Revolutionizing Carbapenem Intermediate Production: Safe and Scalable Synthetic Routes
Introduction to Advanced Carbapenem Intermediate Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic classes, particularly carbapenems, which remain vital for treating resistant bacterial infections. Patent CN101255171A introduces a significant technological advancement in the production of 2-substituted carbapenem derivative intermediates, specifically targeting the compound of formula (1) and its subsequent conversion to the active formula (A). This innovation addresses long-standing challenges in the field by providing a process that is not only efficient but also markedly safer and more cost-effective than previous methodologies. The core breakthrough lies in the strategic redesign of the coupling and cyclization steps, moving away from hazardous reagents while maintaining high stereochemical integrity. For stakeholders in the global supply chain, this represents a pivotal shift towards more sustainable and reliable manufacturing practices for high-purity pharmaceutical intermediates.
Historically, the synthesis of complex beta-lactam structures involved intricate multi-step sequences that often relied on toxic organometallic reagents. The present invention, as detailed in the patent documentation, offers a refined approach that streamlines these operations. By focusing on the preparation of the imidazothiazole side chain and its conjugation with the azetidinone core through novel phosphorus-mediated reactions, the process mitigates risks associated with heavy metal contamination. This is particularly crucial for a reliable carbapenem intermediate supplier aiming to meet stringent regulatory standards. The technical depth of this patent provides a foundation for scaling production from laboratory quantities to commercial tons, ensuring that the supply of these critical medical ingredients remains uninterrupted and compliant with evolving environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
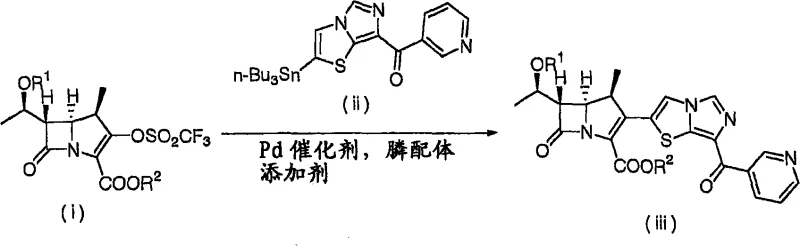
Prior art methods, such as those disclosed in WO 02/42312, typically rely on the use of organotin compounds, specifically trialkyltin chlorides, for the introduction of key side chains onto the carbapenem nucleus. While chemically effective, these reagents are notoriously toxic and pose significant health and environmental hazards during manufacturing. The handling of organotin species requires specialized equipment and rigorous waste treatment protocols to prevent contamination of the final product and the surrounding ecosystem. Furthermore, the conventional routes often necessitate the use of expensive palladium catalysts and phosphine ligands for coupling reactions. These precious metals not only drive up the raw material costs substantially but also introduce complex purification challenges. Removing trace amounts of palladium from the final active pharmaceutical ingredient (API) is a resource-intensive process that can bottleneck production timelines and increase the overall cost reduction in pharmaceutical intermediates manufacturing efforts.
In addition to safety and cost concerns, the older synthetic pathways often suffer from issues related to stereochemical control. The harsh conditions sometimes required for organotin-mediated reactions can lead to epimerization at sensitive chiral centers, resulting in lower yields of the desired biologically active isomer. This impurity profile complicates downstream processing and requires additional chromatographic steps to achieve the necessary purity levels for clinical use. The reliance on these legacy methods creates a fragile supply chain where any disruption in the availability of specialized catalysts or strict enforcement of environmental laws regarding tin waste can halt production. Consequently, there is a pressing industrial need for alternative routes that maintain high efficiency while eliminating these critical vulnerabilities associated with toxic reagents and expensive catalytic systems.
The Novel Approach
The methodology presented in CN101255171A fundamentally reengineers the synthetic strategy by eliminating the need for organotin compounds and substantially reducing the dependence on palladium catalysts. Instead, the invention utilizes a sequence involving Vilsmeier formylation, Grignard reactions, and phosphorus ylide chemistry to construct the critical carbon-carbon bonds. This shift allows for the preparation of the compound of formula (1) under milder conditions that are inherently safer for operators and the environment. By replacing toxic tin reagents with more benign alternatives, the process simplifies the workup procedures and reduces the burden on waste management systems. This transition is not merely a substitution of chemicals but a holistic optimization of the reaction pathway that enhances the overall robustness of the manufacturing process for complex pharmaceutical intermediates.
