Advanced Manufacturing of High-Purity Cefotiam Hexetil Hydrochloride for Global Pharma Supply Chains
Advanced Manufacturing of High-Purity Cefotiam Hexetil Hydrochloride for Global Pharma Supply Chains
The global demand for second-generation cephalosporins continues to drive innovation in antibiotic intermediate manufacturing, specifically regarding the oral prodrug Cefotiam Hexetil. Patent CN101948476A discloses a groundbreaking method for preparing and purifying Cefotiam Hexetil Hydrochloride that addresses critical historical challenges in yield and impurity profiles. This technology represents a significant leap forward for pharmaceutical manufacturers seeking a reliable Cefotiam Hexetil Hydrochloride supplier capable of delivering material that meets stringent international quality standards. The core innovation lies in a refined esterification process coupled with a unique purification strategy that utilizes water-soluble reducing agents to prevent product discoloration without the need for solid adsorbents. By shifting from traditional activation methods to a bicarbonate salt-mediated pathway, this process ensures superior stereochemical control and operational efficiency.
Cefotiam itself, while potent, suffers from poor oral bioavailability, necessitating its conversion into the hexetil ester form for effective clinical application. However, the chemical instability of the hexetil ester has historically forced the industry to adopt the hydrochloride salt form for final drug formulation. The structural evolution from the parent acid to the stable hydrochloride salt involves complex chemistry that must be meticulously managed to avoid degradation. 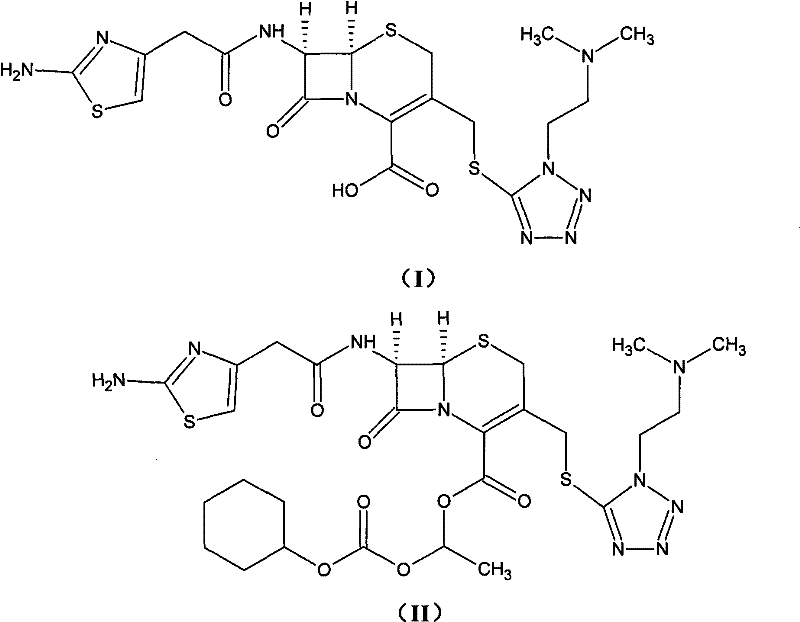 This transformation is not merely a functional group modification but a critical stabilization step that ensures the drug remains intact until absorption in the intestinal mucosa. The patent highlights that achieving a white, crystalline final product with minimal isomeric contamination is the key differentiator for high-quality API production.
This transformation is not merely a functional group modification but a critical stabilization step that ensures the drug remains intact until absorption in the intestinal mucosa. The patent highlights that achieving a white, crystalline final product with minimal isomeric contamination is the key differentiator for high-quality API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefotiam Hexetil has been plagued by inefficient reaction kinetics and difficult purification hurdles that severely impact commercial viability. Prior art methods, such as those described in early literature involving reactions in dimethylformamide (DMF), often resulted in unacceptably low yields, sometimes hovering around merely 20 percent in optimized attempts, and typically failing to exceed 60 percent in broader applications. A major technical bottleneck in these legacy processes is the uncontrolled formation of the Delta-2 isomer, a structural byproduct that frequently exceeds the 2 percent threshold mandated by the Japanese Pharmacopoeia, thereby rendering batches unsuitable for therapeutic use. Furthermore, the crude product obtained from these conventional routes invariably exhibits significant discoloration, ranging from faint yellow to pink, which traditionally necessitates the use of activated carbon for decolorization. This reliance on solid-phase adsorption introduces substantial processing complexity, including additional filtration steps that lead to unavoidable mechanical losses of the valuable active pharmaceutical ingredient.
