Advanced One-Pot Synthesis of Aztreonam: Enhancing Purity and Scalability for Global API Markets
Advanced One-Pot Synthesis of Aztreonam: Enhancing Purity and Scalability for Global API Markets
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics like aztreonam, a monobactam essential for treating Gram-negative bacterial infections. Patent CN101171251A introduces a transformative one-pot preparation method that fundamentally alters the production landscape by integrating condensation and hydrolysis steps without the intermediary isolation of tert-butylaztreonam. This technical breakthrough addresses long-standing inefficiencies in legacy synthetic routes, offering a streamlined approach that enhances overall process throughput while maintaining stringent purity standards required for active pharmaceutical ingredients. By leveraging a specific solvent system comprising C1-C3 tertiary amines and ketones, this methodology facilitates the direct conversion of azetidine and TAEM into the final active compound, thereby minimizing unit operations and reducing the environmental footprint associated with solvent exchange and waste generation. For R&D directors and process chemists, this represents a significant opportunity to optimize existing manufacturing lines for high-purity aztreonam production.
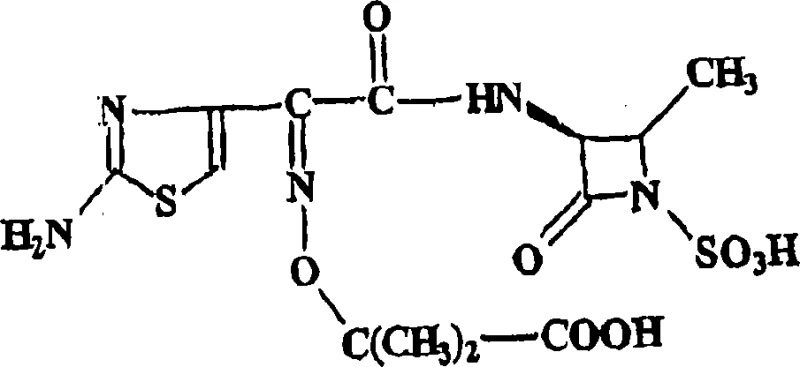
The structural complexity of aztreonam necessitates precise control over stereochemistry and functional group integrity throughout the synthesis. As illustrated in the molecular architecture, the presence of the beta-lactam ring and the sulfonic acid moiety requires mild yet effective reaction conditions to prevent degradation. The patented process achieves this by carefully managing pH levels and temperature profiles during the transition from the protected intermediate to the final acid form. This level of control is paramount for ensuring that the resulting API meets the rigorous specifications demanded by global regulatory bodies, making this synthesis route particularly attractive for established reliable pharmaceutical intermediate supplier networks aiming to secure long-term supply contracts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aztreonam has been plagued by cumbersome multi-step procedures that involve the isolation of unstable intermediates and the use of hazardous reagents. Prior art, such as that described in U.S. Patent No. 4,775,670, relies heavily on acylation reactions utilizing carbodiimides like dicyclohexylcarbodiimide (DCC) and additives like N-hydroxybenzotriazole, which are not only toxic but also expensive and difficult to handle on a large industrial scale. Furthermore, the deprotection steps in these conventional methods often require anhydrous conditions and strong acids like trifluoroacetic acid in the presence of anisole, creating significant safety hazards and waste disposal challenges for manufacturing facilities. The necessity to isolate the tert-butylaztreonam intermediate in these traditional routes introduces additional unit operations, including extraction and drying, which inevitably lead to product loss and increased cycle times. These inefficiencies accumulate to drive up the cost of goods sold and complicate the supply chain logistics for high-purity antibiotic intermediates.
The Novel Approach
In stark contrast, the novel one-pot approach detailed in the patent data circumvents these bottlenecks by combining the condensation of azetidine and TAEM directly with the subsequent hydrolysis step in a unified reactor system. This methodology eliminates the isolation of the tert-butylaztreonam intermediate, thereby saving considerable time and reducing the potential for yield loss associated with physical separation processes. By replacing the traditional acetonitrile solvent system with a mixture of acetone, 2-butanone, or methyl isobutyl ketone (MIBK) alongside a C1-C3 tertiary amine, the process achieves superior solubility for both reactants and products, significantly reducing the requirement for excess TAEM. This solvent innovation not only lowers raw material costs but also simplifies the workup procedure, as the removal of the solvent can be achieved through straightforward distillation with water. The result is a drastically simplified workflow that enhances plant capacity and operational efficiency without compromising the chemical integrity of the final product.
