Optimizing Telmisartan Production: A High-Yield Synthetic Route for Global Supply Chains
The pharmaceutical landscape for Angiotensin II Receptor Blockers (ARBs) demands increasingly efficient and sustainable manufacturing processes to meet global demand while adhering to strict environmental regulations. Patent CN113307775B introduces a transformative preparation method for Telmisartan, a critical antihypertensive agent, addressing key bottlenecks in yield and safety that have plagued previous synthetic strategies. This technical insight analyzes the proprietary route which utilizes 3-methyl-4-n-butylamidobenzoic acid as a foundational building block, diverging significantly from legacy pathways that rely on hazardous reagents or expensive catalysts. By shifting the synthetic logic towards a nitrile-based intermediate generated through safe dehydration rather than direct cyanation, the process offers a robust framework for industrial scale-up. For R&D directors and supply chain leaders, understanding the mechanistic nuances of this approach is vital for evaluating potential technology transfers or procurement partnerships that ensure long-term supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
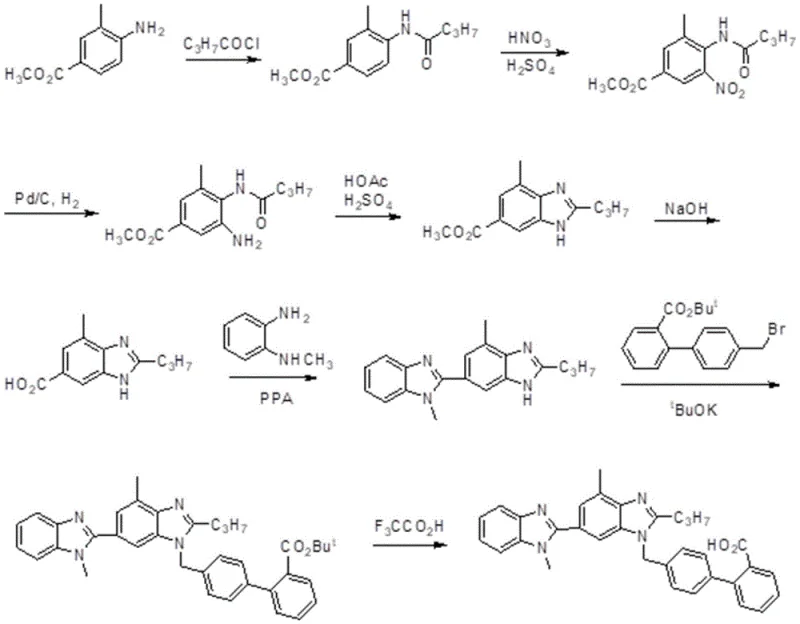
Historically, the synthesis of Telmisartan has been fraught with inefficiencies that impact both cost and environmental compliance. As illustrated in prior art such as EP 0502314, traditional routes often involve multi-step sequences that result in dismal overall yields, reported as low as 21% in some literature. These conventional pathways frequently necessitate the use of 4'-bromomethylbiphenyl-2-carbonitrile, a precursor that is not only costly but also introduces significant handling challenges due to its reactivity. Furthermore, alternative methods disclosed in patents like US200602264644 rely on palladium acetate catalysis for coupling reactions. While effective, the dependence on precious metal catalysts introduces substantial cost volatility and necessitates rigorous downstream purification to remove trace metal impurities, a critical quality attribute for API manufacturing. The accumulation of impurities in these older routes often requires complex recrystallization protocols, further eroding the final mass balance and increasing waste generation.
