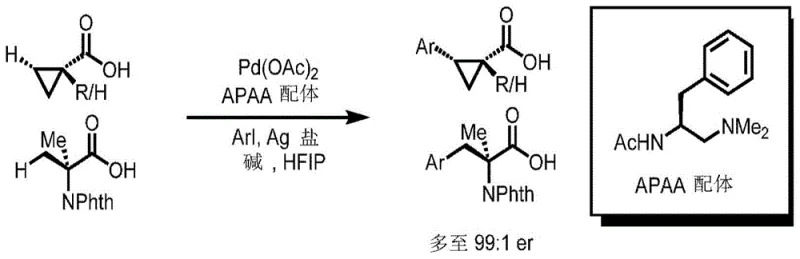
Revolutionizing Chiral Intermediate Synthesis: Direct C-H Arylation for Commercial Scale-Up
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for more efficient and sustainable manufacturing processes for complex organic molecules. Patent CN112312902B introduces a groundbreaking methodology for the stereoselective beta-arylation of carboxylic acids, specifically targeting cyclopropanecarboxylic acids and related structures without the necessity of exogenous directing groups. This innovation addresses a long-standing challenge in organic chemistry where the weak coordinating ability of free carboxyl groups typically hinders effective C-H activation. By leveraging a novel palladium(II) catalytic system combined with specially designed acetyl-protected aminoethylamine (APAA) ligands, this technology enables the direct construction of beta-chiral centers with exceptional enantioselectivity. For R&D directors and process chemists, this represents a pivotal shift away from multi-step protection-deprotection sequences towards more atom-economical routes. The ability to functionalize free carboxylic acids directly not only streamlines the synthetic pathway but also opens new avenues for the rapid diversification of chiral scaffolds essential in modern medicinal chemistry and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, achieving enantioselective C-H functionalization on aliphatic carboxylic acids has been fraught with significant synthetic inefficiencies and operational complexities. Conventional strategies often rely heavily on the installation of strong, exogenous directing groups such as amides or esters to coordinate the metal catalyst effectively. This requirement necessitates additional synthetic steps prior to the key coupling reaction, followed by rigorous conditions to remove the directing group after functionalization is complete. These extra transformations inevitably lead to increased material consumption, higher waste generation, and prolonged overall processing times, which are critical bottlenecks in commercial manufacturing. Furthermore, the harsh conditions required for installing and cleaving these directing groups can sometimes compromise the integrity of sensitive functional groups present elsewhere in the molecule, limiting the substrate scope and overall yield of the desired pharmaceutical intermediate. The cumulative effect of these limitations results in elevated production costs and a larger environmental footprint, making traditional routes less desirable for large-scale applications.
The Novel Approach
In stark contrast to legacy methodologies, the technology disclosed in Patent CN112312902B utilizes a sophisticated Pd(II)-catalyzed system that activates the inherent weak coordinating ability of the free carboxylic acid group itself. This approach eliminates the need for any pre-installation of directing groups, thereby collapsing multiple synthetic steps into a single, efficient transformation. The reaction proceeds smoothly in 1,1,1,3,3,3-hexafluoroisopropanol (HFIP) solvent, which plays a crucial role in stabilizing the catalytic intermediates and enhancing reactivity. By employing chiral APAA ligands, the system achieves precise stereocontrol, delivering products with high enantiomeric ratios often exceeding 97:3 er. This direct arylation strategy not only simplifies the workflow but also significantly improves the overall mass balance of the process. For procurement and supply chain teams, this translates to a more robust and reliable supply of complex chiral intermediates, as the reduced step count minimizes the risk of yield loss at each stage and lowers the dependency on specialized protecting group reagents.
Mechanistic Insights into Pd(II)-Catalyzed Enantioselective C-H Activation
The success of this transformative chemistry lies in the intricate interplay between the palladium catalyst and the unique structure of the APAA ligand. Mechanistically, the free carboxylic acid substrate coordinates to the Pd(II) center, facilitated by the bidentate nature of the chiral ligand which creates a rigid and well-defined coordination sphere. This specific geometry is critical for distinguishing between the enantiotopic C-H bonds at the beta-position of the cyclopropane ring. The acetyl-protected amino moiety of the ligand acts as a transient directing anchor, allowing the metal center to access the proximal C-H bond while the bulky side chains of the ligand exert steric pressure to enforce facial selectivity. This delicate balance ensures that the subsequent oxidative addition of the aryl iodide and reductive elimination steps occur with high fidelity, preserving the stereochemical integrity of the newly formed chiral center. Understanding this mechanism is vital for process optimization, as it highlights the importance of ligand purity and the precise stoichiometry of the silver salt and base used to regenerate the active catalytic species throughout the reaction cycle.
From an impurity control perspective, this catalytic system offers distinct advantages over radical-based or non-catalytic alternatives. The concerted metalation-deprotonation pathway minimizes the formation of racemic byproducts that often plague free-radical arylation methods. Additionally, the use of HFIP as a solvent helps to suppress side reactions such as homocoupling of the aryl iodide or over-arylation of the substrate. The high specificity of the APAA ligand ensures that even substrates with diverse electronic properties on the aryl iodide coupling partner are tolerated without significant erosion of enantioselectivity. For quality assurance teams, this means a cleaner crude reaction profile, which simplifies downstream purification and reduces the burden on analytical resources. The ability to consistently produce high-purity chiral acids with minimal diastereomeric contamination is a key factor in meeting the stringent regulatory requirements for pharmaceutical ingredients and advanced intermediates.
