Scaling High-Performance Dehydroabietic Acid Triarylamine D-π-A Compounds for Next-Gen Solar Cells
Scaling High-Performance Dehydroabietic Acid Triarylamine D-π-A Compounds for Next-Gen Solar Cells
The rapid evolution of renewable energy technologies has placed immense pressure on the supply chain for advanced optoelectronic materials. Patent CN110642830B introduces a groundbreaking class of dehydroabietic acid triarylamine D-π-A type compounds that utilize thiophene derivatives as π-bridges. This innovation addresses critical limitations in current dye-sensitized solar cell (DSSC) architectures by significantly red-shifting absorption and emission spectra. For R&D directors and procurement leaders in the electronic chemicals sector, this patent represents a viable pathway to higher efficiency photovoltaic materials. The structural design integrates a robust dehydroabietic acid triarylamine electron donor with a thiophene π-bridge and a cyanoacetic acid electron acceptor, creating a molecule optimized for charge transfer and stability.
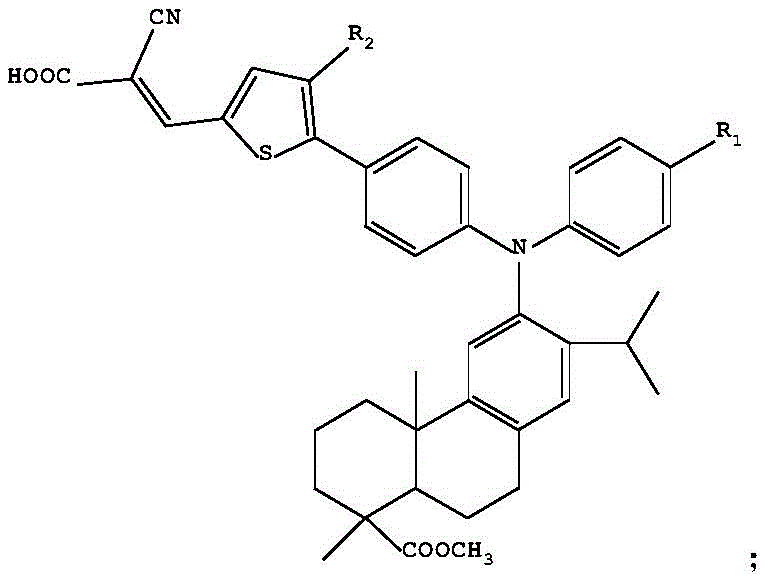
As a reliable electronic chemical supplier, understanding the nuances of this synthesis is vital for ensuring supply continuity. The patent details a robust three-step synthetic route that avoids exotic reagents, relying instead on well-established cross-coupling and condensation chemistries. This approach not only enhances the photoelectric conversion efficiency potential but also simplifies the manufacturing process, making it an attractive candidate for cost reduction in dye-sensitized solar cell material manufacturing. The ability to tune the R1 and R2 substituents allows for further optimization of solubility and energy levels, providing a versatile platform for custom material development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional dehydroabietic acid triarylamine compounds, often designed with simple D-A structures, have historically struggled to meet the rigorous spectral requirements of modern photovoltaic applications. As noted in the background art of the patent, previous iterations exhibited ultraviolet absorption wavelengths primarily below 400nm. This limitation severely restricts their utility in DSSCs, where harvesting visible light is paramount for achieving high photoelectric conversion efficiencies. Furthermore, the lack of an extended conjugated system in these conventional D-A structures often results in insufficient charge separation and lower fluorescence quantum yields. For supply chain heads, relying on these older generation materials means procuring components that may soon become obsolete as industry standards shift towards broader spectrum absorption.
