Advanced Green Synthesis of Aztreonam: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
Advanced Green Synthesis of Aztreonam: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
The global demand for effective monobactam antibiotics continues to rise, driven by the critical need for treatments against Gram-negative bacterial infections that are resistant to other classes of antibiotics. Aztreonam, a synthetic monocyclic beta-lactam, stands out due to its unique stability against many beta-lactamases and its favorable safety profile. However, the manufacturing of this vital active pharmaceutical ingredient (API) has historically been plagued by complex synthetic routes involving hazardous reagents and expensive solvents. A significant technological breakthrough is detailed in patent CN102127068B, which introduces a novel, environmentally benign method for synthesizing Aztreonam. This innovation replaces traditional toxic organic solvents with a sustainable aqueous-alcoholic system, fundamentally altering the economic and ecological landscape of production. By optimizing reaction conditions and solvent selection, this process not only enhances the final product's purity but also streamlines the workflow for industrial scale-up.
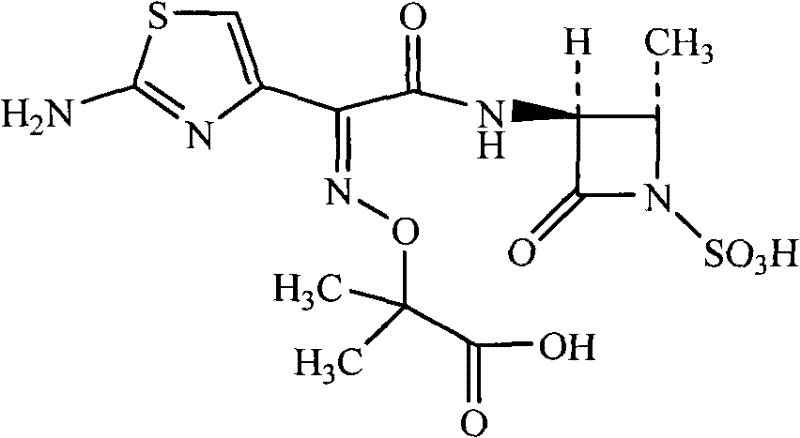
For R&D directors and process chemists, the structural integrity of the beta-lactam ring is paramount. The provided chemical structure illustrates the delicate balance of functional groups required for biological activity. The patent addresses the challenge of constructing this molecule without compromising its stability. The method described utilizes a condensation reaction between trans-3(S)-amino-4-methyl-2-oxo-1-azetidinyl sulfonic acid and a specifically activated thiazole ester. This approach minimizes side reactions that typically lead to difficult-to-remove impurities. Furthermore, the integration of a controlled crystallization process ensures that the final API meets the stringent specifications required by regulatory bodies worldwide. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is crucial for delivering consistent quality to downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for Aztreonam synthesis, such as those described in earlier patents like US4775670A, have relied heavily on harsh chemical environments that pose significant operational and safety challenges. These traditional routes often employ solvents like tetrahydrofuran (THF), acetonitrile, and dichloromethane, which are not only costly but also present substantial environmental hazards due to their volatility and toxicity. Moreover, the use of strong acids like trifluoroacetic acid for deprotection steps necessitates specialized corrosion-resistant equipment and rigorous waste treatment protocols, driving up capital expenditure. Another critical drawback in prior art, such as the method in CN101514200A, involves the use of 1,4-dioxane, a solvent with known carcinogenic potential, making it increasingly undesirable for modern GMP manufacturing. These legacy processes often suffer from inconsistent product quality, requiring extensive downstream purification that erodes overall yield and extends production lead times.
The Novel Approach
The methodology outlined in CN102127068B represents a paradigm shift towards green chemistry principles in antibiotic manufacturing. By substituting hazardous organic solvents with a mixture of purified water and water-miscible alcohols or ketones (specifically methanol, ethanol, or acetone), the process drastically reduces the environmental footprint. This solvent system is not merely a substitution; it is engineered to optimize the solubility of both the polar sulfonic acid substrate and the organic ester reactant. The reaction proceeds under mild conditions, typically between -40°C and 5°C, which preserves the stereochemical integrity of the chiral centers. Additionally, the deprotection step utilizes a simple acetic acid-water mixture, eliminating the need for aggressive mineral acids. This simplification of the chemical environment facilitates easier solvent recovery and recycling, directly contributing to cost reduction in pharmaceutical intermediates manufacturing while enhancing operator safety.
Mechanistic Insights into pH-Controlled Condensation and Crystallization
The core of this synthetic innovation lies in the precise control of the condensation reaction between the azetidine sulfonic acid derivative and the activated ester. The mechanism involves the nucleophilic attack of the primary amine on the carbonyl carbon of the ester, facilitated by the presence of triethylamine as a base. However, the sensitivity of the beta-lactam ring to hydrolysis requires a delicate balance. The patent specifies maintaining the reaction pH between 5.0 and 9.0 during the addition of reagents. This narrow window ensures that the amine remains sufficiently unprotonated to act as a nucleophile while preventing the base-catalyzed opening of the four-membered lactam ring. The use of a benzothiazole-2-sulfenyl carbonyl leaving group further enhances the electrophilicity of the ester, allowing the reaction to proceed efficiently at lower temperatures, thereby suppressing thermal degradation pathways.
