Advanced Synthesis of Dehydroabietic Acid Triarylamine D-π-A Compounds for Optoelectronic Applications
Advanced Synthesis of Dehydroabietic Acid Triarylamine D-π-A Compounds for Optoelectronic Applications
The rapid evolution of the renewable energy sector has placed immense pressure on the development of next-generation photovoltaic materials, specifically within the realm of dye-sensitized solar cells (DSSC). Patent CN110668975A introduces a groundbreaking class of D-π-A type compounds that utilize dehydroabietic acid triarylamine as the electron-donating group (D), benzene derivatives as the π-bridge, and cyanoacetic acid as the electron-withdrawing group (A). This technological leap addresses the critical limitation of traditional triarylamine dyes, which often suffer from insufficient absorption ranges in the visible spectrum. By strategically extending the conjugated system through a robust benzene-based π-bridge, this invention achieves a substantial red-shift in both ultraviolet absorption and fluorescence emission, positioning these materials as high-value candidates for advanced electronic chemical manufacturing.
For research and development directors overseeing material innovation, the structural integrity and purity of these intermediates are paramount. The patent outlines a precise synthetic pathway that ensures the formation of the desired D-π-A architecture with high fidelity. The process begins with the functionalization of the dehydroabietic acid core, followed by a palladium-catalyzed cross-coupling to install the π-bridge, and concludes with a condensation reaction to attach the acceptor group. This modular approach not only allows for fine-tuning of the electronic properties by varying substituents on the benzene ring but also ensures that the final products meet the stringent purity specifications required for high-efficiency optoelectronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic sensitizers for DSSCs has been hindered by the narrow absorption profiles of simple donor-acceptor (D-A) systems. Traditional triarylamine-based dyes, while effective hole transport materials in OLEDs, often exhibit ultraviolet absorption wavelengths confined within the 400nm range. This spectral limitation severely restricts their ability to harvest solar energy efficiently, as a significant portion of the solar spectrum remains unutilized. Furthermore, the lack of a rigid and extended π-conjugated bridge in these conventional structures often leads to weaker intramolecular charge transfer (ICT), resulting in lower photoelectric conversion efficiencies. The inability to independently modulate the donor, bridge, and acceptor components without compromising stability has been a persistent bottleneck in the commercialization of cost-effective solar dyes.
The Novel Approach
The methodology described in CN110668975A overcomes these hurdles by introducing a sophisticated D-π-A architecture that decouples the electronic functions of the molecule. By inserting a benzene derivative as a π-bridge between the bulky dehydroabietic acid triarylamine donor and the cyanoacetic acid acceptor, the conjugation length is significantly extended. This structural modification facilitates a more efficient push-pull electron mechanism, evidenced by the emergence of new absorption peaks in the 362-562nm range. The maximum absorption wavelength shifts to 428nm, and critically, the fluorescence emission undergoes a massive red-shift of 220nm, reaching 594nm. This demonstrates a clear advantage in light-harvesting capability, making these compounds superior candidates for reliable electronic chemical supplier portfolios focused on renewable energy applications.
Mechanistic Insights into Suzuki Coupling and Knoevenagel Condensation
The core of this synthesis relies on two pivotal catalytic transformations: the Suzuki-Miyaura cross-coupling and the Knoevenagel condensation. The Suzuki coupling step is particularly critical for constructing the carbon-carbon bond between the brominated dehydroabietic acid triarylamine intermediate and the benzene boronic acid derivative. In this mechanism, a palladium(0) species, generated in situ from precursors like Pd(PPh3)4 or Pd(dppf)Cl2·CH2Cl2, undergoes oxidative addition with the aryl bromide. This is followed by transmetallation with the organoboron species in the presence of a base such as sodium carbonate or potassium acetate. The use of mixed solvent systems, typically involving THF or toluene with water, ensures optimal solubility for both organic substrates and inorganic bases, driving the reaction to completion while minimizing side reactions.
