatographic separation entirely, resulting in a drastic simplification of the workflow, a significant boost in overall yield to over 60%, and a final product quality that consistently meets the rigorous specifications of the Japanese Pharmacopoeia.
Mechanistic Insights into Bromo-Esterification and Salt Formation
The chemical elegance of this synthesis rests on the precise reactivity of the cefotiam salt intermediate when exposed to the bromo-carbonate electrophile. By first converting cefotiam chloride into a potassium or sodium salt using mild carbonates in methanol, the nucleophilicity of the carboxylate group is enhanced, promoting a smoother attack on the electrophilic carbon of the bromo-ester. The use of tetrahydrofuran (THF) as the reaction solvent is critical, as its low boiling point facilitates easy removal under reduced pressure post-reaction, while its polarity supports the dissolution of the ionic salt species. The reaction is conducted at controlled low temperatures, typically around 3°C, to suppress side reactions and maintain the stereochemical integrity of the beta-lactam ring, which is highly susceptible to thermal degradation. This careful orchestration of reaction conditions ensures that the esterification proceeds with high conversion rates, minimizing the formation of regio-isomers and other structural impurities that often plague cephalosporin synthesis.
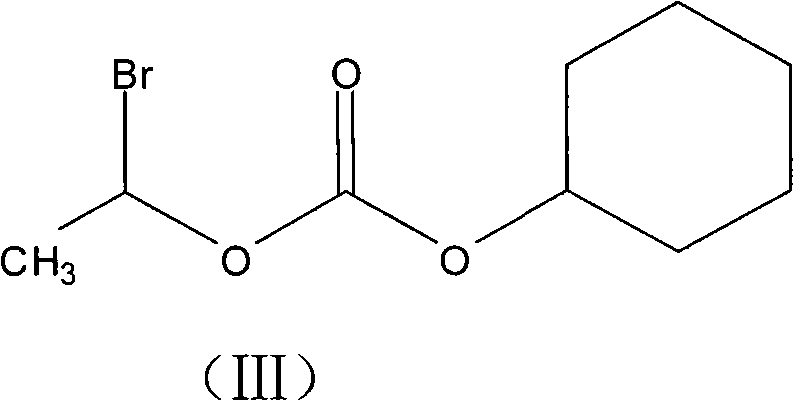
Following the esterification, the mechanism shifts to a physical-chemical separation driven by solubility differences. The crude cefotiam hexetil is dissolved in a selected organic solvent, with ethyl acetate proving superior to dichloromethane or acetonitrile for crystal growth. The introduction of dry hydrogen chloride gas creates an acidic environment that protonates the amine functionalities, forming the hydrochloride salt which has significantly lower solubility in the cold organic medium. This precipitation is not merely a phase change but a purification event; as the pure hydrochloride crystals lattice forms, many soluble organic impurities remain in the mother liquor. The final recrystallization from a methanol and petroleum ether mixture further refines the crystal lattice, ensuring that the isomer ratio (Aa/(Aa+Ab)) remains within the tight specification of approximately 0.49, thereby guaranteeing the biological efficacy and safety profile required for oral antibiotic formulations.
How to Synthesize Cefotiam Hexetil Hydrochloride Efficiently
The synthesis protocol described herein offers a reproducible framework for manufacturing high-purity cefotiam hexetil hydrochloride suitable for clinical and commercial applications. The process begins with the generation of the cefotiam salt, followed by the key esterification step using the bromo-reagent, and concludes with gas-phase salting out and recrystallization. This route is designed to maximize throughput while minimizing waste generation, aligning with green chemistry principles. For process chemists looking to implement this technology, strict adherence to temperature profiles and solvent ratios is paramount to achieving the reported yields and purity levels. The detailed standardized synthesis steps below outline the specific operational parameters required to replicate this success in a pilot or production plant setting.
- Preparation of Cefotiam Salt: React cefotiam chloride with potassium carbonate or sodium carbonate in methanol at 0°C to form the stable cefotiam potassium or sodium salt.
- Esterification Reaction: React the cefotiam salt with 1-bromoethyl cyclohexyl carbonate in THF solvent at low temperatures (approx. 3°C) using anhydrous carbonate as a catalyst.
