Scalable Manufacturing of Paxlovid Azabicyclo Intermediates via Advanced Halogenation Cyclization
Scalable Manufacturing of Paxlovid Azabicyclo Intermediates via Advanced Halogenation Cyclization
The global demand for antiviral therapeutics has necessitated the development of robust, scalable supply chains for critical pharmaceutical intermediates. A recent technological breakthrough disclosed in patent CN114605308A introduces a highly efficient preparation method for 6,6-dimethyl-3-azabicyclo[3.1.0]hexane, a pivotal azabicyclo medical intermediate used in the synthesis of Paxlovid. This novel approach fundamentally reimagines the synthetic pathway by utilizing a dihalo-acyl chloride cyclization strategy, departing from the hazardous and low-yielding oxidation methods traditionally employed in the industry. By leveraging easily accessible raw materials like methyl chrysanthemate and implementing a streamlined halogenation-amidation sequence, this technology offers a compelling solution for manufacturers seeking to optimize their production of high-purity pharmaceutical intermediates. The process is characterized by its operational simplicity, environmental friendliness, and exceptional suitability for industrial scale-up, addressing key pain points in the current supply chain for antiviral drug components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
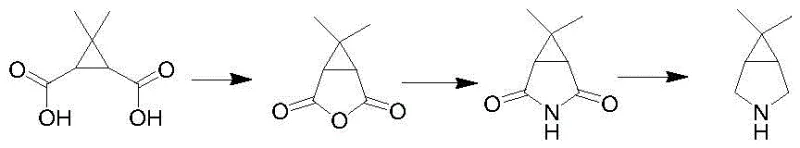
Historically, the synthesis of caronic acid derivatives and subsequent azabicyclo intermediates has been plagued by significant technical and economic hurdles that hinder efficient commercial manufacturing. Traditional routes often rely on the oxidation of ethyl chrysanthemate using large quantities of potassium permanganate, a process that is not only operationally dangerous due to the exothermic nature of the reaction but also generates substantial amounts of manganese-containing waste, creating severe environmental disposal challenges. Furthermore, these conventional pathways frequently suffer from low overall yields, particularly in the dehydration steps to form anhydrides and the subsequent reduction of bis-amide structures, where yields often struggle to exceed 70%. The reliance on expensive solvents like acetone in large volumes further inflates production costs, while the use of specialized ligands in alternative diazo-insertion methods has proven difficult to reproduce consistently, leading to batch-to-batch variability that is unacceptable for GMP manufacturing environments.
The Novel Approach
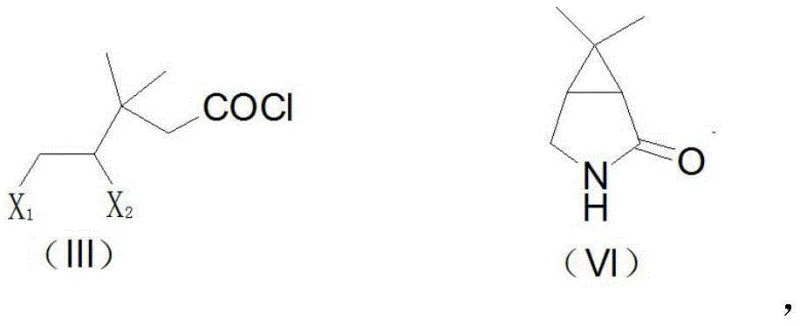
In stark contrast to these legacy processes, the methodology outlined in CN114605308A presents a paradigm shift by introducing a halogen-mediated cyclization strategy that bypasses the need for harsh oxidants and complex catalytic systems. This innovative route begins with the conversion of methyl chrysanthemate into a dihalo-acyl chloride intermediate, specifically compound (III), through a highly efficient halogenation reaction that can achieve near-quantitative yields. The subsequent transformation involves a sophisticated one-pot or multi-step cyclization and amidation reaction under the influence of strong organic bases and ammonia gas at elevated temperatures, directly forming the crucial lactam structure (VI). This approach not only eliminates the generation of heavy metal waste associated with permanganate oxidation but also drastically simplifies the purification workflow, as the intermediate compounds can often be used without extensive isolation. The result is a streamlined process that significantly enhances throughput while maintaining rigorous quality standards required for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Base-Catalyzed Cyclization and Amidation
The core of this technological advancement lies in the precise mechanistic execution of the cyclization and amidation steps, where the dihalo-acyl chloride (III) undergoes an intramolecular nucleophilic substitution to form the bicyclic lactam ring. In this critical transformation, the compound (III) is treated with an organic strong base, such as potassium tert-butoxide or sodium methoxide, in a polar organic solvent like methanol or DMF at moderate temperatures ranging from 40°C to 65°C. This initial step facilitates the formation of an enolate or activated intermediate, which is then subjected to high-temperature ammonolysis at 100°C to 300°C in the presence of ammonia gas or an ammonia-saturated organic liquid. The use of specific catalysts, such as sodium pivalate, further accelerates the ring-closing kinetics, ensuring that the reaction proceeds smoothly to generate the cyclization product (VI) with yields reaching between 50% and 62%, a marked improvement over the erratic outcomes of previous methods. This controlled thermal activation allows for the effective displacement of halogen atoms and the simultaneous formation of the amide bond, constructing the strained azabicyclo framework with high fidelity.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of bis-amide byproducts that typically complicate downstream processing in traditional routes. Because the cyclization constructs the lactam ring directly from the acyl chloride precursor, the subsequent reduction step only needs to target a single carbonyl group rather than a bis-amide structure, which traditionally requires excessive amounts of reducing agents and generates significant salt waste. The specificity of the base-catalyzed ring closure ensures that the stereochemical integrity of the gem-dimethyl group is preserved, minimizing the formation of diastereomeric impurities that could affect the biological activity of the final API. Furthermore, the ability to tune the reaction parameters, such as the molar ratio of the strong base to the substrate (optimized between 1:1.2 and 1:5), provides process chemists with robust levers to suppress side reactions, thereby delivering a crude product profile that is amenable to straightforward distillation or crystallization purification techniques.
