Advanced Synthesis of Dehydroabietic Acid Triarylamine D-pi-A Compounds for High-Efficiency Solar Cells
Advanced Synthesis of Dehydroabietic Acid Triarylamine D-pi-A Compounds for High-Efficiency Solar Cells
The rapid evolution of renewable energy technologies has placed dye-sensitized solar cells (DSSCs) at the forefront of photovoltaic research, driving an urgent demand for novel organic sensitizers with superior light-harvesting capabilities. Patent CN110668975B introduces a groundbreaking class of D-pi-A type compounds that utilize dehydroabietic acid triarylamine as a robust electron-donating group (D), benzene derivatives as conjugated pi-bridges, and cyanoacetic acid as a potent electron-withdrawing group (A). This structural architecture is specifically engineered to overcome the limitations of earlier generations of dyes, which often suffered from narrow absorption bands and insufficient spectral overlap with the solar spectrum. By integrating a rigid dehydroabietic acid scaffold with a flexible triarylamine core, the invention achieves a unique balance of steric stability and electronic delocalization. The resulting materials demonstrate a remarkable red-shift in optical properties, with maximum absorption wavelengths extending to 428nm and fluorescence emission peaking at 594nm. For procurement specialists and R&D directors seeking reliable electronic chemical suppliers, this technology represents a significant leap forward in the design of next-generation optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of efficient sensitizers for DSSCs has been hindered by the inherent trade-offs between molecular stability and charge transfer efficiency. Traditional triarylamine-based dyes, while possessing good hole-transport properties, often exhibit absorption spectra confined to the ultraviolet region, typically below 400nm, which severely limits their ability to harvest visible light. Furthermore, many conventional synthetic routes rely on harsh reaction conditions or expensive transition metal catalysts that are difficult to remove, leading to impurity profiles that degrade device performance over time. The lack of effective pi-conjugation bridges in earlier designs resulted in poor electronic communication between the donor and acceptor moieties, causing rapid charge recombination and low photocurrent generation. Additionally, the planar nature of many standard dyes promotes molecular aggregation on the semiconductor surface, leading to energy loss through self-quenching. These structural and processing deficiencies have created a bottleneck in cost reduction in electronic chemical manufacturing, as complex purification steps and low yields drive up the final price of the active materials.
The Novel Approach
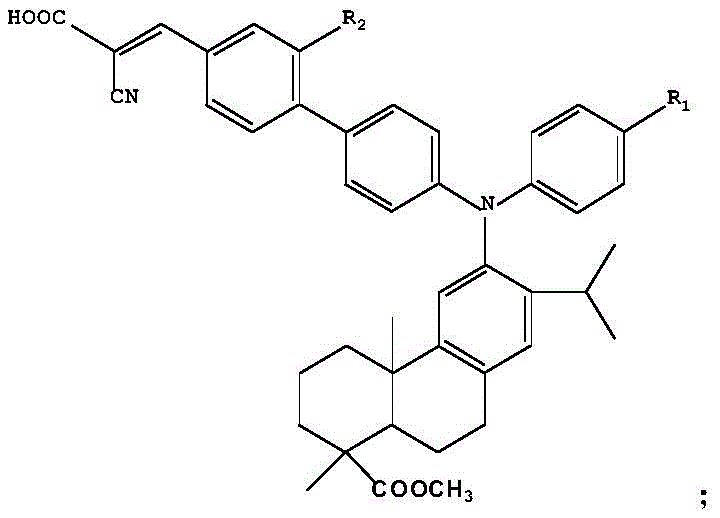
The methodology disclosed in the patent offers a transformative solution by employing a modular synthetic strategy that precisely constructs the D-pi-A architecture through well-defined catalytic cycles. The core innovation lies in the strategic functionalization of the dehydroabietic acid triarylamine backbone, first through selective bromination and subsequently via palladium-catalyzed cross-coupling reactions to install the pi-bridge. This approach allows for the systematic extension of the conjugated