Moreover, the new approach offers superior control over the reaction parameters, leading to improved yields and purity profiles. The use of phosphorus-based reagents for the cyclization step to form the carbapenem ring is particularly advantageous as it avoids the generation of heavy metal byproducts. This results in a cleaner reaction mixture that requires less intensive purification, thereby accelerating the production cycle. The ability to produce high-purity carbapenem derivatives without the baggage of toxic contaminants makes this method highly attractive for commercial scale-up of complex pharmaceutical intermediates. It aligns perfectly with modern green chemistry principles, offering a sustainable solution that meets the dual demands of economic efficiency and environmental responsibility, ensuring a stable supply of essential antibacterial agents for the global market.
Mechanistic Insights into Phosphorus-Mediated Cyclization
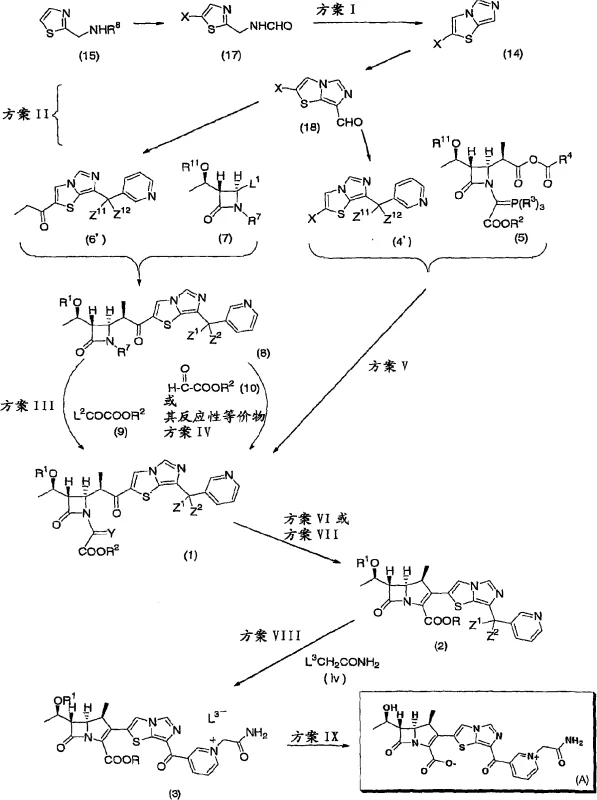
The core of this technological advancement lies in the intricate mechanistic steps that facilitate the formation of the carbapenem ring without traditional toxic aids. The process begins with the preparation of the imidazothiazole side chain, where a Vilsmeier complex is employed to introduce a formyl group onto the thiazole ring. This electrophilic aromatic substitution is carefully controlled to ensure regioselectivity, setting the stage for subsequent nucleophilic attacks. The resulting aldehyde is then reacted with a 3-metal pyridine, typically a Grignard reagent, to establish the carbon framework connecting the thiazole and pyridine moieties. This step is critical as it defines the spatial arrangement of the side chain, which is essential for the antibacterial activity of the final molecule. The precision required in these early stages underscores the importance of rigorous process control to prevent the formation of structural isomers that could compromise efficacy.
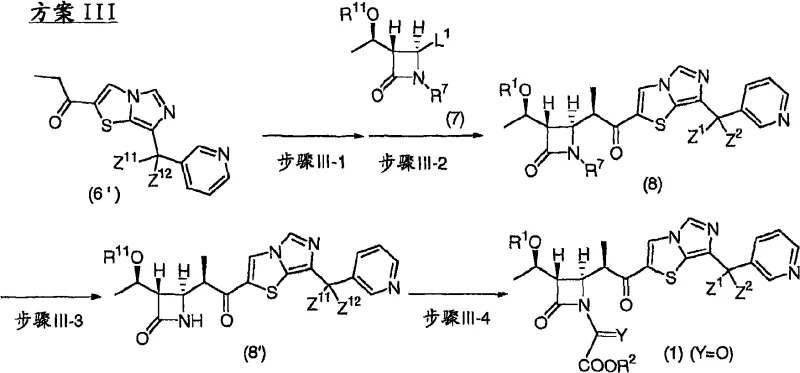
Following the assembly of the side chain, the coupling with the azetidinone core is achieved through metal enolate chemistry. The patent describes treating a propionic acid derivative with a base to generate an enolate, which then attacks the activated azetidinone. This reaction is performed at low temperatures to maintain stereochemical integrity, preventing racemization at the chiral centers. The subsequent cyclization to close the carbapenem ring utilizes phosphorus reagents, such as triethyl phosphite or diethyl methylphosphonite. This intramolecular reaction proceeds through a mechanism that expels the phosphorus oxide byproduct, driving the equilibrium towards the formation of the double bond in the carbapenem system. This elegant mechanistic pathway avoids the need for external oxidants or metal catalysts that could introduce impurities, ensuring that the final product meets the stringent specifications required for a reliable carbapenem intermediate supplier.