Following the successful construction of the aldehyde-functionalized intermediate, the final step involves a Knoevenagel condensation with cyanoacetic acid. This reaction creates the vital electron-withdrawing anchor group that facilitates electron injection into the semiconductor substrate (typically TiO2) in a DSSC device. The reaction proceeds via the formation of an enolate intermediate from cyanoacetic acid, catalyzed by a weak base like piperidine. The subsequent nucleophilic attack on the aldehyde carbonyl, followed by dehydration, yields the conjugated alkene linkage. This specific linkage is essential for maintaining planarity and ensuring effective orbital overlap across the D-π-A system, which is crucial for the observed red-shift in optical properties and overall device performance.
How to Synthesize Dehydroabietic Acid Triarylamine Derivatives Efficiently
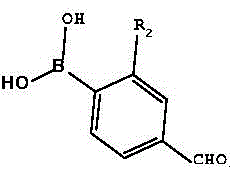
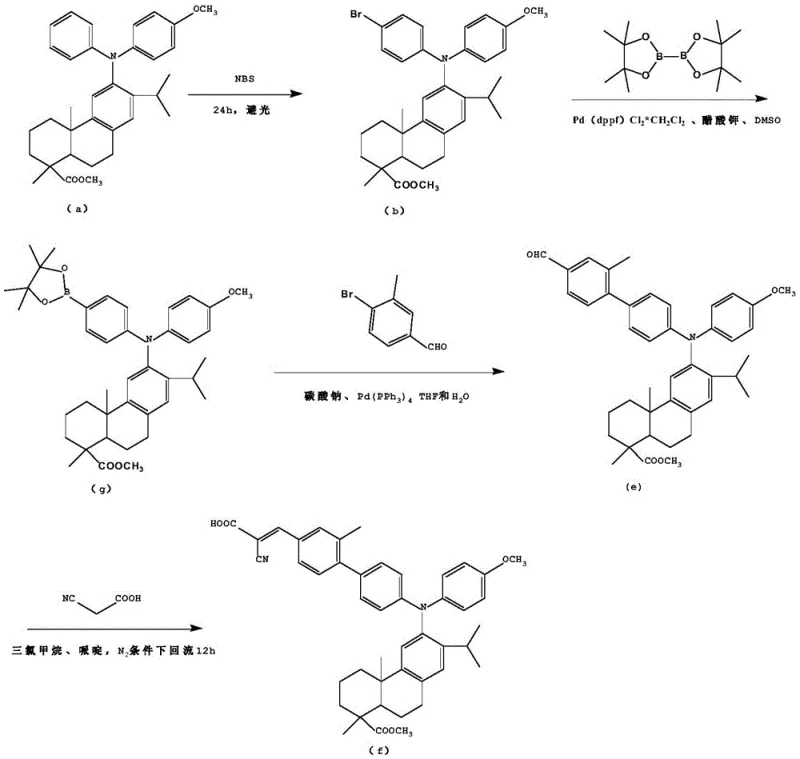
The synthesis of these high-performance D-π-A compounds is designed to be operationally straightforward while maintaining high chemical selectivity. The process leverages widely available starting materials and standard laboratory equipment, making it highly adaptable for scale-up. The initial bromination step requires careful control of light exposure to prevent radical side reactions, ensuring the selective formation of the mono-brominated species. Subsequent coupling reactions utilize robust palladium catalysts that tolerate various functional groups, allowing for the introduction of diverse substituents (R1 and R2) to fine-tune the electronic characteristics of the final dye. For a detailed breakdown of the specific reaction conditions, stoichiometry, and purification protocols, please refer to the standardized guide below.
- Bromination of the dehydroabietic acid triarylamine precursor using NBS in acetonitrile under dark conditions to form the brominated intermediate.
- Palladium-catalyzed Suzuki cross-coupling reaction between the brominated intermediate and a benzene derivative boronic acid to construct the π-bridge aldehyde.