Mechanistic Insights into One-Pot Condensation and Hydrolysis
The core of this technological advancement lies in the precise orchestration of the condensation reaction between the azetidine nucleus and the activated side chain precursor, TAEM. The reaction initiates with the formation of an amide bond under basic conditions provided by the tertiary amine, which acts as both a solvent component and a proton scavenger. 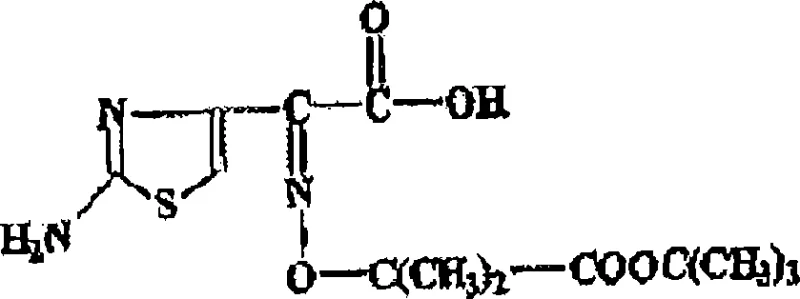 The use of ketone solvents is critical here, as they provide a homogeneous medium that supports the reaction kinetics while remaining compatible with the subsequent aqueous hydrolysis step. Unlike previous methods that required a complete solvent swap, this system allows for the seamless transition from the organic phase to the hydrolysis phase by simply adding water and adjusting the acidity. This continuity prevents the exposure of the sensitive beta-lactam ring to harsh conditions prematurely, ensuring high stereochemical fidelity throughout the transformation.
The use of ketone solvents is critical here, as they provide a homogeneous medium that supports the reaction kinetics while remaining compatible with the subsequent aqueous hydrolysis step. Unlike previous methods that required a complete solvent swap, this system allows for the seamless transition from the organic phase to the hydrolysis phase by simply adding water and adjusting the acidity. This continuity prevents the exposure of the sensitive beta-lactam ring to harsh conditions prematurely, ensuring high stereochemical fidelity throughout the transformation.
A crucial aspect of this mechanism is the management of impurities, specifically the removal of the mercaptobenzothiazole (MBT) by-product generated during the activation of the side chain. 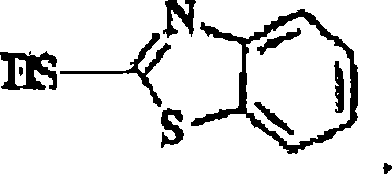 In the patented process, the pH of the reaction mixture is adjusted to a range of approximately 4.5 to 7.5 prior to hydrolysis, which induces the precipitation of MBT. This solid by-product is then physically separated via filtration, effectively purifying the reaction mixture before the critical deprotection step occurs. This strategic removal prevents MBT from interfering with the hydrolysis or contaminating the final crystal lattice of the aztreonam. Following filtration, the pH is lowered to less than 2 using mineral acids like HCl, and the mixture is heated to facilitate the cleavage of the tert-butyl ester group. This controlled acidic hydrolysis converts the intermediate directly into the zwitterionic form of aztreonam, which is then recovered through cooling and crystallization.
In the patented process, the pH of the reaction mixture is adjusted to a range of approximately 4.5 to 7.5 prior to hydrolysis, which induces the precipitation of MBT. This solid by-product is then physically separated via filtration, effectively purifying the reaction mixture before the critical deprotection step occurs. This strategic removal prevents MBT from interfering with the hydrolysis or contaminating the final crystal lattice of the aztreonam. Following filtration, the pH is lowered to less than 2 using mineral acids like HCl, and the mixture is heated to facilitate the cleavage of the tert-butyl ester group. This controlled acidic hydrolysis converts the intermediate directly into the zwitterionic form of aztreonam, which is then recovered through cooling and crystallization.