Impurity control is achieved through a sophisticated crystallization strategy rather than relying solely on chromatography. After the initial condensation, the intermediate t-butyl aztreonam is isolated by adjusting the pH to an acidic range of 1.0 to 1.5. At this pH, the zwitterionic nature of the molecule shifts, reducing its solubility in the aqueous-alcoholic medium and promoting the formation of high-quality crystals. This crystallization step acts as a powerful purification barrier, excluding unreacted starting materials and soluble by-products. Subsequent washing with freezing non-aqueous solvents (at temperatures between -40°C and 0°C) removes surface impurities without dissolving the product. This rigorous solid-state processing ensures that the final high-purity Aztreonam exhibits an HPLC purity of over 99.0%, meeting the strict requirements for parenteral administration.
How to Synthesize Aztreonam Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and addition rates to maximize yield and safety. The process is designed to be robust, accommodating slight variations in raw material quality while maintaining consistent output. The following guide outlines the critical operational parameters derived from the patent examples, serving as a foundational protocol for process engineers looking to adopt this technology. For a comprehensive, step-by-step Standard Operating Procedure (SOP) tailored to your specific reactor configuration, please refer to the detailed technical documentation below.
- Condense trans-3(S)-amino-4-methyl-2-oxo-1-azetidinyl sulfonic acid with the activated thiazole ester in a water-methanol/ethanol/acetone mixture at -40 to 5°C, maintaining pH 5.0-9.0 with triethylamine.
- Isolate the t-butyl aztreonam intermediate by adjusting pH to 1.0-1.5 and crystallizing, then wash with freezing solvent.
- Deprotect the t-butyl group using an acetic acid-water mixture at 50-55°C, followed by cooling and crystallization to obtain pure Aztreonam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this green synthesis route offers tangible strategic benefits beyond mere regulatory compliance. The elimination of expensive and tightly regulated solvents like THF and dioxane simplifies the sourcing of raw materials, reducing the risk of supply disruptions caused by environmental regulations or vendor shortages. The use of commodity chemicals such as ethanol, acetone, and acetic acid ensures a stable and cost-effective supply base. Furthermore, the simplified workup procedure, which avoids complex extraction steps and relies on direct crystallization, significantly reduces the cycle time per batch. This efficiency gain allows for higher throughput in existing facilities, effectively increasing capacity without the need for major capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The substitution of premium-priced specialty solvents with bulk commodity solvents results in a direct decrease in variable production costs. Additionally, the aqueous-based system simplifies wastewater treatment, as the solvents are biodegradable and easier to separate compared to halogenated organics. The removal of toxic reagents also lowers the costs associated with hazardous waste disposal and worker safety monitoring. By streamlining the synthesis into fewer operational steps with higher inherent yields, the overall cost of goods sold (COGS) is substantially optimized, providing a competitive pricing advantage in the global market.
- Enhanced Supply Chain Reliability: Relying on widely available solvents like methanol and ethanol mitigates the risk of supply chain bottlenecks that often plague specialty chemical markets. The robustness of the reaction conditions, particularly the tolerance for a broad pH range (5.0-9.0) during addition, reduces the likelihood of batch failures due to minor process deviations. This reliability translates to more predictable delivery schedules for downstream API manufacturers. The ability to produce high-quality intermediates consistently ensures that the supply chain remains resilient against market fluctuations and regulatory audits.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples ranging from gram-scale laboratory experiments to multi-kilogram pilot runs. The exothermic nature of the reaction is easily managed in large reactors due to the high heat capacity of the water-containing solvent system. From an environmental perspective, the reduction in volatile organic compound (VOC) emissions aligns with increasingly stringent global environmental standards. This compliance future-proofs the manufacturing asset, ensuring long-term operational viability without the need for costly retrofits to meet evolving emission limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Aztreonam synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios.
Q: How does the new solvent system improve upon traditional Aztreonam synthesis methods?
A: Traditional methods often rely on toxic and expensive solvents like THF, acetonitrile, or trifluoroacetic acid. The patented method replaces these with a greener, cost-effective mixture of water and water-miscible solvents such as methanol, ethanol, or acetone, significantly reducing environmental impact and raw material costs.
Q: What purity levels can be achieved with this synthetic route?
A: Experimental data from the patent indicates that this method consistently yields Aztreonam with HPLC purity exceeding 99.0%, with specific embodiments demonstrating purity levels of 99.1% to 99.2%.
Q: Why is pH control critical during the condensation step?
A: Maintaining the pH between 5.0 and 9.0 during the addition of triethylamine is essential to balance the nucleophilicity of the amine substrate while preventing the hydrolysis of the sensitive beta-lactam ring, ensuring high yield and minimal impurity formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Supplier
The synthesis route described in CN102127068B exemplifies the kind of process innovation that drives value in the modern pharmaceutical industry. At NINGBO INNO PHARMCHEM, we specialize in translating such complex laboratory methodologies into robust, commercial-scale manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of green chemistry are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Aztreonam meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to collaborate with us on optimizing their supply chains for this critical antibiotic. Whether you require custom synthesis services or bulk supply of intermediates, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis for your specific project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
