Advanced Synthesis of Chiral Oxazoline Catalysts for High-Purity Pharmaceutical Manufacturing
Advanced Synthesis of Chiral Oxazoline Catalysts for High-Purity Pharmaceutical Manufacturing
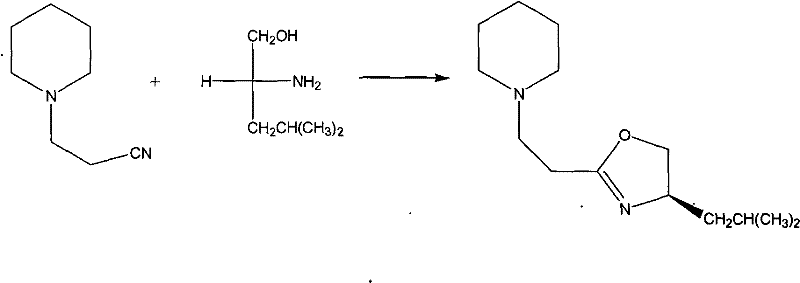
The landscape of asymmetric catalysis is constantly evolving, driven by the relentless demand for enantiomerically pure pharmaceutical intermediates that meet stringent regulatory standards. Patent CN102199130A introduces a significant advancement in this field by detailing a robust and efficient preparation method for a specific chiral oxazoline derivative, identified chemically as 1-[2-4-isobutyl-4,5-dihydro-2-oxazolinyl]-ethyl piperidine. This compound serves as a potent chiral ligand capable of inducing high stereoselectivity in critical carbon-carbon bond-forming reactions, such as the Henry reaction. The innovation lies not just in the molecular structure itself, but in the streamlined synthetic pathway that utilizes accessible starting materials and a cost-effective zinc-based catalytic system. For R&D directors and process chemists, this represents a viable route to generating high-value chiral scaffolds without relying on prohibitively expensive precious metal catalysts or cryogenic conditions. The structural integrity and stereochemical fidelity of this molecule are paramount for its function as a catalyst, ensuring that downstream drug synthesis maintains the necessary optical purity.
![Chemical structure of the chiral oxazoline product 1-[2-4-isobutyl-4,5-dihydro-2-oxazolinyl]-ethyl piperidine](/insights/img/chiral-oxazoline-zinc-catalysis-pharma-supplier-20260312072324-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral oxazolines has often been plagued by significant operational challenges that hinder their widespread adoption in large-scale industrial manufacturing. Traditional routes frequently necessitate the use of harsh dehydrating agents, extremely high temperatures, or toxic solvents that complicate waste management and increase the overall environmental footprint of the process. Furthermore, many conventional methods rely on precious metal catalysts or complex chiral auxiliaries that drive up the raw material costs substantially, making the final intermediate economically unviable for cost-sensitive generic drug production. Purification steps in these older methodologies are often labor-intensive, requiring multiple recrystallizations or preparative HPLC separations to remove trace metal contaminants and diastereomeric impurities. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and inconsistent batch-to-batch quality, which are unacceptable for pharmaceutical companies aiming for rapid time-to-market. The reliance on moisture-sensitive reagents also demands specialized equipment and rigorous anhydrous conditions throughout the entire workflow, adding another layer of complexity and expense to the manufacturing protocol.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in patent CN102199130A offers a streamlined and economically superior alternative that addresses the core pain points of modern chemical manufacturing. By utilizing anhydrous zinc chloride as a Lewis acid catalyst in a chlorobenzene solvent system, this novel approach facilitates the cyclization reaction under standard reflux conditions, eliminating the need for exotic reagents or extreme thermal parameters. The reaction between 3-(1-piperidinyl)propionitrile and D-leucinol proceeds with high efficiency, leveraging the inherent chirality of the amino alcohol to dictate the stereochemical outcome of the final oxazoline ring. This strategy significantly simplifies the workup procedure; post-reaction processing involves simple solvent removal followed by aqueous dissolution and organic extraction, avoiding the need for complex quenching protocols. The result is a cleaner crude product that requires less intensive purification, thereby reducing solvent consumption and waste generation. This shift towards a more atom-economical and operationally simple process directly translates to enhanced scalability, allowing manufacturers to transition from gram-scale laboratory synthesis to multi-kilogram production with minimal process re-engineering.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
To fully appreciate the technical merit of this synthesis, one must delve into the mechanistic role played by the zinc chloride catalyst in driving the cyclization forward. Zinc chloride acts as a potent Lewis acid, coordinating with the nitrogen atom of the nitrile group in the 3-(1-piperidinyl)propionitrile substrate. This coordination significantly increases the electrophilicity of the nitrile carbon, rendering it much more susceptible to nucleophilic attack by the hydroxyl group of the D-leucinol. Once the initial addition occurs, a series of proton transfers and dehydration steps follow, ultimately closing the five-membered oxazoline ring. The presence of the piperidine moiety on the side chain adds a layer of steric bulk that, combined with the isobutyl group from the leucinol, creates a well-defined chiral pocket. This specific three-dimensional architecture is crucial for the molecule's subsequent function as a ligand, as it dictates how the catalyst coordinates with metal centers during asymmetric transformations. Understanding this electronic activation is key for process chemists looking to optimize reaction times or adapt the conditions for continuous flow processing.