- Salting Out and Purification: Dissolve the crude ester in ethyl acetate, feed dry hydrogen chloride gas at -5°C to precipitate the hydrochloride salt, and recrystallize using a methanol/petroleum ether mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refined synthesis route translates into tangible strategic advantages that extend beyond simple unit cost metrics. The elimination of column chromatography and the reduction of processing steps significantly lower the operational expenditure (OPEX) associated with solvent consumption, energy usage, and labor hours. By replacing the expensive iodo-based reagent with a bromine-based alternative, the raw material bill of materials (BOM) is substantially reduced, providing a buffer against market volatility in halogen pricing. Furthermore, the simplified workup procedure reduces the dependency on specialized purification equipment, allowing for greater flexibility in manufacturing site selection and capacity allocation. These factors collectively enhance the resilience of the supply chain, ensuring consistent availability of this critical antibiotic intermediate even during periods of high global demand.
- Cost Reduction in Manufacturing: The primary driver of cost efficiency in this process is the strategic substitution of raw materials and the removal of expensive purification stages. By utilizing 1-bromoethyl cyclohexyl carbonate instead of its iodo-counterpart, manufacturers avoid the premium pricing associated with iodine chemistry, which is often subject to supply constraints. Additionally, the ability to skip the purification of the intermediate ester and proceed directly to salting out eliminates the need for silica gel columns and vast quantities of elution solvents. This streamlining results in a drastic reduction in waste disposal costs and solvent recovery overheads, creating a leaner, more profitable manufacturing model that can withstand competitive pricing pressures in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply continuity is heavily dependent on the availability of key starting materials, and the shift to bromo-based reagents significantly de-risks the supply chain. Bromine derivatives are generally more commoditized and widely available than specialized iodo-compounds, reducing the likelihood of stockouts or long lead times from specialty chemical vendors. The robustness of the reaction conditions, which tolerate standard industrial solvents like ethyl acetate and THF, further ensures that production can be scaled across multiple facilities without requiring exotic or hard-to-source consumables. This reliability is crucial for maintaining uninterrupted production schedules for finished dosage forms, preventing costly delays in the distribution of essential oral antibiotics to healthcare providers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers a cleaner profile that aligns with increasingly stringent regulatory standards. The reduction in solvent usage and the elimination of solid waste generated by column chromatography media significantly lower the environmental footprint of the manufacturing process. The use of gas-phase hydrogen chloride for salting out is a well-established, scalable unit operation that can be easily controlled in large reactors, ensuring consistent product quality from kilogram to tonne scales. This ease of scale-up minimizes the technical risks associated with technology transfer, allowing for rapid capacity expansion to meet market surges while maintaining compliance with environmental discharge limits regarding organic solvents and heavy metal residues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining process. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on yield expectations, purity standards, and operational requirements. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: Why is 1-bromoethyl cyclohexyl carbonate preferred over the iodo-analog in this synthesis?
A: The patent highlights that the bromo-derivative is significantly cheaper and easier to prepare than the iodo-derivative traditionally used, leading to substantial raw material cost reductions without compromising reaction efficiency.
Q: How does this process improve product purity compared to conventional methods?
A: By eliminating complex column chromatography purification steps and utilizing a direct gas-phase salting out method followed by specific recrystallization, the process achieves purity levels meeting Japanese Pharmacopoeia standards with reduced byproduct content.
Q: What are the critical temperature controls required for the esterification step?
A: The esterification reaction requires strict temperature control, typically maintained between -5°C and 10°C (optimally around 3°C), to ensure high yield and prevent degradation of the sensitive beta-lactam structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Hexetil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial infections. Our technical team has extensively analyzed advanced synthesis routes like the one described in CN102127093A to ensure our manufacturing capabilities are aligned with the latest industry innovations. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot batches for clinical trials or bulk quantities for commercial launch, our facilities can deliver. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art HPLC and analytical instrumentation to verify that every batch meets the required isomer ratios and impurity profiles.
We invite pharmaceutical partners to collaborate with us to leverage these process efficiencies for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential clients to contact us directly to obtain specific COA data from recent batches and to discuss route feasibility assessments for integrating this refined cefotiam hexetil hydrochloride into your existing manufacturing portfolio. Let us be your partner in delivering safe, effective, and affordable antibiotic solutions to the market.
