Industrial Scale-Up of Drospirenone: A Safer TEMPO-Catalyzed Oxidation Route
Industrial Scale-Up of Drospirenone: A Safer TEMPO-Catalyzed Oxidation Route
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex steroid intermediates that balance high purity with environmental sustainability. Patent CN101065396A introduces a transformative methodology for the preparation of drospirenone, a synthetic progestogen widely utilized in contraceptive formulations. This technical disclosure addresses critical bottlenecks in existing synthesis routes by replacing hazardous oxidants with a catalytic TEMPO-based system. The core innovation lies in the oxidation of 17α-(3-hydroxypropyl)-6β,7β;15β,16β-dimethylene-5β-androstane-3β,5,17β-triol directly to the final lactone structure. By integrating this advanced oxidation step with a streamlined purification protocol, the process delivers a reliable pharma intermediate supplier solution that mitigates the risks associated with heavy metal contamination and toxic solvent usage.
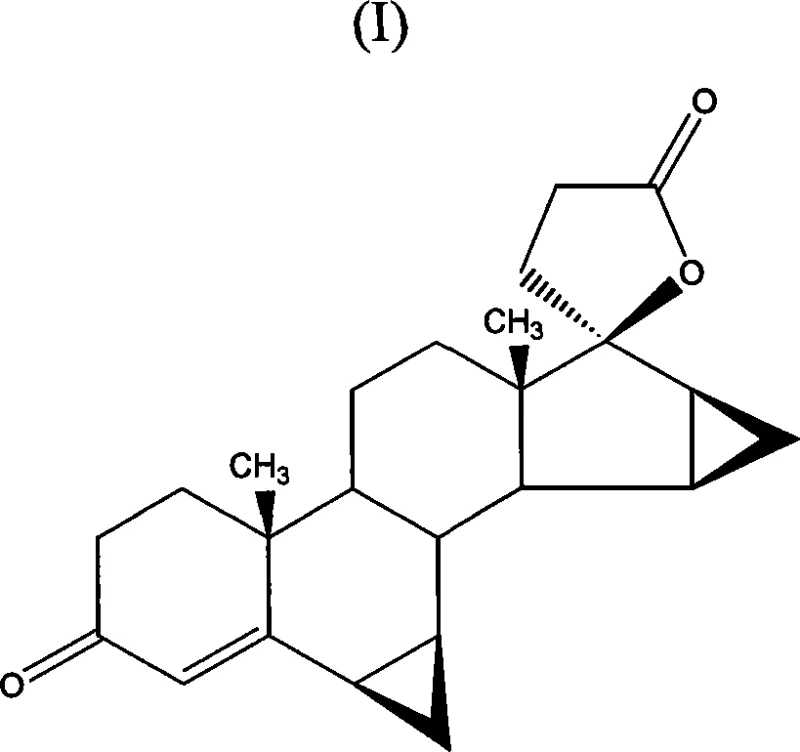
The strategic value of this patent extends beyond mere chemical conversion; it represents a paradigm shift towards greener chemistry in steroid manufacturing. Traditional methods often rely on stoichiometric amounts of dangerous reagents that complicate waste disposal and increase operational costs. In contrast, the disclosed method utilizes catalytic amounts of 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO) derivatives alongside benign oxidants like calcium hypochlorite. This approach not only enhances the safety profile of the production facility but also simplifies the downstream processing requirements. For procurement managers and supply chain heads, understanding the implications of such technological upgrades is vital for securing long-term supply continuity and cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of drospirenone has been plagued by significant safety and efficiency challenges inherent to older patent literature, such as EP 0075189. A primary drawback of these conventional routes is the reliance on carbon tetrachloride (CCl4) as both a reactant and solvent during the bromination steps. CCl4 is a highly toxic substance with severe environmental restrictions, necessitating specialized containment equipment and rigorous monitoring protocols that drive up capital expenditure. Furthermore, the final oxidation step in traditional processes frequently employs chromium trioxide (CrO3) in pyridine, a reagent classified as a known carcinogen. The legislative restrictions surrounding Cr(VI) compounds impose heavy burdens on manufacturers, requiring extensive safety measures for handling, storage, and waste treatment, which ultimately inflates the cost of goods sold and complicates regulatory compliance.