Beyond the formation of the ring itself, the preservation of stereochemical integrity throughout the reaction is a critical quality attribute that defines the utility of this intermediate. The use of D-leucinol as the chiral source ensures that the stereocenter at the 4-position of the oxazoline ring is established with high fidelity, as the reaction conditions are mild enough to prevent racemization. The patent data confirms that the resulting product exhibits specific optical rotation values consistent with the expected enantiomer, validating the robustness of the synthetic route. In downstream applications, such as the Henry reaction depicted in the patent, this chiral environment allows the catalyst to differentiate between the prochiral faces of the aldehyde substrate. When complexed with a metal ion, the oxazoline ligand creates a rigid coordination sphere that directs the incoming nitromethane nucleophile to attack from a specific trajectory. This precise control over the transition state geometry is what enables the high enantioselectivity observed in the formation of beta-nitro alcohols, which are valuable precursors for chiral amines and amino acids in drug discovery.

How to Synthesize 1-[2-4-isobutyl-4,5-dihydro-2-oxazolinyl]-ethyl piperidine Efficiently
Implementing this synthesis in a production environment requires strict adherence to the anhydrous and oxygen-free conditions specified in the patent to maximize yield and minimize side reactions. The process begins with the careful charging of anhydrous zinc chloride and the organic substrates into a reactor equipped with a reflux condenser and inert gas inlet. Maintaining a dry atmosphere is essential, as moisture can deactivate the Lewis acid catalyst and hydrolyze the sensitive nitrile intermediate, leading to reduced conversion rates. Following the 24-hour reflux period in chlorobenzene, the reaction mixture is cooled, and the solvent is removed under reduced pressure to isolate the crude residue. The subsequent purification involves dissolving the residue in water and extracting the product into an organic phase using chloroform, effectively separating the organic product from inorganic zinc salts and water-soluble impurities. Final purification via column chromatography using a petroleum ether and dichloromethane eluent system yields the target chiral oxazoline as a colorless oil with high purity. For detailed standard operating procedures and safety guidelines regarding this specific synthesis, please refer to the technical documentation below.
- Combine 3-(1-piperidinyl)propionitrile and D-leucinol with anhydrous ZnCl2 in chlorobenzene under inert atmosphere.
- Reflux the mixture for 24 hours to ensure complete cyclization and formation of the oxazoline ring.
