Advanced Synthesis of Ketorolac Tromethamine Intermediates for Commercial Scale Production
Advanced Synthesis of Ketorolac Tromethamine Intermediates for Commercial Scale Production
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with environmental sustainability, particularly for potent analgesics like Ketorolac tromethamine. Patent CN113087619A introduces a groundbreaking approach to synthesizing a critical intermediate, Compound IV, which serves as a pivotal building block in the production of this nonsteroidal anti-inflammatory drug. This novel methodology addresses long-standing challenges associated with traditional oxidation processes, offering a cleaner, more efficient route that aligns with modern green chemistry principles. By shifting away from heavy metal oxidants towards a controlled acyl chloride formation strategy, manufacturers can achieve superior product quality while significantly mitigating environmental liabilities. The structural integrity of the final active pharmaceutical ingredient relies heavily on the precision of these upstream intermediate steps.
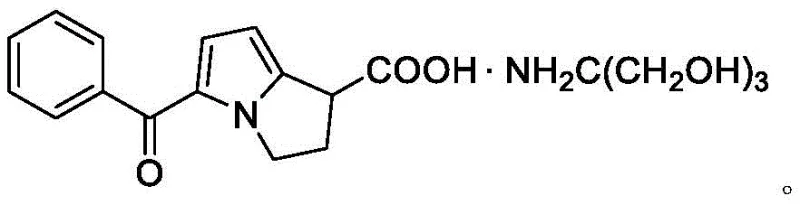
Ketorolac tromethamine is a powerful analgesic widely used for managing moderate to severe postoperative pain, functioning through the inhibition of prostaglandin synthesis. The commercial viability of this API is directly linked to the efficiency of its supply chain, specifically the availability of high-quality intermediates. The introduction of Compound IV represents a strategic advancement in pharmaceutical intermediates manufacturing, providing a reliable foundation for downstream cyclization and functionalization. For procurement leaders and R&D directors, understanding the nuances of this new pathway is essential for optimizing cost structures and ensuring consistent supply continuity in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ketorolac tromethamine has relied on oxidative cyclization strategies that present significant operational and environmental hurdles. As illustrated in prior art, such as the route reported by Wanghua et al., the process typically involves the use of excessive potassium permanganate and manganese acetate to construct the key diester-based intermediate. While chemically feasible, this traditional approach generates substantial quantities of manganese-containing wastewater, which is notoriously difficult and costly to treat to meet regulatory discharge standards. Furthermore, the use of strong oxidants often leads to over-oxidation side reactions, resulting in a crude product with a dark color that necessitates rigorous purification.
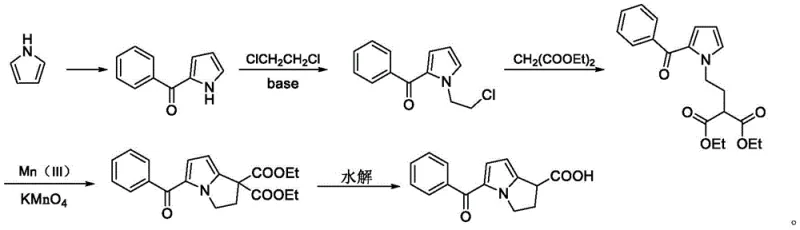
The post-treatment requirements for these conventional methods are equally burdensome, often demanding multiple decolorization steps using activated carbon or silica gel to remove colored impurities and residual metal salts. This not only increases the consumption of auxiliary materials but also generates significant solid waste, complicating the disposal process and inflating the overall production cost. From a supply chain perspective, the reliance on heavy metal oxidants introduces variability in batch consistency and poses potential risks regarding residual metal limits in the final API. These factors collectively diminish the economic attractiveness and sustainability of the legacy manufacturing processes, creating a pressing need for innovation in cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
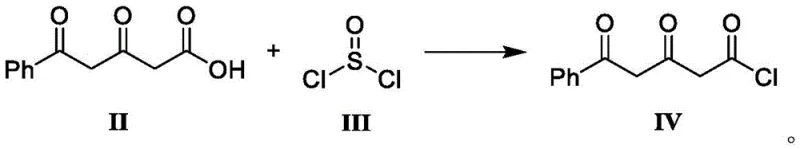
In stark contrast to the oxidative struggles of the past, the novel approach detailed in CN113087619A leverages a precise acyl chloride intermediate strategy to bypass the need for harsh oxidants entirely. The core of this innovation lies in the preparation of Compound IV, a specialized acid chloride derivative that acts as a highly reactive electrophile for subsequent coupling reactions. By converting the corresponding carboxylic acid (Compound II) into its acid chloride form using thionyl chloride under mild conditions, the process establishes a clean and controllable pathway for carbon-carbon bond formation. This shift fundamentally alters the impurity profile, eliminating the source of dark-colored byproducts and heavy metal contamination at the source.
