Scalable Copper-Catalyzed Synthesis of Lasmiditan for Commercial Pharmaceutical Manufacturing
Scalable Copper-Catalyzed Synthesis of Lasmiditan for Commercial Pharmaceutical Manufacturing
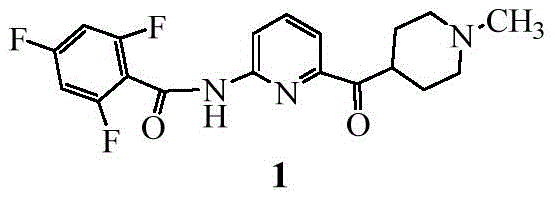
The pharmaceutical industry is constantly seeking robust and cost-effective synthetic routes for high-value active pharmaceutical ingredients (APIs), particularly for novel migraine treatments like Lasmiditan. Patent CN111943930B discloses a groundbreaking synthesis process for Lasmiditan, a selective 5-HT1F receptor agonist, which addresses critical limitations found in earlier manufacturing methods. This innovative approach utilizes a copper-catalyzed amidation reaction between 2,4,6-trifluorobenzamide and a specific bromopyridine derivative, operating under remarkably mild conditions. By shifting away from noble metal catalysts and harsh acylating agents, this technology offers a pathway to high-purity intermediates with significantly improved operational safety and environmental profiles. For global supply chain leaders, this represents a pivotal opportunity to optimize the production of complex pharmaceutical intermediates while ensuring consistent quality and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lasmiditan has relied on routes that present substantial challenges for industrial scale-up and cost efficiency. Early discovery routes, often referred to as Route A, utilized 2-chloropyridine derivatives coupled with benzophenone imine using expensive palladium catalysts like Pd2dba3 and phosphine ligands such as BINAP. This method suffered from extremely low yields, often around 10%, during the initial pyridine functionalization steps. Furthermore, subsequent steps involved the use of trifluorobenzoyl chloride, a highly corrosive and volatile reagent that requires rigorous distillation purification. The handling of such acyl chlorides generates significant amounts of acidic waste and poses severe corrosion risks to reactor equipment. Additionally, the reactivity of acyl chlorides often leads to over-acylation, forming di-acylated byproducts that require complex hydrolysis steps to revert to the desired product, thereby increasing raw material consumption and processing time.
The Novel Approach
In stark contrast, the novel process described in the patent employs a direct copper-catalyzed coupling between a stable amide and an aryl bromide, effectively bypassing the need for reactive acyl chlorides and noble metals. This method leverages a cuprous salt catalyst in conjunction with specific diamine ligands to facilitate the formation of the amide bond under mild thermal conditions, typically between 0°C and 60°C. By utilizing 2,4,6-trifluorobenzamide directly, the process eliminates the hazardous generation of hydrochloric acid and the associated corrosion issues inherent in acyl chloride chemistry. The reaction demonstrates excellent compatibility with a range of benign organic solvents, allowing for simpler work-up procedures and higher overall yields. This strategic shift not only simplifies the synthetic sequence but also drastically reduces the environmental footprint by minimizing toxic waste streams and avoiding the use of precious metal residues that require stringent removal protocols in final drug substances.
Mechanistic Insights into Copper-Catalyzed Amidation
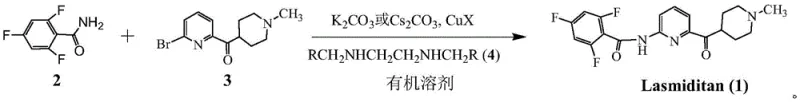
The core of this technological advancement lies in the optimization of the Goldberg-type amidation reaction, traditionally known for requiring harsh conditions. The patent reveals that the successful coupling at room temperature is heavily dependent on the precise selection of the ligand and solvent system. While literature often suggests polar aprotic solvents like DMF or DMSO for copper catalysis, this process surprisingly finds that low molecular weight esters, ethers, and ketones are superior. This is attributed to the unique solubility profile of the trifluorobenzamide substrate; in highly polar solvents, the reaction kinetics are sluggish at low temperatures, whereas in solvents like tetrahydrofuran or ethyl acetate, the catalyst system remains highly active. The mechanism likely involves the formation of a soluble copper-diamine complex that facilitates the oxidative addition of the aryl bromide and subsequent coordination with the amide nitrogen, enabling the C-N bond formation without the thermal energy typically required for unactivated aryl halides.
