Advanced Total Synthesis of Trans-Resveratrol for Industrial Scale Pharmaceutical Manufacturing
Advanced Total Synthesis of Trans-Resveratrol for Industrial Scale Pharmaceutical Manufacturing
The global demand for trans-resveratrol, a potent polyphenolic antioxidant with significant applications in cardiovascular health, anti-aging formulations, and oncology research, has historically outpaced the sustainable supply capabilities of traditional extraction methods. As natural resources face depletion and regulatory scrutiny regarding botanical sourcing intensifies, the pharmaceutical and nutraceutical industries are urgently seeking robust, synthetic alternatives that guarantee purity and continuity. Patent CN103570508A presents a breakthrough total synthesis method that addresses these critical supply chain vulnerabilities through a refined five-step chemical pathway. This innovative approach transforms readily available 3,5-dihydroxybenzoic acid into high-purity trans-resveratrol, bypassing the limitations of biological extraction while achieving exceptional yields and stereo-selectivity. By leveraging a strategic combination of methylation, reduction, halogenation, Wittig-Horner coupling, and Lewis acid-mediated demethylation, this process establishes a new benchmark for the reliable trans-resveratrol supplier market, ensuring that downstream manufacturers can secure high-quality active ingredients without reliance on fluctuating agricultural outputs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of trans-resveratrol has been fraught with significant logistical and technical challenges that hinder consistent commercial scale-up of complex pharmaceutical intermediates. The traditional reliance on extraction from sources such as Polygonum cuspidatum or grape skins introduces inherent variability in potency and purity, driven by seasonal harvest cycles, geographical origin differences, and the co-extraction of unwanted plant metabolites that require costly purification steps. Furthermore, alternative synthetic routes like the Heck reaction, while theoretically viable, often necessitate the use of expensive palladium catalysts and severe reaction conditions that pose safety risks and complicate the removal of trace heavy metals to meet stringent pharmacopeial standards. Similarly, the Perkin reaction, though utilizing accessible raw materials, frequently suffers from long reaction schemes and the generation of cis-isomers alongside the desired trans-product, drastically reducing overall efficiency and increasing waste disposal burdens. These conventional methodologies collectively create a bottleneck for procurement managers seeking cost reduction in fine chemical manufacturing, as the cumulative effect of low yields, expensive catalysts, and complex purification protocols inflates the final cost of goods sold.
The Novel Approach
In stark contrast to these legacy methods, the synthesis route disclosed in CN103570508A offers a streamlined, economically viable pathway that prioritizes mild reaction conditions and high atom economy. This novel approach initiates with the efficient methylation of 3,5-dihydroxybenzoic acid, followed by a highly selective reduction using potassium borohydride and lithium chloride, which avoids the use of more hazardous reducing agents like lithium aluminum hydride. The subsequent conversion to the benzyl bromide intermediate and the pivotal Wittig-Horner coupling reaction proceed with remarkable precision, ensuring the exclusive formation of the thermodynamically stable trans-isomer without the need for difficult chromatographic separations. The final demethylation step utilizes aluminum trichloride in a triethylamine system, a modification that significantly enhances reaction control compared to traditional strong acid hydrolysis, thereby preserving the integrity of the sensitive stilbene double bond. This comprehensive strategy not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
Mechanistic Insights into Wittig-Horner Coupling and Demethylation
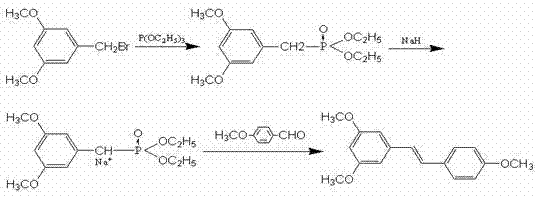
The core of this synthetic success lies in the meticulous optimization of the Wittig-Horner reaction, a variation of the classic Wittig olefination that utilizes phosphonate esters to achieve superior E-selectivity. In this specific embodiment, the reaction between 3,5-dimethoxybenzyl bromide and triethyl phosphite generates a phosphonate intermediate in situ, which is subsequently deprotonated by sodium hydride to form a stabilized carbanion. This nucleophilic species then attacks the carbonyl carbon of anisaldehyde (4-methoxybenzaldehyde), proceeding through a betaine intermediate to form the carbon-carbon double bond with high stereospecificity favoring the trans-configuration. The use of phosphonate esters rather than phosphonium ylides reduces the basicity of the reaction medium, minimizing side reactions such as aldol condensation of the aldehyde, which is a common impurity source in less optimized processes. This mechanistic precision ensures that the resulting 3,5,4'-trimethoxystilbene is obtained with minimal isomeric contamination, drastically reducing the burden on downstream purification units.