The Novel Approach
The methodology described in CN101065396A offers a compelling alternative by fundamentally redesigning the oxidation and purification stages. Instead of relying on stoichiometric chromium oxidants, the novel approach leverages a catalytic cycle involving TEMPO and hypohalites, which operates under mild conditions (20-25°C) and generates significantly less hazardous waste. Crucially, this process eliminates the absolute necessity for industrial-scale chromatographic purification, a unit operation that is notoriously expensive and difficult to scale for steroid batches. By achieving crude purity levels exceeding 96.5% through reaction control and subsequent crystallization, the new route bypasses the logistical and economic hurdles of column chromatography. This shift enables a more streamlined production flow, enhancing the commercial scale-up of complex pharmaceutical intermediates while maintaining the stringent quality standards required for active pharmaceutical ingredients.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Lactonization
The heart of this technological advancement is the selective oxidation mechanism mediated by the nitroxyl radical catalyst. In the presence of a co-oxidant such as calcium hypochlorite, the TEMPO catalyst facilitates the conversion of the primary alcohol side chain on the steroid backbone into an aldehyde intermediate, which subsequently undergoes intramolecular cyclization. This reaction is performed in a biphasic system comprising dichloromethane and tetrahydrofuran, buffered with aqueous sodium bicarbonate to maintain optimal pH levels. The mild reaction conditions prevent the degradation of sensitive functional groups elsewhere on the steroid nucleus, such as the cyclopropane rings and the enone system, which are prone to isomerization under harsh acidic or basic conditions. The precision of this catalytic system ensures high regioselectivity, minimizing the formation of over-oxidized byproducts that typically plague non-catalytic oxidation methods.
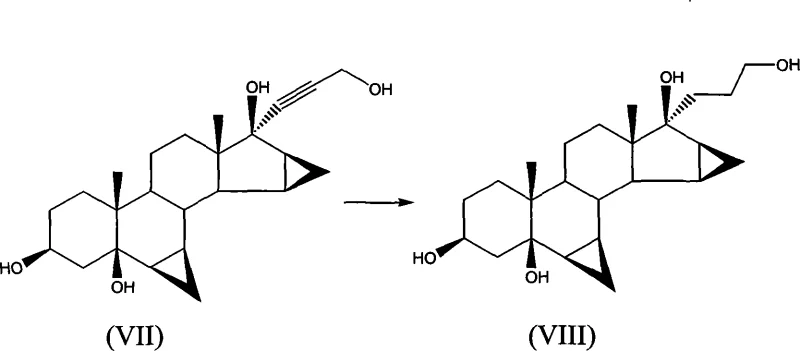
Following the oxidation, the process incorporates a direct acid-mediated lactonization step within the same reactor vessel. By introducing a protonic acid, such as p-toluenesulfonic acid monohydrate, directly into the organic phase, the intermediate hemiacetal is dehydrated to form the stable 17α-lactone ring characteristic of drospirenone. This telescoped operation reduces the number of isolation steps, thereby improving overall yield and reducing solvent consumption. From an impurity control perspective, the subsequent purification protocol involving silica gel and decolorizing carbon is specifically designed to remove the "reversed lactone" impurity (ZK35096). This specific structural isomer is a known challenge in drospirenone synthesis, and its effective removal without chromatography demonstrates the robustness of the crystallization parameters defined in the patent, ensuring the final product meets high-purity pharmaceutical intermediate specifications.
How to Synthesize Drospirenone Efficiently
The synthesis of this complex steroid requires a precise sequence of transformations to construct the unique 6β,7β;15β,16β-dimethylene framework before the final oxidation can occur. The precursor preparation involves a series of well-defined steps including bromination, epoxide opening, and methylenation, each optimized to maximize stereochemical integrity. The detailed standardized synthesis steps see the guide below outlines the specific reagents and conditions required to navigate this multi-step pathway successfully. Adhering to these protocols ensures that the substrate fed into the final oxidation reactor possesses the necessary purity profile to support the high-efficiency TEMPO catalysis.