- Knoevenagel condensation of the resulting aldehyde intermediate with cyanoacetic acid in the presence of piperidine to yield the final D-π-A compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers significant strategic benefits. The reliance on dehydroabietic acid, a naturally occurring resin acid derived from pine trees, provides a sustainable and potentially cost-stable feedstock compared to purely petrochemical-derived alternatives. This bio-based origin aligns with growing corporate sustainability goals and may offer long-term price stability against volatile oil markets. Furthermore, the synthetic steps utilize commodity chemicals such as N-bromosuccinimide (NBS), cyanoacetic acid, and common palladium catalysts, which are readily available from multiple global suppliers, reducing the risk of single-source dependency.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for exotic or highly specialized reagents, relying instead on well-established organic transformations like Suzuki coupling and Knoevenagel condensation. These reactions are known for their scalability and high atom economy when optimized. By avoiding complex multi-step protection and deprotection sequences often found in other dye syntheses, the overall process mass intensity (PMI) is reduced. This streamlined approach translates directly to lower operational expenditures (OPEX) in terms of solvent usage, waste disposal, and labor hours, facilitating substantial cost savings in display material manufacturing.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible production scheduling. Intermediates such as the brominated triarylamine and the aldehyde-functionalized bridge can potentially be stockpiled, enabling a just-in-time manufacturing model for the final condensation step. Additionally, the use of standard purification techniques like column chromatography and recrystallization, as described in the patent examples, ensures that quality control can be easily implemented at various stages. This reduces the likelihood of batch failures and ensures a consistent supply of high-purity electronic chemical intermediates to downstream device manufacturers.
- Scalability and Environmental Compliance: The reaction conditions specified, such as temperatures ranging from 25°C to 80°C and the use of solvents like ethanol, toluene, and THF, are compatible with existing large-scale reactor infrastructure. There is no requirement for extreme high-pressure or cryogenic conditions, which simplifies engineering controls and safety protocols. Moreover, the aqueous workups described in the patent facilitate the separation of inorganic salts and catalyst residues, aiding in waste stream management. This ease of handling supports the commercial scale-up of complex polymer additives and specialty chemicals while adhering to increasingly strict environmental regulations regarding heavy metal discharge and solvent emissions.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new class of materials is essential for stakeholders evaluating their integration into existing product lines. The following questions address common inquiries regarding the stability, application scope, and synthesis specifics of these dehydroabietic acid-based dyes. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the spectral advantages of these new D-π-A compounds compared to traditional triarylamines?
A: According to patent CN110668975A, the new compounds exhibit a significant red-shift in both absorption and emission. Specifically, the maximum fluorescence emission wavelength shifts from 374nm in the parent compound to 594nm in the new D-π-A structure, making them far more suitable for dye-sensitized solar cells.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the process utilizes standard organic transformations such as NBS bromination, Suzuki coupling, and Knoevenagel condensation. These reactions use commercially available catalysts like Pd(PPh3)4 and common solvents, facilitating scale-up from laboratory to commercial tonnage.
Q: What is the role of the benzene derivative in this molecular architecture?
A: The benzene derivative serves as the π-conjugated bridge connecting the electron-donating dehydroabietic acid triarylamine group and the electron-withdrawing cyanoacetic acid group. This extension of the conjugated system is critical for lowering the energy gap and shifting absorption into the visible region.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroabietic Acid Triarylamine Supplier
As the demand for high-efficiency organic photovoltaics continues to surge, securing a supply chain partner with deep technical expertise is critical. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed couplings and sensitive condensation reactions, ensuring that every batch meets stringent purity specifications. With rigorous QC labs dedicated to analyzing impurity profiles and optical properties, we guarantee that our dehydroabietic acid triarylamine derivatives deliver the performance metrics required for next-generation DSSC applications.
We invite industry leaders to collaborate with us to optimize the production of these advanced optoelectronic materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your transition to these high-performance D-π-A compounds is seamless, cost-effective, and scientifically sound.