The Novel Approach
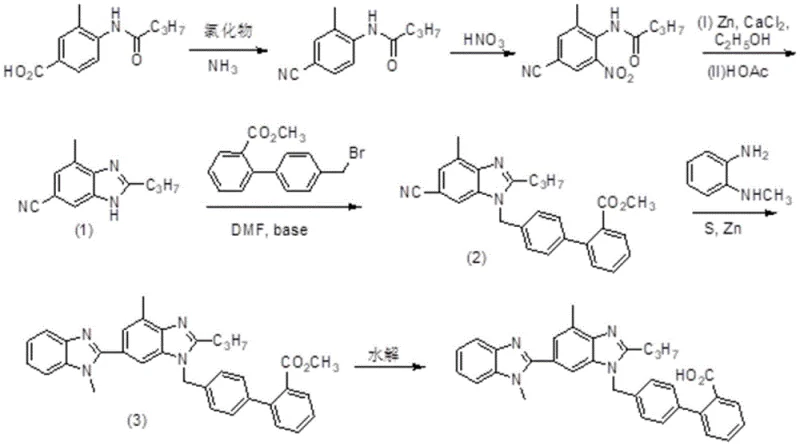
In stark contrast, the methodology outlined in CN113307775B presents a streamlined and chemically elegant solution that bypasses these historical hurdles. The core innovation lies in the strategic construction of the benzimidazole scaffold starting from 3-methyl-4-n-butylamidobenzoic acid, a commercially accessible and affordable raw material. Instead of introducing the nitrile functionality through dangerous cyanide salts, the process employs a safer dehydration strategy using thionyl chloride and ammonia gas to convert the carboxylic acid moiety into a nitrile group in situ. This fundamental shift eliminates the need for toxic cyanide reagents, drastically improving the safety profile of the manufacturing plant. The subsequent nitration and zinc-mediated reduction steps are optimized to proceed under mild conditions, preserving the integrity of the sensitive amide side chain while efficiently constructing the heterocyclic core. This approach not only boosts the overall yield to approximately 54% but also simplifies the purification workflow, making it highly attractive for large-scale production.
Mechanistic Insights into Zinc-Mediated Cyclization and Sulfur-Assisted Condensation
A deep dive into the reaction mechanism reveals why this specific pathway offers superior control over impurity profiles. The formation of the first benzimidazole ring (Intermediate 1) utilizes a combination of Zinc powder and Calcium Chlor2 in an ethanol solvent system. Mechanistically, the Zinc serves as a potent reducing agent for the nitro group, while the Calcium Chloride likely acts as a Lewis acid promoter, facilitating the intramolecular cyclization of the resulting amine with the adjacent nitrile group. This dual-action system ensures high conversion rates without the need for high-pressure hydrogenation equipment, which can be a capital expenditure barrier for some facilities. The subsequent reflux in glacial acetic acid drives the cyclization to completion, effectively locking in the benzimidazole structure with high regioselectivity. This precise control is essential for preventing the formation of isomeric byproducts that are notoriously difficult to separate in ARB synthesis.
Furthermore, the construction of the second benzimidazole ring via condensation with N-methyl o-phenylenediamine employs a sulfur-mediated protocol. In this step, elemental sulfur acts as an oxidant and dehydrogenating agent, facilitating the oxidative cyclization required to close the second heterocyclic ring. The addition of Zinc powder in the later stage of this reaction helps to scavenge excess sulfur and reduce any sulfurous byproducts, ensuring a cleaner reaction matrix. This specific stoichiometry—balancing the diamine, sulfur, and zinc—is critical for maximizing the yield of Intermediate 3. By avoiding harsh acidic or basic conditions during this delicate coupling phase, the process preserves the stereochemical and structural integrity of the biphenyl linkage, which is crucial for the biological activity of the final Telmisartan molecule. The final hydrolysis step is straightforward, utilizing standard alkaline conditions to unmask the carboxylic acid, completing the synthesis with a purity exceeding 99.7%.
How to Synthesize Telmisartan Efficiently
The operational execution of this synthesis requires careful attention to temperature control and reagent addition rates, particularly during the exothermic nitration and cyclization phases. The patent provides a detailed roadmap that balances reaction kinetics with safety, ensuring that the process is reproducible on a kilogram to tonne scale. Operators must strictly adhere to the specified molar ratios, especially regarding the Zinc and Calcium Chloride loading in the reduction step, to prevent incomplete reduction or over-reduction side reactions. The workup procedures, involving pH adjustments and solvent extractions, are designed to maximize recovery at each stage, contributing to the impressive overall yield. For a comprehensive breakdown of the exact temperatures, stirring times, and quenching protocols required to replicate this high-efficiency route, please refer to the standardized operating procedure below.