- Prepare the precursor 17α-(3-hydroxypropyl)-6β,7β;15β,16β-dimethylene-5β-androstane-3β,5,17β-triol through bromination, epoxide opening, hydrolysis, methylenation, propargyl addition, and hydrogenation.
- Oxidize the triol precursor using catalytic TEMPO and calcium hypochlorite in a dichloromethane/THF solvent system at 20-25°C.
- Add protonic acid directly to the reaction mixture to induce lactonization, followed by filtration and crystallization to achieve >99.5% purity without chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the adoption of this patented process translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of chromatographic purification represents a major operational improvement, as industrial chromatography requires significant investment in specialized resins, large volumes of solvents, and dedicated equipment that often becomes a bottleneck in production schedules. By replacing this with a filtration and crystallization workflow, manufacturers can drastically simplify their production lines, leading to substantial cost savings in both fixed assets and variable operating expenses. Furthermore, the avoidance of restricted substances like carbon tetrachloride and chromium trioxide removes the regulatory friction that often delays batch releases or triggers costly environmental audits, thereby smoothing the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this TEMPO-based route is driven primarily by the reduction in raw material costs and waste disposal fees. Chromium salts and toxic halogenated solvents carry high procurement prices and even higher costs for compliant disposal. By utilizing cheaper, safer oxidants like calcium hypochlorite and common solvents, the variable cost per kilogram of product is significantly lowered. Additionally, the catalytic nature of the TEMPO reagent means that only minute quantities are needed compared to stoichiometric oxidants, further driving down the material cost basis. This efficiency allows for a more competitive pricing structure without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized or regulated chemicals. Since this process relies on commodity chemicals such as sodium bicarbonate, calcium hypochlorite, and standard organic solvents, the risk of supply disruption due to regulatory bans or vendor shortages is minimized. The robustness of the synthesis also means that batch failure rates are likely reduced, as the process is less sensitive to the variations that can occur with hazardous reagents. This stability ensures that procurement managers can secure long-term contracts with greater confidence, knowing that the manufacturing partner has a resilient and compliant production capability.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, particularly when dealing with exothermic oxidations or toxic volatiles. The mild temperature range (20-25°C) and the absence of highly toxic reagents make this process inherently safer to scale from pilot plant to commercial tonnage. The simplified waste stream, devoid of heavy metals and persistent organic pollutants, aligns with modern environmental, social, and governance (ESG) goals. This compliance advantage not only protects the manufacturer from liability but also appeals to downstream pharmaceutical clients who are increasingly scrutinizing the environmental footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these details helps technical teams evaluate the feasibility of adopting this process for their own manufacturing needs or sourcing strategies.
Q: How does this process improve safety compared to traditional methods?
A: This method eliminates the use of carcinogenic chromium trioxide (CrO3) and toxic carbon tetrachloride (CCl4), replacing them with safer TEMPO catalysts and calcium hypochlorite, significantly reducing regulatory and handling risks.
Q: Is chromatographic purification required for high purity?
A: No, the process achieves high purity (>99.5%) through a specialized purification sequence involving silica gel and decolorizing carbon filtration followed by crystallization, removing the need for expensive industrial chromatography.
Q: What specific impurity is removed by the new purification method?
A: The purification process effectively eliminates the reversed lactone moiety known as ZK35096, a critical impurity identified in prior art, ensuring the final API meets stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Drospirenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthesis routes is critical for the future of pharmaceutical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the TEMPO-catalyzed oxidation of drospirenone can be seamlessly transferred from the lab to the plant. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped to detect trace impurities such as the reversed lactone moiety. Our commitment to quality assurance means that every batch of drospirenone intermediate we supply meets the highest global regulatory standards, providing peace of mind to our partners in the generic and branded drug sectors.
We invite you to explore how our advanced manufacturing capabilities can optimize your supply chain. By leveraging our expertise in green chemistry and process intensification, we can help you achieve significant efficiencies in your production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our drospirenone intermediates can enhance your final product's quality and market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
