Advanced Refining Technology for Cefotiam Hexetil Hydrochloride: Enhancing Yield and Purity for Commercial Scale-up
Advanced Refining Technology for Cefotiam Hexetil Hydrochloride: Enhancing Yield and Purity for Commercial Scale-up
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotic intermediates, and the refining process detailed in patent CN102127093B represents a significant leap forward for Cefotiam hexetil hydrochloride production. This oral cephalosporin prodrug is vital for treating various bacterial infections, yet its historical synthesis has been plagued by low efficiency and complex purification requirements. The disclosed technology introduces a streamlined approach that transitions from expensive iodo-ester reagents to more accessible bromo-esters, fundamentally altering the economic landscape of this API intermediate. By optimizing the esterification conditions and implementing a direct gas-phase salt formation strategy, this method achieves superior purity profiles that meet stringent Japanese Pharmacopoeia standards. For R&D directors and supply chain managers, understanding this shift from laborious chromatographic purification to efficient crystallization is key to securing a reliable pharmaceutical intermediates supplier partnership.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
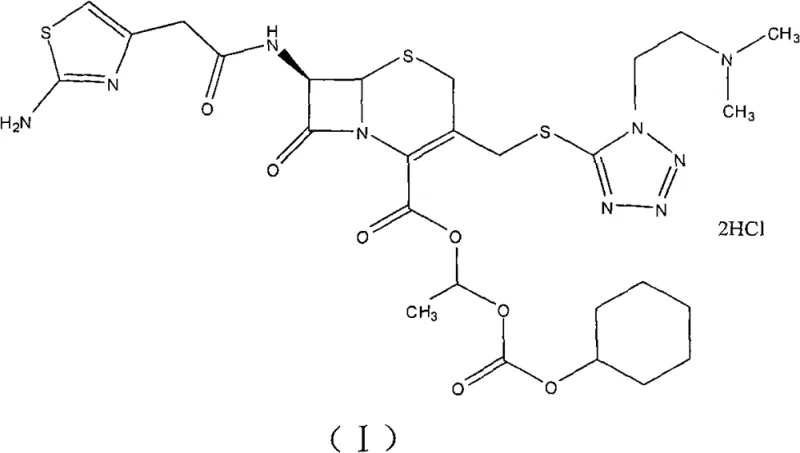
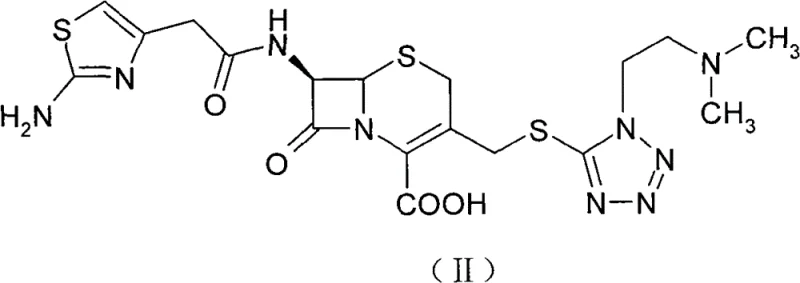
Historically, the industrial preparation of Cefotiam hexetil hydrochloride relied heavily on the direct esterification of Cefotiam (II) using 1-iodoethyl cyclohexyl carbonate. This traditional pathway suffers from severe economic and technical drawbacks that hinder large-scale viability. Foremost among these issues is the abysmal total recovery rate, which bibliographical data indicates hovers around merely 20%, rendering the production cost prohibitively high for competitive markets. Furthermore, the reliance on iodo-esters introduces significant raw material cost pressures, as iodine-containing reagents are inherently more expensive and less stable than their bromine counterparts. The post-reaction treatment is equally problematic, typically necessitating multiple extractions and tedious column chromatography to remove byproducts that often exceed 2%, failing to meet strict pharmacopoeial limits. These factors combine to create a fragile supply chain vulnerable to yield fluctuations and purification bottlenecks.
The Novel Approach
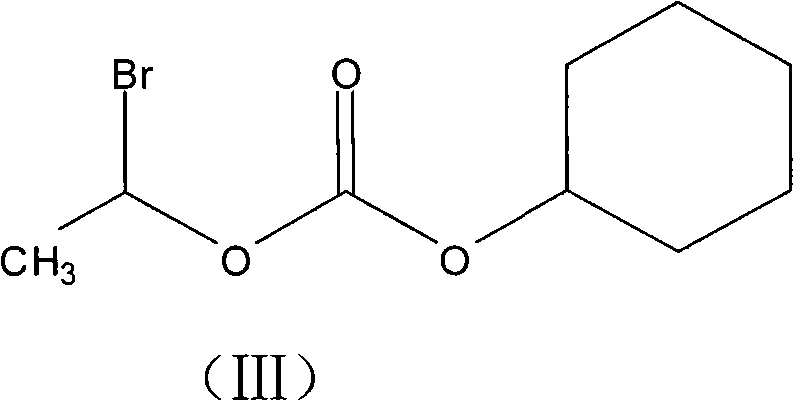
The innovative process described in the patent circumvents these historical inefficiencies by initiating the synthesis with Cefotiam salts (potassium or sodium) rather than the free acid form. This strategic modification enhances the nucleophilicity of the beta-lactam core, facilitating a smoother esterification reaction with the more economical 1-bromoethyl cyclohexyl carbonate (III). Crucially, the novel method eliminates the need for intermediate purification of the ester; the crude reaction mixture is directly subjected to salt formation using dry hydrogen chloride gas in an optimized organic solvent system. This telescoping of steps not only drastically reduces solvent consumption and processing time but also minimizes product loss associated with isolation. The final recrystallization from a methanol and petroleum ether mixture ensures the removal of residual impurities, delivering a high-purity product suitable for immediate formulation without further extensive cleaning.