How to Synthesize Chiral Cyclopropane Derivatives Efficiently
Implementing this advanced C-H activation protocol requires careful attention to reaction conditions and reagent quality to maximize yield and selectivity. The general procedure involves charging a reaction vessel with the cyclopropanecarboxylic acid substrate, the appropriate aryl iodide, and the catalytic system comprising Pd(OAc)2 and the chiral APAA ligand in HFIP solvent. The addition of stoichiometric silver carbonate and sodium carbonate is essential to drive the reaction forward by scavenging iodide ions and maintaining the necessary basicity for C-H cleavage. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the cyclopropanecarboxylic acid substrate, aryl iodide, Pd(OAc)2 catalyst, and the chiral APAA ligand in HFIP solvent.
- Add stoichiometric amounts of Ag(I) salt and carbonate base to facilitate the C-H activation and neutralize acidic byproducts generated during the cycle.
- Heat the reaction mixture to 80°C for approximately 16 hours, then purify the resulting chiral product via preparative TLC or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this direct C-H arylation technology offers profound economic and logistical benefits for organizations managing the supply of complex chemical building blocks. By removing the requirement for directing group installation and removal, the overall material throughput is significantly improved, leading to substantial cost savings in raw material procurement. The reduction in synthetic steps directly correlates to lower labor costs and decreased consumption of solvents and reagents associated with intermediate isolations and purifications. For procurement managers, this means a more predictable cost structure and reduced exposure to price volatility of specialized protecting group reagents. Furthermore, the simplified workflow enhances operational efficiency, allowing manufacturing facilities to increase throughput without proportional increases in capital expenditure or floor space. This efficiency gain is critical for maintaining competitiveness in the fast-paced pharmaceutical and fine chemical markets where time-to-market is a decisive factor.
- Cost Reduction in Manufacturing: The elimination of protecting group manipulations removes entire sequences of chemical transformations, which drastically reduces the consumption of expensive reagents and solvents. This streamlining of the synthetic route leads to a lower cost of goods sold (COGS) by minimizing waste disposal fees and energy consumption associated with heating and cooling multiple reaction vessels. The high atom economy of the direct arylation process ensures that a greater proportion of the starting materials end up in the final product, optimizing resource utilization. Additionally, the use of commercially available aryl iodides and simple carboxylic acid precursors avoids the need for custom-synthesized starting materials, further driving down input costs and simplifying the supply chain logistics for raw material sourcing.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the number of potential failure points in the manufacturing process, thereby increasing the overall reliability of supply. With fewer steps involved, the lead time for producing batches of chiral intermediates is significantly shortened, allowing for more responsive inventory management and faster fulfillment of customer orders. The robustness of the catalytic system across a wide range of substrates means that production schedules are less likely to be disrupted by substrate-specific optimization issues. This stability is crucial for supply chain heads who need to guarantee continuous availability of critical intermediates for downstream drug substance manufacturing, mitigating the risk of production delays that could impact clinical trial timelines or commercial product launches.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are amenable to scale-up, utilizing standard equipment and moderate temperatures that do not require extreme pressure or cryogenic conditions. This facilitates a smoother transition from laboratory discovery to pilot plant and eventually to commercial-scale production. From an environmental perspective, the reduction in chemical waste and solvent usage aligns with green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability goals. The ability to produce high-value chiral intermediates with a smaller environmental footprint enhances the corporate social responsibility profile of the manufacturing operation and can provide a competitive advantage in markets where eco-friendly sourcing is prioritized by end customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd-catalyzed C-H activation technology. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing clarity on substrate compatibility and process expectations. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing development pipelines.
Q: What is the primary advantage of this C-H activation method over traditional cross-coupling?
A: The key advantage is the elimination of exogenous directing groups. Traditional methods require installing and subsequently removing directing groups, which adds synthetic steps and waste. This patent describes a direct arylation of free carboxylic acids, significantly simplifying the workflow and reducing material costs.
Q: How does the APAA ligand influence enantioselectivity in this reaction?
A: The acetyl-protected aminoethylamine (APAA) ligand coordinates with the Pd(II) center to create a chiral environment. This specific coordination geometry controls the approach of the aryl iodide to the beta-carbon of the carboxylic acid, ensuring high enantiomeric ratios (up to 99:1 er) without needing pre-functionalized substrates.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process uses commercially available reagents like Pd(OAc)2 and aryl iodides under relatively mild conditions (80°C). The absence of complex protecting group manipulations makes it highly scalable and attractive for the commercial production of high-purity chiral building blocks used in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in Patent CN112312902B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust manufacturing processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the enantiomeric excess and chemical purity of every batch. We understand that consistency is key in the supply of chiral building blocks, and our team of expert process chemists is ready to optimize this C-H arylation route to meet your specific volume and timeline requirements.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your development timeline while reducing overall production costs. Let us be your partner in turning complex chemical challenges into commercial successes.