system without compromising the thermal stability provided by the bulky dehydroabietic acid group. As illustrated in the reaction scheme below, the process utilizes accessible starting materials and proceeds through high-yielding intermediates, ensuring a consistent supply of high-purity intermediates. The final condensation step with cyanoacetic acid efficiently installs the anchoring carboxyl group, facilitating strong adsorption onto titanium dioxide surfaces. This novel route not only enhances the photophysical properties, achieving a 220nm red-shift in fluorescence compared to the precursor, but also simplifies the overall production workflow, making it an attractive option for commercial scale-up of complex organic semiconductors.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling and Condensation
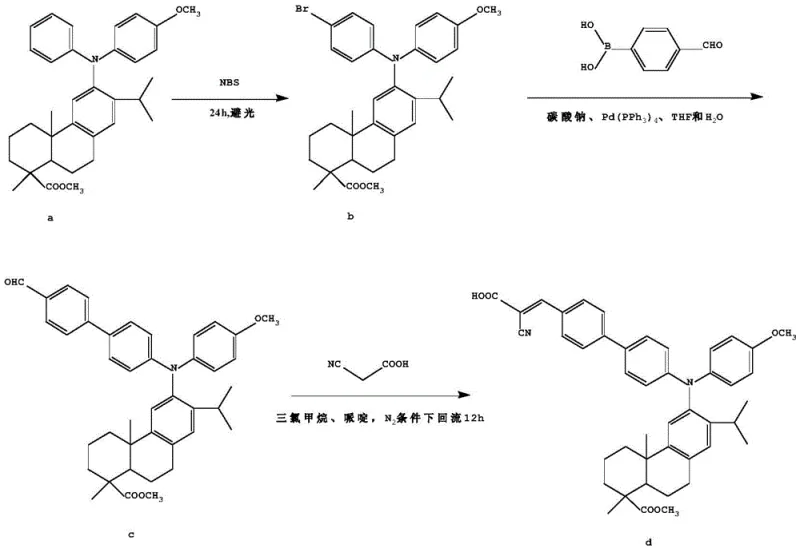
The heart of this synthesis lies in the sophisticated application of transition metal catalysis to forge carbon-carbon bonds with high regioselectivity. The initial bromination step utilizes N-bromosuccinimide (NBS) to selectively activate the para-position of the phenyl ring attached to the nitrogen atom, creating a reactive handle for subsequent coupling. This is followed by a Suzuki-Miyaura coupling reaction, where the brominated intermediate reacts with arylboronic acids in the presence of a palladium catalyst such as Pd(PPh3)4 or Pd(dppf)Cl2. The mechanism involves the oxidative addition of the palladium species into the carbon-bromine bond, followed by transmetallation with the boronate species activated by the inorganic base, and finally reductive elimination to form the extended biaryl pi-system. This step is critical for establishing the electronic pathway between the donor and the future acceptor. The versatility of this mechanism is further highlighted by the alternative route shown below, which utilizes a pinacol boronate ester intermediate, demonstrating the robustness of the catalytic cycle under varying solvent conditions like DMSO or THF. Such mechanistic flexibility ensures that the process can be optimized for different scales and substrate variations.
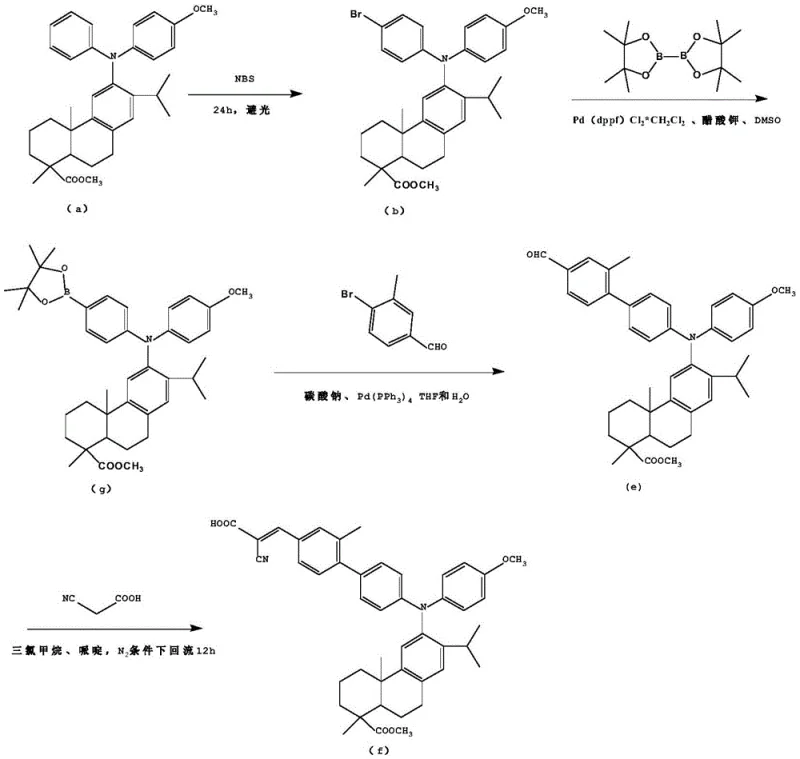
Following the construction of the pi-bridge, the final transformation involves a Knoevenagel condensation between the aldehyde-functionalized intermediate and cyanoacetic acid. This reaction is typically catalyzed by a weak base like piperidine in a solvent such as chloroform or acetonitrile. The mechanism proceeds through the deprotonation of the active methylene group in cyanoacetic acid, generating a nucleophilic carbanion that attacks the carbonyl carbon of the aldehyde. Subsequent dehydration yields the alpha,beta-unsaturated nitrile-acid structure, which serves as the electron-accepting anchor. This specific structural motif is crucial for injecting electrons into the conduction band of the semiconductor upon photoexcitation. The careful control of reaction temperature, typically around 60-80°C, and the use of inert atmospheres prevent side reactions such as polymerization or oxidation of the sensitive triarylamine core. Understanding these mechanistic nuances is vital for R&D teams aiming to replicate the high purity and consistent batch-to-batch quality required for high-performance optoelectronic devices.
