Advanced Synthesis of Carbapenem Antibiotic Intermediate 4-AA via Cost-Effective Chiral Resolution
Advanced Synthesis of Carbapenem Antibiotic Intermediate 4-AA via Cost-Effective Chiral Resolution
The pharmaceutical industry continuously seeks robust and economical pathways for synthesizing critical beta-lactam antibiotics, particularly carbapenems which serve as last-resort treatments for severe bacterial infections. A pivotal intermediate in this class is (3R,4R)-3-[(R)-1-tert-butyldimethylsilyloxyethyl]-4-acetoxy-2-azetidinone, commonly referred to as 4-AA, which possesses a complex structure featuring a strained four-membered beta-lactam ring and three distinct chiral centers. As illustrated in the chemical structure below, the precise stereochemical configuration of these centers is paramount for biological activity, making the synthesis of this molecule a significant challenge in process chemistry.
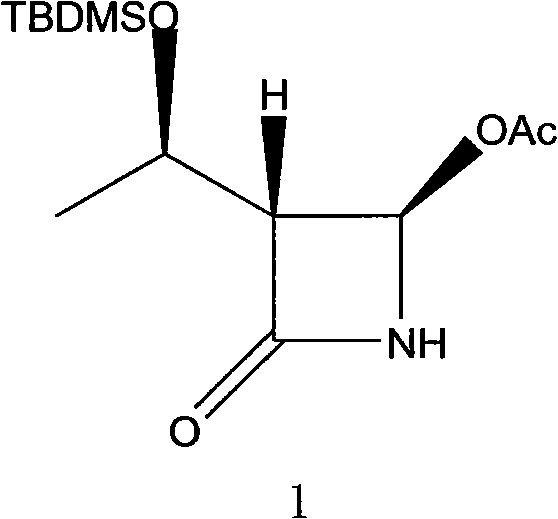
Patent CN101838282B, filed in 2012, introduces a groundbreaking preparation method that fundamentally shifts the paradigm from expensive catalytic asymmetric synthesis to a more accessible chemical resolution strategy. This innovation addresses the long-standing economic bottlenecks associated with traditional routes by leveraging classical resolution techniques using inexpensive chiral acids. For R&D directors and procurement managers alike, this patent represents a viable pathway to secure a reliable carbapenem intermediate supplier while drastically reducing the dependency on scarce noble metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the chiral centers in 4-AA has relied heavily on asymmetric hydrogenation using sophisticated transition metal complexes, specifically the BINAP-Ruthenium (Ru) catalyst system. As depicted in the reaction scheme below, this prior art method involves the reduction of a keto-ester precursor followed by cyclization to form the beta-lactam ring. While chemically elegant, this approach suffers from severe commercial drawbacks, primarily the exorbitant cost of the chiral ligand BINAP and the ruthenium metal itself, which renders the process economically unfeasible for large-scale manufacturing.
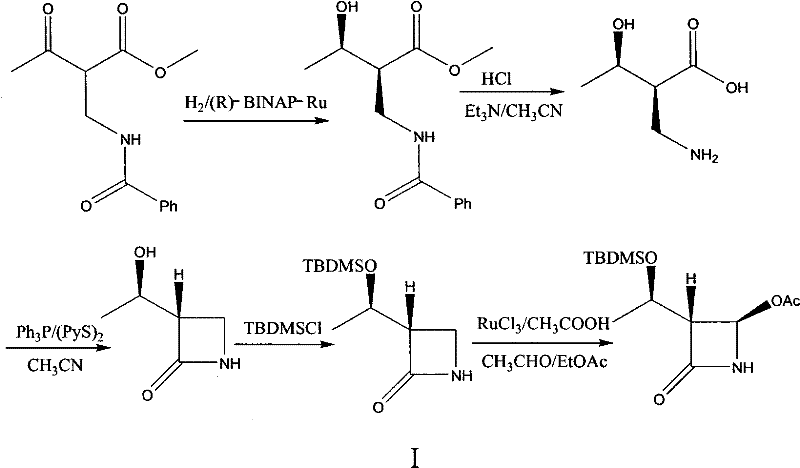
Furthermore, the use of heavy metal catalysts introduces stringent regulatory hurdles regarding residual metal limits in the final Active Pharmaceutical Ingredient (API). Removing trace amounts of ruthenium to meet pharmacopeial standards often requires additional purification steps, such as specialized scavenging resins or repeated crystallizations, which further erode yield and increase processing time. These factors combined create a fragile supply chain where cost volatility in precious metals can directly impact the availability and pricing of the final antibiotic, posing a significant risk for supply chain heads managing global inventory.