- Convert 3-methyl-4-n-butylamidobenzoic acid to 3-methyl-4-n-butylamidobenzonitrile using thionyl chloride and ammonia gas.
- Perform nitration with fuming nitric acid to introduce the nitro group at the 5-position.
- Execute reduction and cyclization using Zinc powder and Calcium Chloride in ethanol followed by acetic acid reflux to form the benzimidazole core.
- Alkylate the intermediate with 4'-bromomethyl biphenyl-2-methyl formate in DMF with a base.
- Condense with N-methyl o-phenylenediamine using sulfur and Zinc powder to form the bis-benzimidazole structure.
- Hydrolyze the ester group under alkaline conditions to yield final Telmisartan.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift away from precious metal catalysts and toxic cyanide salts translates into a significantly simplified regulatory burden and reduced operational costs. By utilizing commodity chemicals like Zinc powder and Thionyl Chloride, the process insulates the supply chain from the price volatility associated with specialized reagents. Moreover, the high overall yield means that less raw material is required to produce the same amount of API, effectively lowering the cost of goods sold (COGS). The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more reliable delivery schedule for downstream formulation partners. This reliability is paramount in the pharmaceutical sector, where supply interruptions can have severe consequences.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the use of low-cost starting materials like 3-methyl-4-n-butylamidobenzoic acid drive substantial cost savings. The process avoids the need for specialized high-pressure hydrogenation reactors, allowing production in standard glass-lined or stainless steel vessels, which reduces capital depreciation costs. Additionally, the higher yield reduces the waste disposal costs associated with failed batches or low-efficiency steps, creating a leaner manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that raw material sourcing is not a bottleneck. Unlike routes dependent on custom-synthesized brominated precursors which may have limited suppliers, the key inputs for this method are commoditized. This diversification of the supply base mitigates the risk of shortages and allows for more flexible procurement strategies. The simplified purification steps also mean faster cycle times, enabling quicker turnaround from raw material intake to finished API release.
- Scalability and Environmental Compliance: The avoidance of toxic cyanide salts significantly lowers the environmental footprint of the manufacturing process, easing the burden on wastewater treatment facilities and reducing the cost of hazardous waste disposal. The mild reaction conditions are inherently safer to scale up, reducing the risk of thermal runaways. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the manufacturing partner, a key metric for modern pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Telmisartan synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety protocols, and scalability factors. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new methodology for commercial production.
Q: How does this new route improve upon traditional Telmisartan synthesis yields?
A: Traditional routes, such as those disclosed in EP 0502314, often suffer from low overall yields around 21%. This novel method achieves a total yield of approximately 54% by optimizing the cyclization steps and utilizing more reactive intermediates.
Q: What are the safety advantages regarding cyanide usage in this process?
A: Unlike conventional methods that may rely on toxic cyanide salts for nitrile introduction, this process generates the nitrile group via dehydration of an amide using thionyl chloride and ammonia, significantly reducing environmental hazards and handling risks.
Q: Is the starting material 3-methyl-4-n-butylamidobenzoic acid readily available?
A: Yes, the process utilizes 3-methyl-4-n-butylamidobenzoic acid as a primary raw material, which is noted for being easy to obtain and cost-effective compared to expensive brominated precursors used in other pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Supplier
The technical superiority of this synthesis route positions it as a prime candidate for commercial adoption, yet successful translation from patent to plant requires experienced partnership. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary infrastructure to handle the specific reagents and conditions outlined in this process, ensuring that the theoretical yields and purity specifications are met consistently. With our rigorous QC labs and commitment to stringent purity specifications, we guarantee that every batch of Telmisartan intermediate or API meets the highest international standards, providing peace of mind for your regulatory filings.
We invite you to discuss how this optimized route can enhance your supply chain resilience and cost structure. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value for your Telmisartan projects.