The Novel Approach
The methodology outlined in CN101948476A fundamentally reengineers the synthesis pathway to overcome these entrenched inefficiencies through a sophisticated salt-formation and reductive purification strategy. Instead of direct esterification under harsh conditions, the process initiates by converting Cefotiam into a soluble bicarbonate salt using reagents like potassium bicarbonate or sodium bicarbonate in a mixed solvent system of water and acetone. This salt intermediate exhibits enhanced reactivity and stability, allowing for a cleaner esterification reaction with 1-iodoethyl cyclohexyl carbonate at controlled low temperatures. Crucially, the purification protocol integrates the addition of water-soluble reducing agents, such as sodium thiosulfate or sulfite, directly into the extraction solvent system. This innovative step quenches color-forming oxidative species in the liquid phase, eliminating the need for activated carbon treatment entirely. The result is a streamlined workflow that delivers a white crystalline product with yields consistently exceeding 95 percent and purity levels above 98.5 percent.
Mechanistic Insights into Bicarbonate-Mediated Esterification
The chemical elegance of this process lies in the precise manipulation of the carboxylic acid functionality on the Cefotiam nucleus. By reacting Cefotiam with an acid bicarbonate, the process generates a carboxylate salt (Structure V) that serves as a superior nucleophile compared to the free acid or traditional metal salts like potassium Cefotiam used in older methods. 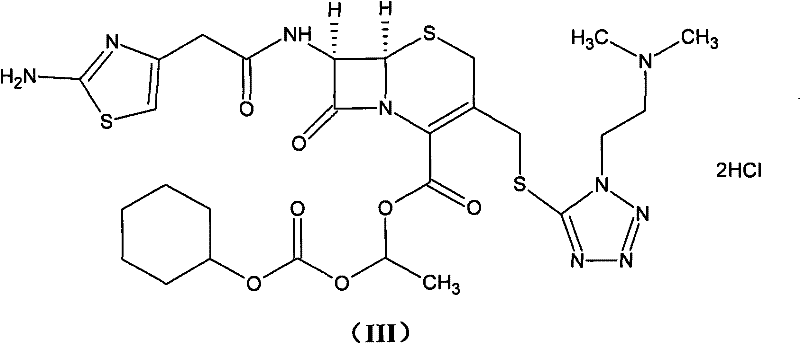 This salt formation occurs in a carefully balanced solvent mixture, typically water and acetone in a ratio of 1:2 to 1:3, which ensures complete dissolution while maintaining the stability of the beta-lactam ring. The subsequent esterification with 1-iodoethyl cyclohexyl carbonate (Structure IV) proceeds via an SN2-type mechanism where the carboxylate oxygen attacks the iodomethyl carbon. Conducting this reaction at temperatures between -10°C and -5°C is critical; it kinetically favors the formation of the desired ester bond while suppressing competing elimination reactions that would lead to the problematic Delta-2 isomer. This temperature control is a vital parameter for any contract development and manufacturing organization (CDMO) aiming to replicate this high-purity output.
This salt formation occurs in a carefully balanced solvent mixture, typically water and acetone in a ratio of 1:2 to 1:3, which ensures complete dissolution while maintaining the stability of the beta-lactam ring. The subsequent esterification with 1-iodoethyl cyclohexyl carbonate (Structure IV) proceeds via an SN2-type mechanism where the carboxylate oxygen attacks the iodomethyl carbon. Conducting this reaction at temperatures between -10°C and -5°C is critical; it kinetically favors the formation of the desired ester bond while suppressing competing elimination reactions that would lead to the problematic Delta-2 isomer. This temperature control is a vital parameter for any contract development and manufacturing organization (CDMO) aiming to replicate this high-purity output.
Beyond the primary reaction mechanism, the impurity control strategy employed during the workup phase is equally sophisticated and vital for ensuring the commercial quality of the API. In traditional syntheses, trace metal ions or oxidative byproducts generated during the reaction can catalyze the degradation of the cephalosporin core, leading to the characteristic yellowing of the product. The patented method counters this by introducing a reducing agent aqueous solution during the organic extraction phase. Agents like sodium bisulfite act as scavengers, neutralizing oxidizing species before they can attack the sensitive thiazole or beta-lactam moieties. Following extraction, the product is converted to its hydrochloride salt form through pH adjustment using hydrochloric acid, typically targeting a pH of 2 to 3. This acidification step not only stabilizes the molecule for storage but also facilitates the final crystallization from a mixed solvent system of acetonitrile, isopropanol, or acetone, ensuring the removal of residual solvents and trace organic impurities to meet rigorous regulatory specifications.
How to Synthesize Cefotiam Hexetil Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the specified solvent ratios and temperature profiles to maximize the economic and quality benefits. The process begins with the preparation of the key alkylating agent, 1-iodoethyl cyclohexyl carbonate, followed by the in situ generation of the Cefotiam bicarbonate salt. These two intermediates are then coupled under cryogenic conditions to form the ester, which is subsequently isolated through a liquid-liquid extraction protocol that incorporates the critical reducing agent wash. The final isolation involves a controlled crystallization step where the concentration of the solution and the cooling rate are managed to promote the growth of large, pure crystals.