How to Synthesize 6,6-dimethyl-3-azabicyclo[3.1.0]hexane Efficiently
The practical implementation of this synthesis route involves a logical sequence of halogenation, cyclization, and reduction steps that are designed for ease of execution in standard chemical manufacturing facilities. The process initiates with the preparation of the key dihalo-acyl chloride intermediate (III) by reacting cardia acyl chloride with halogens like chlorine or bromine at low temperatures (0-5°C), a step that proceeds with near-quantitative efficiency and requires minimal workup. Following this, the cyclization is performed either as a one-pot reaction where the base and ammonia are introduced sequentially, or via a stepwise approach involving the isolation of an ester or amide intermediate (IV) prior to ring closure. The final stage involves the reduction of the lactam (VI) to the target amine (VII) using borohydride reagents in the presence of boron trifluoride, a transformation that achieves yields exceeding 90% under mild conditions. For detailed operational parameters, including specific solvent volumes, heating rates, and quenching protocols, please refer to the standardized synthesis guide below.
- Prepare 4,5-dihalo-3,3-dimethylvaleryl chloride (III) by halogenating cardia acyl chloride with chlorine or bromine at 0-5°C.
- Perform cyclization and amidation by reacting compound (III) with an organic strong base and ammonia gas at 100-300°C to form the lactam (VI).
- Reduce the lactam (VI) using a borohydride reducing agent in the presence of boron trifluoride to obtain the final amine product (VII).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend far beyond simple yield improvements. By shifting away from oxidation-heavy processes that rely on volatile commodity prices for permanganate and acetone, manufacturers can stabilize their cost structures and reduce exposure to supply chain disruptions associated with hazardous chemical logistics. The elimination of heavy metal waste streams significantly lowers the environmental compliance burden, reducing the overhead costs related to waste treatment and regulatory reporting, which is increasingly critical in the current global regulatory landscape. Moreover, the use of common, non-proprietary reagents such as chlorine, methanol, and sodium alkoxides ensures that the supply chain remains resilient, as these materials are widely available from multiple global sources, mitigating the risk of single-source dependency that often plagues complex pharmaceutical syntheses.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic sequence, which removes the need for expensive transition metal catalysts and complex ligand systems that were previously required to achieve acceptable conversion rates. By achieving near-quantitative yields in the halogenation step and high efficiency in the reduction phase, the overall material throughput is maximized, meaning less raw material is wasted per kilogram of final product. Additionally, the ability to perform the cyclization in a one-pot manner reduces the number of unit operations, saving on energy consumption, labor hours, and equipment occupancy time, all of which contribute to a substantially lower cost of goods sold without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on methyl chrysanthemate as a starting material provides a robust foundation for supply continuity, as this feedstock is produced on a massive scale for the agrochemical and fragrance industries, ensuring consistent availability even during market fluctuations. The process conditions, which operate within standard temperature and pressure ranges for most of the sequence, allow for flexible manufacturing across different geographic locations without requiring specialized high-pressure or cryogenic infrastructure. This flexibility enables companies to diversify their manufacturing footprint, reducing lead times for high-purity pharmaceutical intermediates and ensuring that production schedules can be met reliably even in the face of regional logistical challenges.
- Scalability and Environmental Compliance: From a scale-up perspective, the chemistry is inherently safe and manageable, avoiding the runaway reaction risks associated with large-scale permanganate oxidations. The waste profile is significantly cleaner, consisting primarily of inorganic salts and recoverable organic solvents, which simplifies the effluent treatment process and aligns with modern green chemistry principles. This environmental compatibility not only facilitates faster regulatory approvals for new manufacturing sites but also future-proofs the supply chain against tightening environmental regulations, making it a sustainable choice for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology, providing clarity on its operational feasibility and strategic value. These insights are derived directly from the experimental data and process descriptions within the patent literature, offering a realistic view of what partners can expect when integrating this route into their production portfolios. Understanding these nuances is essential for R&D and procurement teams evaluating the potential for technology transfer or contract manufacturing partnerships.
Q: What are the primary advantages of this new synthesis route over traditional methods?
A: The new route avoids the use of dangerous potassium permanganate oxidants and expensive transition metal catalysts found in prior art. It utilizes readily available methyl chrysanthemate and achieves near-quantitative yields in the halogenation step, significantly simplifying waste treatment and lowering raw material costs.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The process employs a specific cyclization mechanism using strong organic bases like potassium tert-butoxide, which minimizes byproduct formation. Furthermore, the final reduction step achieves yields over 90%, compared to less than 70% in traditional succinimide reduction routes, resulting in a cleaner impurity profile suitable for API manufacturing.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial amplification. It features simple operations such as one-pot cyclization and uses common solvents like methanol and ethanol. The avoidance of ozone-depleting chlorinating agents and the use of standard pressure reactors for the ammonia reaction make it highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-dimethyl-3-azabicyclo[3.1.0]hexane Supplier
As the pharmaceutical industry continues to evolve, the ability to rapidly scale innovative synthetic routes from the laboratory to commercial production is a defining capability for any leading CDMO partner. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane meets the exacting standards required for antiviral drug synthesis. We understand the critical nature of supply continuity in the pharma sector and have optimized our operations to deliver consistent quality and reliability, making us a trusted ally for companies navigating the complexities of global API supply chains.
We invite you to engage with our technical procurement team to discuss how this novel cyclization technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers compared to your current supply sources. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your manufacturing operations.