The Novel Approach
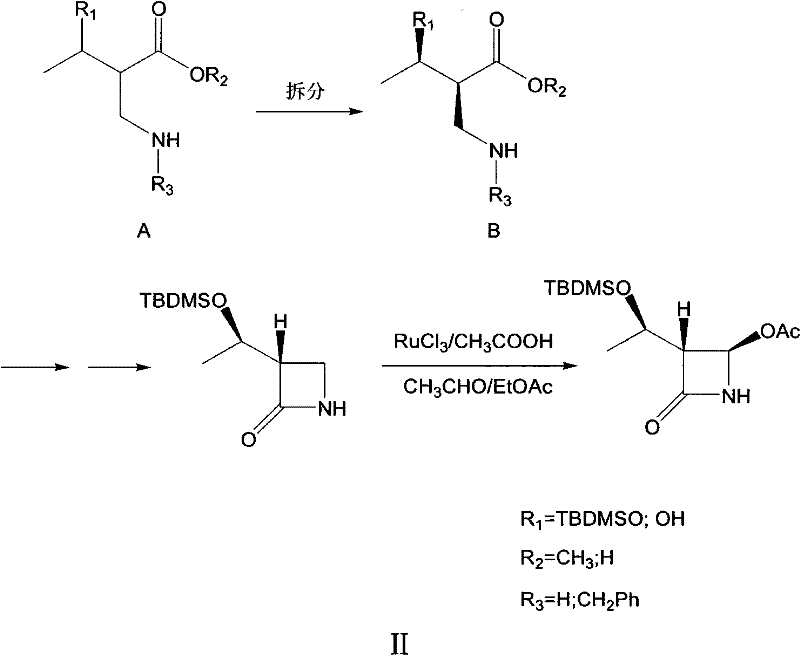
In stark contrast, the novel approach detailed in the patent circumvents these issues by employing a chemical resolution method that separates enantiomers through diastereomeric salt formation. Instead of creating chirality de novo via catalysis, this method starts with a racemic mixture and selectively crystallizes the desired enantiomer using cheap, naturally occurring chiral resolving agents like L-tartaric acid or D-dibenzoyltartaric acid. This shift eliminates the need for expensive transition metals entirely, replacing them with abundant organic acids that are easily sourced from the bulk chemical market.
The operational simplicity of this new route is another major advantage, as it relies on standard unit operations such as dissolution, heating, cooling crystallization, and filtration, which are well-understood and easily scalable in existing multipurpose reactors. By avoiding the sensitive handling requirements of air- and moisture-sensitive catalysts, the process becomes more robust and less prone to batch-to-batch variability. This reliability is crucial for ensuring the commercial scale-up of complex pharmaceutical intermediates without the technical risks associated with homogeneous catalysis.
Mechanistic Insights into Chiral Resolution and Cyclization
The core mechanism of this invention lies in the thermodynamic control of diastereomeric salt crystallization. When a racemic amine or ester substrate is mixed with a single enantiomer of a chiral acid (the resolving agent) in a suitable solvent system, two diastereomeric salts are formed. Due to differences in their crystal lattice energies and solubilities, one salt (containing the desired stereoisomer) precipitates out of the solution upon cooling, while the other remains dissolved. The patent specifies optimal solvent systems, such as mixtures of butanone and methanol, which fine-tune the solubility profile to maximize the recovery and optical purity of the target isomer.
Following the isolation of the chiral salt, the free base or acid is liberated through a basic workup, typically using carbonates or hydroxides, and extracted into an organic phase. This resolved chiral building block then serves as the precursor for the subsequent construction of the beta-lactam ring. The patent outlines that this resolved intermediate can be converted into the final 4-AA structure through established cyclization and acetylation protocols, ensuring that the stereochemical integrity established during the resolution step is preserved throughout the synthesis. This mechanistic clarity provides R&D teams with a predictable framework for optimizing impurity profiles.
