Advanced Manufacturing of Ketorolac Tromethamine: A Breakthrough in Process Efficiency and Purity Control
Introduction to Next-Generation Ketorolac Tromethamine Manufacturing
The pharmaceutical landscape for non-steroidal anti-inflammatory drugs (NSAIDs) is constantly evolving, driven by the demand for higher purity analgesics suitable for parenteral administration. Patent CN101575340A introduces a transformative preparation method for Ketorolac Tromethamine, a potent analgesic widely used for postoperative and cancer-related pain management. This technical disclosure addresses critical bottlenecks in traditional synthesis, specifically the issues of low purity, poor coloration due to oxidation, and complex processing steps. By leveraging a unique alkaline hydrolysis pathway coupled with spontaneous decarboxylation, this method achieves a total two-step yield of up to 95% and a purity exceeding 99.9%. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, ensuring consistent quality for injection-grade formulations while mitigating the risks associated with oxidative degradation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ketorolac Tromethamine has been plagued by inefficiencies and safety concerns inherent to older patent methodologies, such as those described in US5082950 and US5532381. Traditional routes typically involve the hydrolysis of diethyl esters in biphasic systems of ether and aqueous sodium hydroxide, a process that is notoriously slow, often requiring reaction times extending up to 24 hours to reach completion. Furthermore, these legacy methods necessitate a distinct and energy-intensive thermal decarboxylation step post-acidification, adding significant operational complexity and cost. Perhaps most critically from a regulatory and safety standpoint, conventional salification processes frequently employ benzene as a solvent, introducing severe toxicity risks and challenging solvent residue limits that complicate downstream purification. The susceptibility of the ketorolac intermediate to oxidation in these environments often results in products with suboptimal color and purity profiles, rendering them unsuitable for high-specification injectable applications without extensive and costly reprocessing.
The Novel Approach
In stark contrast, the methodology disclosed in CN101575340A offers a streamlined, robust alternative that fundamentally reengineers the reaction pathway. The core innovation lies in the discovery that decarboxylation occurs spontaneously during the acidification of the hydrolyzed intermediate, effectively merging two distinct unit operations into one seamless transition. This eliminates the need for the cumbersome thermal decarboxylation step entirely. The process utilizes water-miscible alcoholic solvents such as methanol or ethanol, which are not only safer and more environmentally benign than benzene but also facilitate homogeneous reaction conditions that drastically reduce reaction times to between 2 and 6 hours. By refining the ketorolac prior to salification and introducing specific reducing agents during the final salt formation, this approach proactively manages oxidative impurities. This results in a white crystalline product with exceptional clarity and stability, perfectly aligned with the rigorous demands of modern pharmaceutical manufacturing.
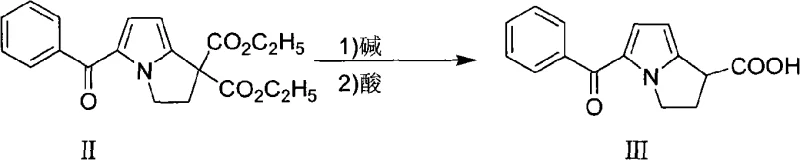
Mechanistic Insights into Alkaline Hydrolysis and Reductive Salification
The chemical elegance of this process is rooted in the precise control of pH and redox potential during the transformation of 5-benzoyl-2,3-dihydro-1H-pyrrole-1,1-diethyl dicarboxylate (Compound II) into the final active salt. During the initial alkaline hydrolysis phase, the ester groups are cleaved under mild thermal conditions (20-90°C) using sodium or potassium hydroxide. Upon subsequent acidification to a pH of 2-3 at controlled low temperatures (-20 to 25°C), the resulting diacid intermediate undergoes immediate and spontaneous decarboxylation. This phenomenon suggests a highly favorable thermodynamic pathway where the instability of the gem-dicarboxylic acid moiety drives the rapid evolution of carbon dioxide, yielding the free acid ketorolac (Compound III) directly in the precipitation step. This mechanistic shortcut not only saves energy but also minimizes the exposure of the sensitive pyrrole ring to prolonged harsh conditions that could otherwise lead to polymerization or degradation.
Furthermore, the integrity of the final product is safeguarded through a sophisticated redox management strategy during the salification with Trometamol. Ketorolac is inherently prone to oxidation, which manifests as discoloration and the formation of trace impurities that are difficult to remove. The patented method counters this by incorporating reducing agents such as sodium thiosulfate, sodium hydrosulfite, or potassium sulfite directly into the reaction matrix. These agents act as sacrificial scavengers, neutralizing dissolved oxygen and reactive oxidative species before they can attack the drug substance. This ensures that the resulting Ketorolac Tromethamine maintains a pristine white appearance and a purity profile greater than 99.9%, effectively solving the chronic 'color difference' problems that have historically plagued the commercial production of this vital analgesic intermediate.