The Novel Approach
The novel approach detailed in CN110642830B overcomes these spectral deficits by inserting a thiophene derivative as a π-bridge between the donor and acceptor units. This structural modification creates a D-π-A architecture that facilitates intramolecular charge transfer more effectively than its D-A predecessors. The result is a dramatic red-shift in the absorption spectrum, with new peaks appearing in the 380-600nm range and a maximum absorption wavelength reaching 474nm. Similarly, the fluorescence emission is pushed to 603nm, a significant improvement over the 374nm observed in non-bridged analogues. This strategic insertion of the thiophene unit not only enhances optical properties but also maintains the steric benefits of the triarylamine group, preventing detrimental molecular aggregation on the photoelectrode surface.
Mechanistic Insights into Suzuki-Miyaura Coupling and Knoevenagel Condensation
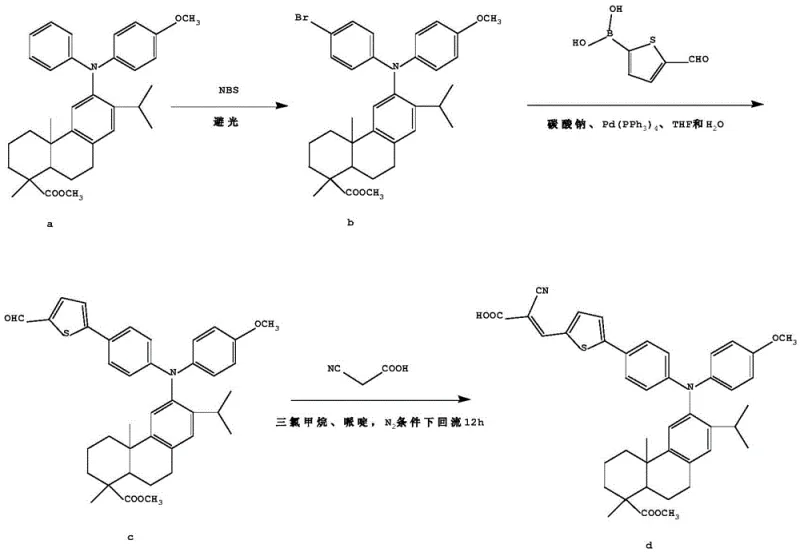
The core of this synthesis relies on two pivotal reactions: the palladium-catalyzed Suzuki-Miyaura cross-coupling and the Knoevenagel condensation. The Suzuki coupling step is particularly critical for constructing the carbon-carbon bond between the brominated triarylamine intermediate and the thiophene boronic acid derivative. In this mechanism, the Pd(0) catalyst undergoes oxidative addition with the aryl bromide, followed by transmetallation with the organoboron species in the presence of a base like sodium carbonate. This step is performed in a biphasic system of THF and water, which aids in the solubility of the inorganic base while maintaining the organic reactants in solution. The choice of ligand, such as triphenylphosphine in Pd(PPh3)4, stabilizes the palladium center and ensures high turnover numbers, which is essential for minimizing residual metal impurities in the final electronic grade material.
Following the formation of the aldehyde intermediate, the Knoevenagel condensation installs the electron-withdrawing cyanoacetic acid group. This reaction proceeds via the deprotonation of the active methylene group in cyanoacetic acid by a weak base like piperidine, generating a nucleophilic carbanion. This species attacks the carbonyl carbon of the thiophene aldehyde, followed by dehydration to form the conjugated double bond. This final step completes the D-π-A system, extending the conjugation length and locking in the favorable electronic properties. The use of chloroform as a solvent and mild reflux conditions ensures that the sensitive dehydroabietic acid skeleton remains intact while driving the equilibrium towards the product through water removal.
How to Synthesize Dehydroabietic Acid Triarylamine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with consistent quality. The process begins with the controlled bromination of the starting triarylamine using N-bromosuccinimide (NBS) in acetonitrile, requiring strict protection from light to prevent side reactions. Subsequent purification via column chromatography ensures the removal of polybrominated impurities before the material enters the coupling stage. The detailed experimental conditions, including specific molar ratios such as 1:1 for NBS and 2:1 for the boronic acid coupling partner, are optimized to maximize yield while minimizing waste. For manufacturers looking to implement this technology, adhering to these precise stoichiometric balances is key to achieving the reported yields of approximately 65-70% per step.