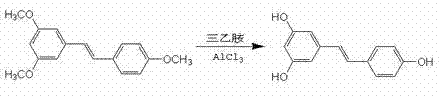
Following the construction of the stilbene backbone, the final transformation involves the cleavage of the methyl ether protecting groups to reveal the bioactive phenolic hydroxyls. This step employs anhydrous aluminum trichloride coordinated with triethylamine in a toluene solvent system, a sophisticated modification of the classic Lewis acid demethylation. The aluminum center coordinates strongly with the ether oxygen, facilitating the nucleophilic attack by chloride ions or the subsequent hydrolysis upon workup, effectively stripping the methyl groups under relatively mild thermal conditions (80 °C to 100 °C). This controlled environment prevents the isomerization of the double bond or the degradation of the electron-rich aromatic rings, which can occur under harsher acidic conditions. The result is a final product with a purity profile exceeding 98%, demonstrating the robustness of this chemical architecture in maintaining molecular integrity throughout the synthesis.
How to Synthesize Trans-Resveratrol Efficiently
The execution of this total synthesis requires precise control over stoichiometry and temperature profiles to maximize yield and minimize byproduct formation, particularly during the exothermic methylation and reduction phases. Operators must ensure strict anhydrous conditions during the formation of the phosphonate intermediate and the subsequent Wittig-Horner coupling to prevent hydrolysis of the reactive species. The detailed standardized synthetic steps, including specific mass ratios for reagents like 3,5-dihydroxybenzoic acid to dimethyl sulfate (1:1 to 1:3) and the specific thermal ramps for the coupling reaction, are critical for reproducibility. For a complete breakdown of the operational parameters and safety protocols required for implementation, please refer to the technical guide below.
- Methylation of 3,5-dihydroxybenzoic acid with dimethyl sulfate and potassium carbonate to form 3,5-dimethoxy methyl benzoate.
- Reduction of the ester to 3,5-dimethoxybenzyl alcohol using potassium borohydride and lithium chloride.
- Nucleophilic substitution with phosphorus tribromide to generate 3,5-dimethoxybenzyl bromide.
- Wittig-Horner reaction with triethyl phosphite and anisaldehyde to form the stilbene backbone.
- Final demethylation using aluminum trichloride and triethylamine to yield pure trans-resveratrol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route represents a strategic shift towards greater stability and cost predictability in the sourcing of high-value antioxidants. By eliminating the dependency on agricultural harvests, manufacturers can decouple their production schedules from seasonal fluctuations and geopolitical risks associated with raw botanical imports. The use of commodity chemicals such as dimethyl sulfate, potassium carbonate, and common solvents like acetone and toluene ensures that the raw material basket remains liquid and competitively priced, shielding the project from the volatility often seen in specialized catalytic reagents. Furthermore, the high yield reported in the patent embodiments suggests a substantial reduction in waste generation per kilogram of product, which translates directly into lower disposal costs and a smaller environmental footprint, aligning with increasingly rigorous corporate sustainability mandates.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts, such as palladium used in Heck couplings, removes a significant line item from the bill of materials and obviates the need for expensive metal scavenging processes to meet residual metal specifications. Additionally, the high selectivity of the Wittig-Horner step minimizes the formation of difficult-to-separate isomers, reducing the consumption of chromatography media and solvents during purification. This streamlined process flow allows for significant operational expenditure savings, making the final trans-resveratrol product more price-competitive in the crowded nutraceutical market while maintaining healthy margins for producers.
- Enhanced Supply Chain Reliability: The starting material, 3,5-dihydroxybenzoic acid, is a widely available industrial chemical with a mature supply chain, ensuring that production can be ramped up quickly to meet surges in market demand without long lead times for precursor sourcing. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic high-pressure or cryogenic setups, further enhances the reliability of the manufacturing process. This accessibility allows for multi-sourcing strategies where different contract manufacturing organizations can replicate the process with high fidelity, mitigating the risk of single-point failures in the supply network.
- Scalability and Environmental Compliance: The patent data indicates successful translation from laboratory scale to 50L pilot reactors, demonstrating that the heat transfer and mixing requirements are manageable at larger volumes. The avoidance of chlorinated solvents in certain steps and the use of recyclable solvents like toluene and ethyl acetate simplify waste stream management and facilitate compliance with environmental regulations. The overall process design favors continuous or semi-continuous operation modes, offering a clear pathway for scaling production from hundreds of kilograms to multi-ton annual capacities without fundamental changes to the chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived from the specific advantages and operational details outlined in the patent documentation. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers provided reflect the consensus on best practices for optimizing yield and purity while maintaining safety standards.
Q: What are the advantages of this synthesis method over plant extraction?
A: Unlike plant extraction which suffers from resource depletion and seasonal variability, this total synthesis method offers a consistent, scalable supply chain with high purity (over 98%) and eliminates the risk of natural impurities.
Q: How does this route compare to traditional Heck or Perkin reactions?
A: This method avoids the harsh conditions and expensive palladium catalysts required for Heck reactions, and prevents the formation of isomers common in Perkin reactions, resulting in a cleaner product profile and lower processing costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful execution in 50L reactors with high yields, utilizing common reagents and mild conditions that are easily adaptable for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis method are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch, guaranteeing that our trans-resveratrol meets the exacting standards required for pharmaceutical and high-end nutraceutical applications.
We invite you to collaborate with us to optimize your supply chain and secure a competitive advantage in the antioxidant market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term growth objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