How to Synthesize Dehydroabietic Acid Triarylamine D-pi-A Compound Efficiently
The synthesis of these advanced dye intermediates requires precise control over reaction parameters to maximize yield and minimize impurities. The process begins with the dissolution of the dehydroabietic acid triarylamine precursor in anhydrous acetonitrile, followed by the slow addition of NBS under strictly dark conditions to prevent radical side reactions. After the bromination is complete, the crude product is purified via column chromatography before proceeding to the coupling stage. In the second stage, the brominated intermediate is combined with the appropriate boronic acid derivative, a palladium catalyst, and an inorganic base in a biphasic solvent system of THF and water. The mixture is heated under reflux to drive the coupling to completion. Finally, the resulting aldehyde is condensed with cyanoacetic acid in the presence of piperidine. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Perform bromination of the dehydroabietic acid triarylamine precursor using N-bromosuccinimide (NBS) in acetonitrile under dark conditions to yield the brominated intermediate.
- Execute a Suzuki-Miyaura cross-coupling reaction between the brominated intermediate and a benzene boronic acid derivative using a palladium catalyst and inorganic base in a mixed solvent system.
- Conduct a Knoevenagel condensation reaction between the resulting aldehyde intermediate and cyanoacetic acid in the presence of piperidine to form the final D-pi-A structured compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthesis route offers substantial strategic benefits, primarily driven by the use of commodity-grade reagents and robust reaction conditions. The reliance on widely available starting materials such as dehydroabietic acid derivatives and common benzene building blocks significantly mitigates the risk of raw material shortages that often plague the specialty chemical sector. Furthermore, the reaction conditions are relatively mild, operating at temperatures between 45°C and 80°C, which reduces energy consumption and lowers the operational burden on manufacturing facilities. The use of standard palladium catalysts, while requiring recovery protocols, is well-established in the industry, meaning that existing infrastructure can often be adapted without massive capital expenditure. This compatibility with existing manufacturing assets translates directly into reduced lead time for high-purity organic semiconductors, allowing companies to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The streamlined three-step sequence eliminates the need for exotic reagents or multi-protection group strategies that typically inflate production costs. By utilizing direct C-H activation or simple halogenation followed by coupling, the atom economy of the process is improved, leading to less waste generation. The high selectivity of the palladium-catalyzed steps minimizes the formation of difficult-to-remove isomers, thereby reducing the load on downstream purification units like preparative HPLC. Consequently, the overall cost of goods sold (COGS) is optimized, making these high-performance dyes more commercially viable for mass-market solar applications.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of intermediate production from final assembly. Key intermediates, such as the brominated dehydroabietic acid derivative, can be stockpiled or sourced from multiple qualified vendors, creating a buffer against supply disruptions. The robustness of the reaction to minor variations in solvent quality or temperature further enhances process reliability, ensuring consistent output even in diverse manufacturing environments. This resilience is critical for maintaining continuous production schedules for downstream clients in the photovoltaic and display industries.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on gram scales in the patent examples, with clear pathways to kilogram and tonne-level production. The use of aqueous workups and standard organic solvents facilitates efficient waste management and solvent recovery systems. Moreover, the final products exhibit excellent photostability, which extends the lifecycle of the end-devices, indirectly contributing to sustainability goals by reducing the frequency of replacement. The elimination of heavy metal contaminants through standard purification protocols ensures compliance with stringent environmental regulations governing electronic waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dehydroabietic acid-based dyes. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation, providing a reliable basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating these materials into their existing product lines or research pipelines.
Q: What is the primary optical advantage of this new D-pi-A compound structure?
A: The compound exhibits a significant red-shift in both absorption and fluorescence emission compared to traditional dehydroabietic acid triarylamine derivatives. Specifically, the maximum absorption wavelength reaches 428nm and fluorescence emission peaks at 594nm, a 220nm red-shift from the precursor, making it highly suitable for broad-spectrum light harvesting in DSSCs.
Q: Which catalysts are recommended for the C-C coupling step in this synthesis?
A: The patent specifies several effective palladium catalysts including tetrakis(triphenylphosphine)palladium(0) [Pd(PPh3)4], 1,1'-bis(diphenylphosphino)ferrocene-palladium(II)dichloride [Pd(dppf)Cl2], and palladium acetate. These are used in conjunction with inorganic bases like sodium carbonate or potassium acetate.
Q: Can this synthesis method be adapted for different substituents on the phenyl ring?
A: Yes, the method is highly versatile. The R1 and R2 groups on the benzene derivatives can be varied to include hydrogen, methyl, methoxy, hexyl, or other electron-donating and withdrawing groups, allowing for fine-tuning of the HOMO-LUMO energy levels for specific electronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroabietic Acid Triarylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the advancement of renewable energy technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of dehydroabietic acid triarylamine derivatives meets the exacting standards required for DSSC and OLED applications. Our commitment to quality assurance means that you can rely on us for materials that deliver reproducible performance in your final devices.
We invite you to contact our technical procurement team to discuss your specific requirements. We offer a Customized Cost-Saving Analysis to help you optimize your sourcing strategy and reduce overall project expenses. Whether you need specific COA data for regulatory filings or detailed route feasibility assessments for new product development, our experts are ready to provide the support you need to accelerate your time to market.