Impurity control is inherently superior in this resolution-based route because the crystallization step acts as a powerful purification tool, rejecting not only the unwanted enantiomer but also many structurally related by-products that do not fit into the crystal lattice. This results in a starting material for the cyclization step that is of significantly higher purity compared to catalytic routes where side reactions like over-reduction or isomerization can occur. Consequently, the downstream processing burden is reduced, leading to a cleaner final product with a simpler impurity spectrum that is easier to characterize and control.
How to Synthesize 4-AA Efficiently
The synthesis protocol described in the patent offers a streamlined workflow that integrates resolution directly into the production line. The process begins with the careful selection of the resolving agent and solvent to match the specific substrate, whether it is a silylated ester or a hydroxy acid derivative. Operators must strictly adhere to the temperature gradients, dissolving the components at 40-80°C to ensure complete homogeneity before initiating the slow cooling phase which drives the selective crystallization. Detailed standardized synthetic steps see the guide below for precise execution parameters.
- Dissolve the resolving agent (e.g., L-tartaric acid) in a solvent mixture like butanone and methanol at 40-80°C.
- Add the racemic raw material equimolarly, stir until clear, and allow to stand at 5-20°C for 10-20 hours to crystallize the salt.
- Filter the solid, recrystallize for purity, then treat with aqueous base and extract with ether to isolate the resolved chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition from catalytic hydrogenation to chemical resolution offers profound strategic benefits that extend beyond simple unit cost savings. The primary advantage is the decoupling of production costs from the volatile precious metals market, as the process no longer depends on ruthenium or specialized phosphine ligands. This stability allows for more accurate long-term budgeting and protects the manufacturing margin from sudden spikes in raw material prices, ensuring a more predictable cost structure for the final API.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts results in substantial cost savings, as the resolving agents used, such as tartaric acid, are commodity chemicals produced on a massive scale. Additionally, the removal of heavy metal scavenging steps reduces the consumption of auxiliary materials and lowers waste disposal costs associated with hazardous metal residues. This leaner material profile translates directly into a lower cost of goods sold (COGS), enhancing the overall competitiveness of the manufactured intermediate in the global market.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral catalysts often involves long lead times and reliance on a limited number of specialized suppliers, creating a single point of failure in the supply chain. In contrast, the reagents required for this resolution method are widely available from multiple global vendors, significantly reducing the risk of supply disruption. This diversification of the supply base ensures continuous production capability even during periods of market instability or logistical constraints.
- Scalability and Environmental Compliance: The process relies on crystallization and filtration, which are inherently scalable operations that do not require the specialized high-pressure equipment often needed for hydrogenation reactions. This ease of scale-up facilitates rapid technology transfer from pilot plant to commercial production. Furthermore, the absence of toxic heavy metals simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent green chemistry regulations and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs.
Q: Why is the chemical resolution method preferred over BINAP-Ru catalysis for 4-AA synthesis?
A: The conventional BINAP-Ru catalytic hydrogenation route relies on extremely expensive noble metal catalysts, which drastically increases production costs and introduces heavy metal removal challenges. The chemical resolution method described in CN101838282B utilizes cheap, readily available organic acids like L-tartaric acid, significantly lowering raw material costs and simplifying the purification process for industrial scale-up.
Q: What are the key operational parameters for the resolution crystallization step?
A: The process requires dissolving the resolving agent at elevated temperatures between 40°C and 80°C, followed by a critical static crystallization period at low temperatures (5-20°C) for 10 to 20 hours. This controlled temperature gradient ensures high stereo-selectivity during salt formation, which is essential for achieving the necessary optical purity before the final extraction step.
Q: Can this resolution method be applied to various protected amino acid precursors?
A: Yes, the patent demonstrates versatility by successfully applying the resolution technique to multiple substrates, including 2-(benzylamino)methyl-3-tert-butyldimethylsilyloxybutyric acid methyl ester and its hydroxy analogues. This flexibility allows manufacturers to adapt the protocol based on their specific upstream synthetic routes while maintaining consistent chiral integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-AA Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against antibiotic-resistant bacteria. Our team of expert process chemists has extensively evaluated the resolution methodology described in CN101838282B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this vital carbapenem intermediate with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the exacting standards required for pharmaceutical synthesis.
We invite you to collaborate with us to optimize your supply chain for carbapenem antibiotics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term production goals efficiently and reliably.