Mechanistic Insights into Esterification and Salt Formation
The core chemical transformation relies on the nucleophilic attack of the Cefotiam carboxylate anion on the electrophilic carbon of the bromo-carbonate reagent. By pre-forming the Cefotiam potassium or sodium salt in methanol at controlled low temperatures (0°C), the reaction system ensures a high concentration of the reactive carboxylate species. The subsequent addition of anhydrous potassium or sodium carbonate in THF acts as an acid scavenger, neutralizing the HBr generated during the substitution reaction and driving the equilibrium toward the desired ester product. The choice of THF as the reaction solvent is critical due to its ability to dissolve both the ionic salt and the organic reagent while maintaining a low boiling point for easy removal. This mechanistic precision prevents the degradation of the sensitive beta-lactam ring, which is prone to hydrolysis under harsher acidic or basic conditions found in older methodologies.
Following esterification, the transition to the hydrochloride salt is governed by solubility dynamics and protonation kinetics. The patent highlights a rigorous solvent screening process where ethyl acetate emerged as the superior medium for precipitating the hydrochloride salt upon introduction of HCl gas. Unlike acetonitrile or THF, which resulted in muddy liquids or failed precipitation, ethyl acetate promotes the formation of well-defined crystalline solids at temperatures between -5°C and 0°C. This phase behavior is essential for impurity rejection, as the crystalline lattice of the hydrochloride salt selectively incorporates the target molecule while excluding structurally related byproducts. The final recrystallization step further refines the crystal habit and purity, ensuring that the isomeric ratio remains within the tight specification of roughly 0.49, consistent with high-quality therapeutic standards.
How to Synthesize Cefotiam Hexetil Hydrochloride Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for manufacturing high-purity Cefotiam hexetil hydrochloride suitable for GMP environments. The process begins with the conversion of Cefotiam chloride to its corresponding potassium salt, followed by esterification with the bromo-reagent in THF. The crude ester solution is then concentrated and redissolved in ethyl acetate for the critical salt formation step using dry HCl gas. Detailed operational parameters, including specific molar ratios, temperature controls, and solvent volumes, are essential for maximizing the yield which can reach over 60% in optimized embodiments. For a comprehensive breakdown of the exact experimental conditions and safety protocols required for implementation, please refer to the standardized guide below.
- Synthesize cefotiam potassium or sodium salt from cefotiam chloride and carbonate base in methanol at 0°C.
- Perform esterification of the cefotiam salt with 1-bromoethyl cyclohexyl carbonate in THF with anhydrous carbonate.
- Dissolve the crude ester in ethyl acetate, pass dry HCl gas to precipitate the hydrochloride salt, and recrystallize from methanol/petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining process translates directly into enhanced operational resilience and cost efficiency. The shift from iodo- to bromo-reagents addresses raw material volatility, leveraging the broader availability and lower price point of brominated intermediates to stabilize input costs. Furthermore, the elimination of column chromatography—a unit operation known for high solvent usage, low throughput, and scalability challenges—significantly simplifies the manufacturing workflow. This simplification allows for larger batch sizes and reduced cycle times, directly addressing the need for cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent quality requirements of the global market.
- Cost Reduction in Manufacturing: The replacement of expensive 1-iodoethyl cyclohexyl carbonate with the more economical 1-bromoethyl analog results in substantial raw material savings. Additionally, the removal of the purification column step drastically cuts down on silica gel and solvent expenses, which are major cost drivers in traditional cephalosporin synthesis. By telescoping the esterification and salt formation steps, the process reduces energy consumption and labor hours, creating a leaner production model that improves overall margin potential.
- Enhanced Supply Chain Reliability: The robustness of the new crystallization-based purification method ensures consistent batch-to-batch quality, reducing the risk of out-of-specification releases that can disrupt supply. The use of common industrial solvents like ethyl acetate and methanol, rather than specialized chromatographic eluents, simplifies logistics and inventory management. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical formulators.
- Scalability and Environmental Compliance: Transitioning from chromatography to crystallization significantly improves the environmental footprint of the process by reducing hazardous waste generation and solvent emissions. Crystallization is inherently more scalable than chromatography, allowing for seamless expansion from pilot plant to multi-ton commercial production without the need for complex equipment modifications. This scalability supports long-term supply agreements and aligns with modern green chemistry initiatives increasingly demanded by regulatory bodies and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing production lines.
Q: Why is 1-bromoethyl cyclohexyl carbonate preferred over the iodo-ester in this synthesis?
A: The bromo-ester reagent is significantly more cost-effective and easier to source industrially compared to the iodo-analog, while maintaining high reactivity for the esterification step.
Q: How does this process improve yield compared to conventional methods?
A: By utilizing the cefotiam salt directly and eliminating complex purification steps like column chromatography, the total yield increases from approximately 20% to over 50%.
Q: What solvents are critical for the salt formation step?
A: Ethyl acetate is the preferred solvent for dissolving the crude ester prior to HCl gas treatment, as it facilitates optimal crystallization of the hydrochloride salt compared to acetonitrile or THF.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Hexetil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes for complex beta-lactam antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical manufacturing setting. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Cefotiam hexetil hydrochloride meets the exacting standards required for oral antibiotic formulations. Our commitment to process optimization allows us to deliver high-quality intermediates that support your drug development and commercialization timelines.
We invite you to engage with our technical procurement team to discuss how this advanced refining process can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical partner available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