- Bromination of dehydroabietic acid triarylamine compound (I) with NBS in acetonitrile under dark conditions to yield compound (II).
- Palladium-catalyzed Suzuki coupling of compound (II) with thiophene boronic acid derivatives in a THF/water mixture to form the aldehyde intermediate (III).
- Knoevenagel condensation of intermediate (III) with cyanoacetic acid using piperidine as a catalyst in chloroform to obtain the final D-π-A product (IV).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in CN110642830B offers substantial advantages in terms of raw material availability and process scalability. The reliance on commodity chemicals such as NBS, cyanoacetic acid, and standard palladium catalysts means that supply chain disruptions are less likely compared to processes requiring bespoke or highly specialized reagents. Furthermore, the reaction conditions—mostly ambient pressure and moderate temperatures—reduce the energy burden on manufacturing facilities. This translates to a more sustainable production profile, aligning with the increasing demand for green chemistry practices in the electronic materials sector. The ability to source precursors from multiple vendors enhances supply security and provides leverage in price negotiations.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences found in alternative dye syntheses significantly lowers the overall cost of goods sold. By utilizing a convergent synthesis strategy where the thiophene bridge is coupled in a single step, the process reduces labor hours and solvent consumption. Additionally, the high selectivity of the Suzuki coupling minimizes the formation of difficult-to-remove byproducts, thereby reducing the load on downstream purification units. This efficiency gain allows for a more competitive pricing structure without compromising the purity specifications required for high-performance solar applications.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch reproducibility, a critical factor for long-term supply agreements. The use of stable intermediates that can be isolated and stored, such as the brominated compound (II) and the aldehyde (III), provides flexibility in production scheduling. This modularity allows manufacturers to stockpile key intermediates during periods of low demand, ensuring rapid fulfillment when market needs surge. Such operational agility is invaluable for maintaining continuity in the fast-paced renewable energy supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable from gram to kilogram and eventually to tonne quantities without fundamental changes to the reaction engineering. The solvent systems employed, primarily THF, water, and chloroform, are well-understood in industrial settings with established recovery and recycling protocols. This facilitates compliance with environmental regulations regarding volatile organic compound (VOC) emissions. Moreover, the absence of heavy metals other than the catalytic amount of palladium, which can be scavenged, simplifies waste treatment and disposal procedures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dehydroabietic acid derivatives. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding material selection and process integration.
Q: What is the primary advantage of introducing a thiophene bridge in dehydroabietic acid triarylamine compounds?
A: Introducing a thiophene derivative as a π-bridge significantly extends the conjugation system, shifting the UV absorption maximum to 474nm and fluorescence emission to 603nm, which is critical for efficient dye-sensitized solar cells.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes standard organic reactions like Suzuki coupling and Knoevenagel condensation with commercially available reagents such as NBS and cyanoacetic acid, making it highly amenable to commercial scale-up.
Q: What are the typical reaction conditions for the key coupling step?
A: The Suzuki coupling step typically employs Pd(PPh3)4 as a catalyst with sodium carbonate as a base in a THF and water solvent system, heated to reflux for approximately 12 hours under nitrogen.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroabietic Acid Triarylamine Supplier
The transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands ready to support your development of next-generation solar materials with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the specific temperature and atmosphere controls required for palladium-catalyzed couplings and sensitive condensations. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of dehydroabietic acid triarylamine derivative meets the exacting standards of the optoelectronic industry.
We invite you to collaborate with us to optimize this synthesis for your specific application needs. Our technical team can provide a Customized Cost-Saving Analysis to identify further efficiencies in your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of these critical D-π-A compounds for your dye-sensitized solar cell projects.