The operational simplicity of this new route is a major advantage for industrial adoption. The reaction conditions are温和 (mild), typically proceeding at temperatures between 10-35°C in common solvents like tetrahydrofuran, which reduces energy consumption and safety risks associated with exothermic runaway reactions. Moreover, the work-up procedure is streamlined; after the reaction is complete, simple filtration and concentration suffice to isolate Compound IV with exceptional purity, often exceeding 99.7%. This elimination of complex purification steps translates directly into reduced cycle times and lower operational expenditures, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acyl Chloride Formation and Cyclization
The mechanistic elegance of this synthesis begins with the activation of the carboxylic acid group in Compound II. Upon treatment with thionyl chloride, the hydroxyl group is replaced by a chlorine atom, generating the highly electrophilic acyl chloride functionality found in Compound IV. This transformation is critical because it primes the molecule for nucleophilic attack by the enolate derived from Compound V. The use of thionyl chloride is particularly advantageous as the byproducts, sulfur dioxide and hydrogen chloride, are gases that easily escape the reaction mixture, driving the equilibrium forward and simplifying the isolation of the product. This gas-evolving mechanism ensures that the reaction proceeds to completion without the need for extensive aqueous work-ups that could hydrolyze the sensitive acid chloride.
Following the formation of Compound IV, the subsequent alkylation and cyclization steps demonstrate remarkable chemoselectivity. When Compound IV reacts with the anion of Compound V (generated using a strong base like sodium hydride), it forms the linear precursor Compound VI. The final cyclization to form the pyrrolizine ring system (Compound I) is achieved through treatment with ammonia. This intramolecular condensation is highly efficient, closing the ring to form the core structure of the Ketorolac skeleton. The entire sequence avoids the chaotic radical mechanisms associated with permanganate oxidation, resulting in a much cleaner reaction profile. This mechanistic control is paramount for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the formation of hard-to-remove regioisomers and degradation products.
How to Synthesize Ketorolac Tromethamine Intermediate Efficiently
The practical execution of this synthesis requires careful attention to temperature control and stoichiometry to maximize yield and purity. The process is divided into distinct stages: the activation of the acid, the coupling reaction, and the final cyclization. Each step has been optimized in the patent examples to demonstrate robustness across different solvent systems and bases. For R&D teams looking to implement this technology, the following guide outlines the standardized operational parameters derived from the patent data, ensuring a smooth transition from laboratory bench to pilot plant.
- Prepare Compound IV by reacting Compound II with thionyl chloride in an organic solvent like tetrahydrofuran at controlled temperatures between 10-35°C.
- Synthesize Compound VI by reacting Compound V with a strong base followed by the addition of Compound IV at low temperatures (-30 to -10°C).
- Complete the cyclization to form Compound I by reacting Compound VI with an ammonia solution in an autoclave at 50-80°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The primary value proposition lies in the drastic simplification of the waste management profile. By eliminating the use of manganese salts and potassium permanganate, manufacturers avoid the costly and logistically complex disposal of heavy metal-contaminated wastewater. This not only reduces direct disposal costs but also mitigates regulatory risks associated with environmental compliance, ensuring uninterrupted production schedules. The ability to operate without specialized waste treatment infrastructure for heavy metals significantly lowers the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the reduction in auxiliary material consumption and energy usage. Traditional methods require significant amounts of decolorizing agents like activated carbon and silica gel to manage the dark color of the crude product, which adds material costs and extends processing time. The new method produces a light-colored product with high inherent purity, effectively removing the need for these expensive purification consumables. Furthermore, the high yields reported (often above 95% for intermediate steps) mean that less raw material is wasted, directly improving the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis and the availability of specialized reagents. This new route utilizes commodity chemicals such as thionyl chloride, sodium hydride, and ammonia, which are readily available in the global chemical market. Unlike specialized oxidants that may face supply constraints or price volatility, these reagents ensure a stable supply chain. Additionally, the simplified work-up procedures reduce the risk of batch failures due to operational errors, leading to more predictable delivery timelines for reliable pharmaceutical intermediates supplier networks.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing challenges, particularly with exothermic oxidations. The mild conditions of this new synthesis (near ambient temperatures) make it inherently safer and easier to scale from kilograms to metric tons. The absence of heavy metal waste aligns perfectly with increasingly stringent global environmental regulations, such as REACH in Europe. This compliance advantage future-proofs the manufacturing process, preventing potential shutdowns due to regulatory changes and positioning the manufacturer as a leader in sustainable high-purity pharmaceutical intermediates production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer.
Q: How does the new method improve impurity profiles compared to traditional routes?
A: The new method avoids the use of excessive potassium permanganate and manganese acetate, which traditionally cause dark product color and difficult wastewater treatment. By utilizing an acyl chloride intermediate (Compound IV), the process achieves HPLC purity exceeding 99.7% without requiring multi-step decolorization with activated carbon or silica gel.
Q: What are the optimal reaction conditions for synthesizing Compound IV?
A: According to patent CN113087619A, the optimal conditions involve using tetrahydrofuran as the solvent with a molar ratio of Compound II to thionyl chloride of approximately 1:1.4. The reaction is best conducted at mild temperatures between 20-25°C to ensure high yield (up to 98.5%) and minimize side reactions.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial scalability. It eliminates toxic heavy metal oxidants, simplifies post-treatment by avoiding complex filtration of metal salts, and uses common organic solvents. The operational simplicity and high yields make it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketorolac Tromethamine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN113087619A are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for API synthesis, providing our partners with peace of mind regarding quality and consistency.
We invite you to collaborate with us to optimize your supply chain for Ketorolac tromethamine production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this new intermediate can lower your overall manufacturing costs. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global analgesic market through superior chemical innovation.