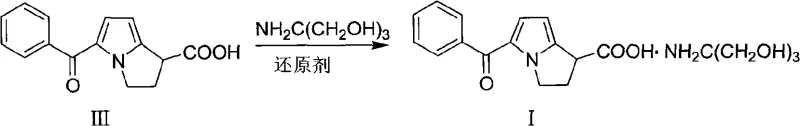
How to Synthesize Ketorolac Tromethamine Efficiently
Implementing this advanced synthesis route requires careful attention to solvent selection and temperature control to maximize the benefits of the spontaneous decarboxylation mechanism. The process begins with the suspension of the diethyl ester starting material in a lower alcohol solvent, followed by the addition of an aqueous alkali solution. Operators must monitor the reaction via TLC to ensure complete hydrolysis before proceeding to the critical acidification stage, where temperature control is paramount to ensure proper precipitation and crystal habit of the intermediate ketorolac. Following isolation and optional recrystallization to further enhance purity, the final salification is conducted in a refluxing alcohol solvent with the stoichiometric addition of Trometamol and the chosen reducing agent. For a comprehensive, step-by-step standard operating procedure including specific molar ratios and workup details, please refer to the standardized guide below.
- Perform alkaline hydrolysis of 5-benzoyl-2,3-dihydro-1H-pyrrole-1,1-diethyl dicarboxylate using NaOH or KOH in alcoholic solvents at 20-90°C.
- Acidify the reaction mixture to pH 2-3 at low temperature (-20 to 25°C) to induce spontaneous decarboxylation and precipitate Ketorolac.
- React purified Ketorolac with Trometamol in alcohol solvent with a reducing agent (e.g., sodium thiosulfate) to form the final salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages regarding cost structure, lead times, and regulatory compliance. By eliminating the thermal decarboxylation step and reducing reaction times from nearly a day to mere hours, the overall throughput of the manufacturing facility is significantly increased, allowing for greater production capacity without additional capital expenditure. The substitution of toxic solvents like benzene with common alcohols simplifies waste treatment protocols and reduces the costs associated with hazardous material handling and disposal. Moreover, the high intrinsic purity of the crude product reduces the burden on downstream purification teams, minimizing yield losses typically associated with aggressive recrystallization or chromatography steps. This efficiency creates a more resilient supply chain capable of responding rapidly to market demand fluctuations for this essential pain management medication.
- Cost Reduction in Manufacturing: The elimination of the separate thermal decarboxylation step represents a direct reduction in energy consumption and processing time. By combining hydrolysis and decarboxylation into a single operational sequence, manufacturers can drastically lower utility costs and labor hours per batch. Additionally, the use of inexpensive, commodity-grade solvents like ethanol and methanol, instead of specialized or toxic solvents like benzene and ethyl acetate, further optimizes the raw material cost base, delivering substantial cost savings in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemical process, characterized by high yields (up to 95% over two steps) and tolerance to standard industrial conditions, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines to downstream API formulators. The simplified workflow reduces the number of potential failure points in the production line, thereby minimizing the risk of batch rejections or delays. Consequently, this leads to reducing lead time for high-purity pharmaceutical intermediates, ensuring that critical medications reach the market without interruption.
- Scalability and Environmental Compliance: Designed with green chemistry principles in mind, this method avoids the use of carcinogenic solvents, aligning perfectly with increasingly stringent global environmental regulations. The aqueous-alcoholic solvent system is easier to recover and recycle, reducing the environmental footprint of the manufacturing process. The high purity of the final product (>99.9%) ensures compliance with strict pharmacopoeial standards for injectables, facilitating smoother regulatory filings and inspections. This makes the commercial scale-up of complex pharmaceutical intermediates not only technically feasible but also environmentally sustainable and commercially viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of yield, purity, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: How does the new method improve the color and purity of Ketorolac Tromethamine?
A: The novel process incorporates specific reducing agents during the salt formation step, which effectively prevents the oxidation of the ketorolac molecule. This results in a product with purity exceeding 99.9% and color quality superior to Light Yellow No. 1, meeting injection-grade standards.
Q: Does this synthesis route require a separate thermal decarboxylation step?
A: No. A key innovation of this method is that decarboxylation occurs spontaneously during the acidification phase of the hydrolysis step. This eliminates the need for a separate, energy-intensive thermal decarboxylation process required in conventional methods.
Q: What solvents are used in this environmentally friendly process?
A: The process utilizes water-miscible alcoholic solvents such as methanol, ethanol, or isopropanol for hydrolysis, and avoids toxic aromatic solvents like benzene, significantly reducing environmental impact and solvent residue risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketorolac Tromethamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-quality synthesis routes for essential pharmaceutical ingredients like Ketorolac Tromethamine. Our team of expert chemists has extensively analyzed the technological potential of the hydrolysis-decarboxylation pathway described in CN101575340A and is fully prepared to translate this laboratory-scale innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are realized in tangible output. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for global pharmaceutical markets.
We invite you to collaborate with us to optimize your supply chain for this vital analgesic intermediate. By leveraging our expertise in process chemistry and scale-up engineering, we can help you achieve significant cost reductions and supply security. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring a seamless partnership from development to commercial delivery.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