Furthermore, the choice of ligand plays a critical role in controlling both reactivity and impurity formation. The patent details extensive screening of N,N'-disubstituted ethylenediamines, identifying that ligands with specific steric and electronic properties, such as N,N'-dimethyl-1,2-ethylenediamine or N,N'-dibenzyl-1,2-ethylenediamine, provide optimal results. Ligands with excessive steric hindrance or strong electron-donating groups on aromatic rings were found to deactivate the catalyst or lead to side reactions. For instance, the use of ligands with bulky alkyl groups resulted in negligible conversion, while those with electron-withdrawing substituents maintained high catalytic activity. This fine-tuning ensures that the reaction proceeds selectively to the mono-acylated product, minimizing the formation of bis-acylated impurities that plagued earlier synthetic routes. The ability to run this reaction at ambient temperature also prevents the thermal degradation of the sensitive pyridine-piperidine scaffold, ensuring a cleaner impurity profile.
How to Synthesize Lasmiditan Efficiently
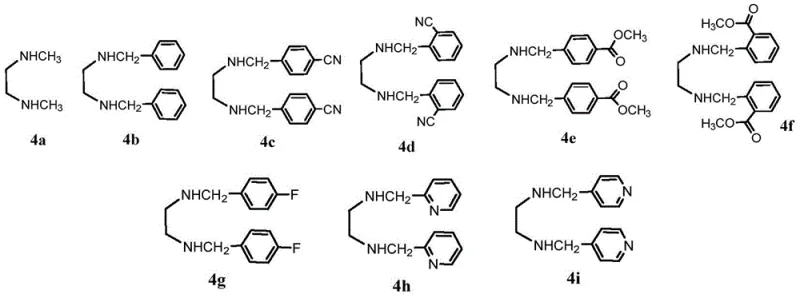
The synthesis of Lasmiditan via this copper-catalyzed route involves a straightforward one-pot coupling reaction that is amenable to large-scale manufacturing. The process begins with the preparation of the key starting materials, 2,4,6-trifluorobenzamide and (6-bromopyridin-2-yl)(1-methylpiperidin-4-yl)methanone, which are readily available or easily synthesized. These precursors are combined in a reactor with a catalytic amount of a cuprous salt, such as copper(I) iodide or copper(I) chloride, and a stoichiometric amount of a weak inorganic base like potassium carbonate. The selection of the solvent is paramount, with tetrahydrofuran, methyltetrahydrofuran, or ethyl acetate proving to be the most effective media for maintaining reaction homogeneity and catalyst stability. The reaction mixture is stirred under an inert atmosphere at controlled temperatures, typically ranging from 20°C to 30°C, until the starting bromide is fully consumed. Detailed standardized synthesis steps for this specific protocol are provided in the guide below.
- Prepare the reaction mixture by combining 2,4,6-trifluorobenzamide, (6-bromopyridin-2-yl)(1-methylpiperidin-4-yl)methanone, a cuprous salt catalyst, and a diamine ligand in an organic solvent.
- Add a weak inorganic solid base such as potassium carbonate and stir the mixture at a controlled temperature between 0°C and 60°C, optimally at room temperature.
- Upon completion, filter the reaction mass, wash with appropriate solvents and aqueous solutions to remove copper residues and ligands, and isolate the pure Lasmiditan product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed synthesis offers transformative benefits in terms of cost structure and operational reliability. The most immediate impact is the elimination of palladium catalysts and expensive phosphine ligands, which are subject to significant price volatility and supply constraints. By substituting these with abundant and inexpensive copper salts and simple diamine ligands, the raw material cost for the key coupling step is drastically reduced. Moreover, the avoidance of acyl chloride chemistry removes the need for specialized corrosion-resistant equipment and complex scrubbing systems for acidic off-gases, leading to substantial capital expenditure savings and lower maintenance costs. The simplified work-up procedure, which avoids difficult distillations and extensive chromatographic purifications, further enhances the economic viability of the process by reducing solvent consumption and processing time.