- Preparation of Cefotiam Salt by reacting Cefotiam with acid bicarbonate (KHCO3 or NaHCO3) in a mixed solvent system of water and acetone.
- Esterification reaction between the prepared Cefotiam salt and 1-iodoethyl cyclohexyl carbonate in DMA or DMF at low temperatures (-10 to -5°C).
- Purification involving extraction with organic solvents containing water-soluble reducing agents to eliminate color, followed by acidification and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative advantages in terms of cost structure and supply reliability. The most immediate impact is observed in the drastic simplification of the downstream processing unit operations. By eliminating the requirement for activated carbon filtration, manufacturers remove a significant bottleneck that traditionally slows down batch turnover and increases equipment cleaning validation burdens. This streamlining translates directly into reduced operational expenditures and a more agile production schedule, allowing for faster response times to market demand fluctuations. Furthermore, the substantial increase in reaction yield, moving from historical averages of roughly 60 percent to over 95 percent, implies a massive reduction in the cost of goods sold (COGS) per kilogram of finished API. This efficiency gain means that less raw material is required to produce the same amount of active ingredient, providing a buffer against volatility in the pricing of starting materials like Cefotiam acid.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of expensive and loss-prone purification steps. Traditional decolorization using activated carbon not only consumes material but also traps a percentage of the product within the filter cake, representing a direct financial loss. By switching to a chemical reduction method using inexpensive inorganic salts like thiosulfate, the process achieves a whiter product with higher recovery rates. Additionally, the use of common industrial solvents such as acetone, ethyl acetate, and acetonitrile ensures that solvent procurement remains cost-effective and logistically simple. The high yield further amortizes the fixed costs of reactor time and labor over a larger output of saleable product, significantly enhancing the overall profit margin for the manufacturer.
- Enhanced Supply Chain Reliability: From a supply continuity perspective, the robustness of this synthetic route minimizes the risk of batch failures due to out-of-specification impurities. The tight control over the Delta-2 isomer content ensures that nearly every batch meets the strict criteria set by major pharmacopoeias, reducing the likelihood of costly rejections or re-processing. This consistency is paramount for maintaining uninterrupted supply lines to downstream formulation partners. Moreover, the scalability of the reaction conditions, which do not require exotic catalysts or extreme pressures, means that production can be easily ramped up from pilot scale to multi-ton commercial campaigns without significant re-engineering. This flexibility allows suppliers to better manage inventory levels and fulfill large-volume orders with shorter lead times.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is notably lower than that of conventional methods, aligning with the increasing regulatory pressure for greener pharmaceutical production. The avoidance of solid waste generation associated with spent activated carbon disposal simplifies waste management protocols and reduces hazardous waste handling costs. The solvent systems employed are amenable to standard recovery and recycling distillation columns, further minimizing volatile organic compound (VOC) emissions. The high atom economy resulting from the improved yield means less chemical waste is generated per unit of product. These factors collectively contribute to a more sustainable manufacturing profile, which is increasingly becoming a key criterion for selection by environmentally conscious multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefotiam Hexetil Hydrochloride using this advanced methodology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their supply chain. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for process development scientists.
Q: How does this method control the formation of the Delta-2 isomer?
A: The novel process utilizes a specific bicarbonate salt formation step and controlled low-temperature esterification conditions which significantly suppress the formation of the unwanted Delta-2 isomer, keeping total impurities below 2 percent.
Q: What is the advantage of using water-soluble reducing agents in purification?
A: Incorporating water-soluble reducing agents like thiosulfate or sulfite during the extraction phase effectively eliminates color-developing factors in situ, removing the need for activated carbon filtration and thereby reducing product loss.
Q: What purity levels can be achieved with this synthesis route?
A: This optimized manufacturing route consistently yields Cefotiam Hexetil Hydrochloride with a purity exceeding 98.5 percent and an overall yield greater than 95 percent, surpassing standard pharmacopoeial requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Hexetil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in patent literature can be reliably replicated on an industrial scale. We understand that for antibiotics like Cefotiam Hexetil Hydrochloride, maintaining stringent purity specifications is non-negotiable. Our rigorous QC labs are equipped to monitor critical quality attributes, including isomer ratios and residual solvent levels, guaranteeing that every shipment meets the exacting standards required for global regulatory filings.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis routes, we can help you reduce your overall cost of acquisition while securing a stable supply of high-quality intermediates. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects. Let us demonstrate how our commitment to process excellence can become a strategic advantage for your pharmaceutical manufacturing operations.