How to Synthesize Aztreonam Efficiently
Implementing this synthesis route requires strict adherence to temperature and pH controls to maximize yield and purity. The process begins with the preparation of the solvent system, followed by the sequential addition of reactants under inert conditions to prevent oxidation. Detailed operational parameters regarding stirring rates, addition speeds, and crystallization times are critical for reproducibility on a commercial scale. For a comprehensive guide on the specific operational steps and safety protocols required to execute this synthesis, please refer to the standardized procedure outlined below.
- Condense azetidine and TAEM in a C1-C3 tertiary amine and ketone solvent mixture at controlled low temperatures.
- Adjust pH to precipitate and filter out the MBT by-product before hydrolysis.
- Acidify the mixture, heat for hydrolysis, and cool to crystallize the final aztreonam product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this one-pot synthesis method offers profound advantages in terms of cost structure and logistical reliability. The elimination of intermediate isolation steps translates directly into reduced labor costs and lower energy consumption, as fewer reactors and separation units are required to produce the same volume of final API. Furthermore, the substitution of expensive and hazardous reagents with more common industrial solvents like acetone and MIBK significantly lowers the raw material expenditure and simplifies the sourcing strategy for purchasing managers. This reduction in chemical complexity also mitigates the risks associated with the storage and handling of dangerous substances, thereby lowering insurance premiums and compliance overheads for manufacturing sites. Consequently, this process enables a more competitive pricing model for high-purity aztreonam without sacrificing quality margins.
- Cost Reduction in Manufacturing: The streamlined nature of the one-pot process inherently drives down manufacturing costs by minimizing the number of unit operations and reducing solvent usage. By avoiding the isolation of the tert-butyl intermediate, the process eliminates the need for extensive washing, drying, and re-dissolution steps, which are typically resource-intensive. Additionally, the ability to recover and recycle the ketone-based solvent system further enhances the economic viability of the production line. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final antibiotic product more accessible in cost-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as acetone, triethylamine, and hydrochloric acid ensures a robust and resilient supply chain that is less susceptible to disruptions caused by specialty chemical shortages. Unlike processes dependent on exotic catalysts or hard-to-source reagents, this method utilizes standard industrial inputs that can be procured from multiple global vendors. This diversification of the supply base reduces lead times for raw material acquisition and provides greater flexibility in production scheduling. For supply chain heads, this means a more predictable delivery schedule for finished aztreonam, ensuring continuity of supply for downstream formulation partners.
- Scalability and Environmental Compliance: The simplicity of the reaction design facilitates easy scale-up from pilot batches to multi-ton commercial production without the need for specialized equipment modifications. The reduction in solvent volume and the elimination of toxic coupling agents like DCC significantly decrease the generation of hazardous waste, aligning the process with increasingly stringent environmental regulations. The efficient removal of the MBT by-product prior to hydrolysis also simplifies wastewater treatment requirements, as the load of organic contaminants is reduced before the final acidic workup. These factors collectively support sustainable manufacturing practices and ensure long-term regulatory compliance for pharmaceutical producers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel aztreonam synthesis pathway. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of adopting this technology within existing production frameworks.
Q: How does the one-pot method improve upon conventional aztreonam synthesis?
A: The one-pot method eliminates the need to isolate the tert-butylaztreonam intermediate, significantly reducing solvent consumption and processing time compared to traditional multi-step acylation and deprotection routes.
Q: What solvents are utilized in this novel aztreonam preparation process?
A: The process utilizes a mixture of C1-C3 tertiary amines and ketone solvents such as acetone, 2-butanone, or methyl isobutyl ketone (MIBK), replacing more hazardous or expensive solvent systems like acetonitrile.
Q: How is the MBT by-product managed in this synthesis route?
A: The MBT by-product is precipitated by adjusting the pH to approximately 5.5 prior to the hydrolysis step, allowing for its physical removal via filtration, which simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antibiotics like aztreonam. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot method are fully realized in practical application. We are committed to delivering high-purity aztreonam that meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By integrating this optimized synthesis technology into our manufacturing portfolio, we can offer our global partners a reliable source of premium quality intermediates that support their own drug development and production goals.
We invite pharmaceutical companies and procurement specialists to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your supply chain efficiency. Contact us today to discuss how we can collaborate to bring cost-effective and high-quality aztreonam solutions to the global market.