- Remove solvent, dissolve residue in water, extract with chloroform, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this zinc-catalyzed synthesis route offers substantial opportunities for cost optimization and supply chain resilience in the manufacturing of chiral intermediates. The primary driver for cost reduction lies in the replacement of expensive noble metal catalysts with abundant and inexpensive zinc chloride, which drastically lowers the raw material bill of goods. Furthermore, the use of chlorobenzene as a solvent, while requiring careful handling, is a standard industrial solvent that is readily available in bulk quantities, avoiding the supply volatility associated with specialized fluorinated or chlorinated solvents often used in fine chemistry. The simplified workup procedure, which avoids complex distillation or crystallization steps, reduces the energy consumption and labor hours required per batch, contributing to a lower overall cost of goods sold. For supply chain managers, the robustness of this method means fewer failed batches and less variability in production schedules, ensuring a steady flow of materials to downstream formulation teams. This reliability is crucial for maintaining continuity in the production of active pharmaceutical ingredients where interruptions can have severe financial consequences.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of a simple reflux setup significantly lower the capital expenditure and operational costs associated with producing this chiral ligand. By avoiding the need for cryogenic cooling or high-pressure reactors, facilities can utilize existing general-purpose equipment, further deferring capital investment. The high yield reported in the patent data suggests that raw material utilization is efficient, minimizing waste disposal costs and maximizing the output per unit of input. Additionally, the straightforward purification process reduces the volume of solvents required for chromatography or recrystallization, leading to tangible savings in solvent procurement and recovery operations. These cumulative efficiencies create a leaner manufacturing process that is highly competitive in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically 3-(1-piperidinyl)propionitrile and D-leucinol, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. This diversity in the supply base allows procurement teams to negotiate better pricing and secure backup sources in case of logistical disruptions. The stability of the reagents under ambient storage conditions simplifies inventory management, as there is no need for specialized cold storage or inert atmosphere warehousing. Moreover, the scalability of the reflux reaction means that production volumes can be easily ramped up to meet surging demand without requiring extensive process redevelopment. This flexibility ensures that pharmaceutical partners can rely on a consistent supply of high-quality intermediates to support their own clinical and commercial timelines.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing the use of hazardous reagents and reducing the generation of heavy metal waste. Zinc salts are generally less toxic and easier to dispose of or recycle compared to heavy metals like palladium or rhodium, simplifying compliance with increasingly stringent environmental regulations. The ability to run the reaction at atmospheric pressure and moderate temperatures enhances operational safety, reducing the risk of thermal runaways or pressure vessel failures. As production scales from pilot plant to commercial tonnage, these safety and environmental advantages become even more pronounced, facilitating smoother regulatory approvals and community acceptance. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this chiral oxazoline derivative, based on the data provided in the patent literature. Understanding these nuances is essential for R&D teams evaluating this technology for integration into their existing synthetic workflows. The answers provided here reflect the specific experimental conditions and results reported, offering a realistic expectation of performance and limitations. For further customization or specific technical data sheets, direct consultation with our technical team is recommended to ensure the material meets your specific project requirements.
Q: What is the primary advantage of using ZnCl2 in this oxazoline synthesis?
A: ZnCl2 acts as an effective Lewis acid catalyst that activates the nitrile group for nucleophilic attack by the amino alcohol, allowing the reaction to proceed under relatively mild reflux conditions compared to harsher traditional methods.
Q: Can this chiral oxazoline be used for other asymmetric reactions besides the Henry reaction?
A: Yes, while the patent specifically demonstrates efficacy in the Henry reaction of 2-methylbenzaldehyde, oxazoline ligands are broadly applicable in Diels-Alder, Michael, and Friedel-Crafts reactions when complexed with various transition metals.
Q: What is the expected yield and purity profile for this synthetic route?
A: The patent data indicates a yield of approximately 70% for the synthesis of the oxazoline ligand itself, with high stereochemical integrity retained from the D-leucinol starting material, ensuring high enantiomeric excess in downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity, assay, and chiral purity. Our expertise in handling sensitive organometallic reactions and anhydrous processes allows us to replicate the high-yield conditions described in patent CN102199130A with precision and consistency. By partnering with us, you gain access to a supply chain that prioritizes quality, reliability, and technical excellence, enabling you to focus on your core drug discovery objectives without worrying about raw material bottlenecks.
We invite you to engage with our technical procurement team to discuss how this specific chiral oxazoline can enhance your asymmetric synthesis campaigns. We offer a Customized Cost-Saving Analysis to help you evaluate the economic benefits of switching to this zinc-catalyzed route for your specific application. Please contact us to request specific COA data and route feasibility assessments tailored to your project needs. Our goal is to be more than just a vendor; we aim to be a strategic partner in your chemical supply chain, providing the technical support and material reliability necessary to accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