How to Synthesize Carbapenem Intermediates Efficiently
The practical implementation of this synthesis requires a deep understanding of the reaction conditions and reagent stoichiometry outlined in the patent. The process is designed to be modular, allowing for the optimization of individual steps such as formylation, coupling, and cyclization independently before integration into a continuous flow or batch process. Operators must pay close attention to temperature controls, particularly during the Grignard addition and enolate formation steps, where exothermic reactions can occur. The use of anhydrous solvents and inert atmospheres is paramount to prevent the degradation of sensitive intermediates. By adhering to the standardized protocols derived from the patent examples, manufacturers can achieve consistent quality and yield. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different production scales.
- Prepare the imidazothiazole core via Vilsmeier formylation and Grignard addition to avoid toxic organotin reagents.
- Couple the side chain using metal enolate chemistry with azetidinone precursors under controlled low-temperature conditions.
- Execute ring closure using phosphorus reagents to form the carbapenem skeleton, followed by deprotection to yield the final active intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond simple chemical efficiency. The elimination of organotin compounds removes a major regulatory hurdle, as tin residues are heavily scrutinized by health authorities. This simplification in compliance requirements translates directly into reduced administrative overhead and faster time-to-market for new drug filings. Furthermore, the avoidance of expensive palladium catalysts significantly lowers the raw material cost base. Precious metals are subject to volatile market pricing, and their removal from the bill of materials stabilizes production costs, allowing for more accurate long-term budgeting and pricing strategies. This stability is crucial for maintaining competitive margins in the highly price-sensitive generic pharmaceutical market.
Supply chain reliability is another critical advantage conferred by this technology. Organotin reagents and specialized palladium catalysts often have limited suppliers and long lead times, creating potential bottlenecks. By shifting to more common and readily available phosphorus reagents and Grignard components, the manufacturing process becomes more resilient to supply disruptions. This diversification of the supply base ensures continuity of supply even during global shortages of specific specialty chemicals. Additionally, the simplified purification process reduces the consumption of solvents and chromatography media, lowering logistical costs associated with waste disposal and material handling. These factors collectively contribute to a more agile and responsive supply chain capable of meeting fluctuating demand for high-purity carbapenem derivatives without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The removal of costly palladium catalysts and toxic organotin reagents drastically simplifies the bill of materials. This change eliminates the need for expensive heavy metal scavenging resins and complex filtration steps, leading to substantial cost savings in both raw materials and processing time. The overall reduction in chemical complexity allows for more efficient resource allocation and lower operational expenditures per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: By relying on widely available phosphorus and magnesium-based reagents instead of scarce precious metals, the production process becomes less vulnerable to geopolitical supply shocks. This ensures a steady flow of materials and reduces the risk of production stoppages due to vendor shortages. The robustness of the supply chain is further strengthened by the reduced dependency on specialized waste treatment facilities required for toxic tin byproducts.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction conditions that are easily managed in large reactors. The absence of highly toxic substances simplifies environmental permitting and reduces the liability associated with hazardous waste storage and transport. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific pain points identified in the background art and the beneficial effects highlighted in the patent summary. Understanding these aspects is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines. The answers provide clarity on safety, purity, and scalability, which are the primary concerns for decision-makers in the pharmaceutical industry.
Q: How does this process improve safety compared to traditional carbapenem synthesis?
A: This method eliminates the use of highly toxic organotin compounds and reduces reliance on expensive palladium catalysts found in prior art, significantly enhancing operational safety and environmental compliance.
Q: What are the key purity advantages of this synthetic route?
A: By utilizing specific protecting group strategies and controlled cyclization conditions, the process minimizes epimerization and side reactions, ensuring high stereochemical purity essential for antibacterial efficacy.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the avoidance of hazardous tin reagents and the use of robust phosphorus chemistry make the process highly scalable and easier to manage within standard GMP facilities without specialized heavy metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to ensure the consistent supply of life-saving antibiotics. Our expertise as a CDMO partner allows us to translate complex patent methodologies like CN101255171A into robust commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every batch for impurities and stereochemical integrity, guaranteeing that the intermediates we supply meet the highest global standards.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, compliant, and cost-effective source of high-value pharmaceutical intermediates, ensuring your production lines remain efficient and competitive in the global marketplace.