- Cost Reduction in Manufacturing: The transition from noble metal catalysis to a copper-based system fundamentally alters the cost equation for Lasmiditan production. Palladium catalysts and ligands like BINAP are not only expensive to purchase but also require costly recovery or scavenging processes to meet strict residual metal limits in pharmaceutical products. By utilizing copper, which is orders of magnitude cheaper and easier to remove via simple aqueous washes, manufacturers can achieve significant cost savings per kilogram of API produced. Additionally, the higher yields and reduced formation of byproducts mean that less starting material is wasted, improving the overall atom economy and reducing the cost of goods sold. The milder reaction conditions also translate to lower energy consumption, as there is no need for prolonged heating or cryogenic cooling, contributing to a more sustainable and cost-efficient manufacturing footprint.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals rather than specialized reagents significantly de-risks the supply chain. Copper salts and diamine ligands are widely available from multiple global suppliers, ensuring continuity of supply even during market disruptions. In contrast, the specialized phosphine ligands and palladium complexes used in older routes often have longer lead times and are sourced from a limited number of vendors. Furthermore, the stability of the amide coupling partners allows for longer shelf-life and easier storage compared to moisture-sensitive acyl chlorides, reducing the risk of raw material spoilage. This robustness enables manufacturers to maintain leaner inventory levels while still guaranteeing production schedules, providing a competitive advantage in meeting market demand for migraine therapies.
- Scalability and Environmental Compliance: The simplicity and safety of this process make it ideally suited for commercial scale-up. The absence of highly corrosive reagents and the use of standard organic solvents simplify the engineering requirements for large-scale reactors, facilitating a smoother technology transfer from pilot plant to full production. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The process generates fewer toxic byproducts and avoids the release of volatile acidic gases, simplifying waste treatment and disposal. This environmental stewardship not only reduces compliance costs but also enhances the corporate sustainability profile, which is becoming a critical factor in supplier selection for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this technology for their own manufacturing pipelines. The answers highlight the specific advantages in terms of yield, purity, and operational simplicity that distinguish this method from prior art.
Q: Why is the copper-catalyzed route preferred over palladium methods for Lasmiditan?
A: The copper-catalyzed route eliminates the need for expensive noble metal catalysts like Pd2dba3 and costly ligands such as BINAP. It operates under milder conditions (room temperature) compared to traditional Goldberg reactions which often require high temperatures, significantly reducing raw material costs and energy consumption.
Q: What are the critical solvent considerations for this synthesis?
A: Unlike traditional couplings that use polar aprotic solvents like DMF or DMSO, this process utilizes low molecular weight esters, ethers, or ketones (e.g., THF, ethyl acetate). These solvents are chosen because they maintain catalyst activity at lower temperatures while avoiding the solubility issues and purification difficulties associated with high-boiling polar solvents.
Q: How does this process improve impurity control?
A: By using a stable amide coupling partner instead of reactive acyl chlorides, the process avoids the formation of di-acylated byproducts common in previous routes. Additionally, the mild reaction conditions prevent the thermal decomposition of sensitive intermediates, leading to a cleaner crude profile and higher final purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lasmiditan Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthetic routes for high-value pharmaceutical intermediates like Lasmiditan. Our team of expert chemists has extensively evaluated the copper-catalyzed amidation technology described in patent CN111943930B and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Lasmiditan intermediate we produce adheres to the highest quality standards required by global regulatory bodies.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for migraine therapeutics. By leveraging our expertise in process chemistry and scale-up engineering, we can help you realize the full cost-saving potential of this novel synthesis route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term supply goals and enhance your product competitiveness in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
