Advanced Synthesis of Dehydroabietic Acid Triarylamine D-pi-A Compounds for High-Performance OLEDs
Advanced Synthesis of Dehydroabietic Acid Triarylamine D-pi-A Compounds for High-Performance OLEDs
The rapid evolution of the optoelectronic industry demands materials with precisely tuned electronic properties, particularly for applications in dye-sensitized solar cells (DSSC) and organic light-emitting diodes (OLED). Patent CN111138389A introduces a groundbreaking class of dehydroabietic acid triarylamine D-pi-A type compounds that utilize furan derivatives as pi-bridges to overcome historical limitations in absorption spectra. This innovation represents a significant leap forward for manufacturers seeking a reliable OLED material supplier capable of delivering next-generation intermediates. By strategically integrating an electron-donating dehydroabietic acid triarylamine group with an electron-withdrawing cyanoacetic acid group via a conjugated furan bridge, the resulting molecules exhibit a maximum absorption wavelength of 477nm and a fluorescence emission maximum of 595nm. 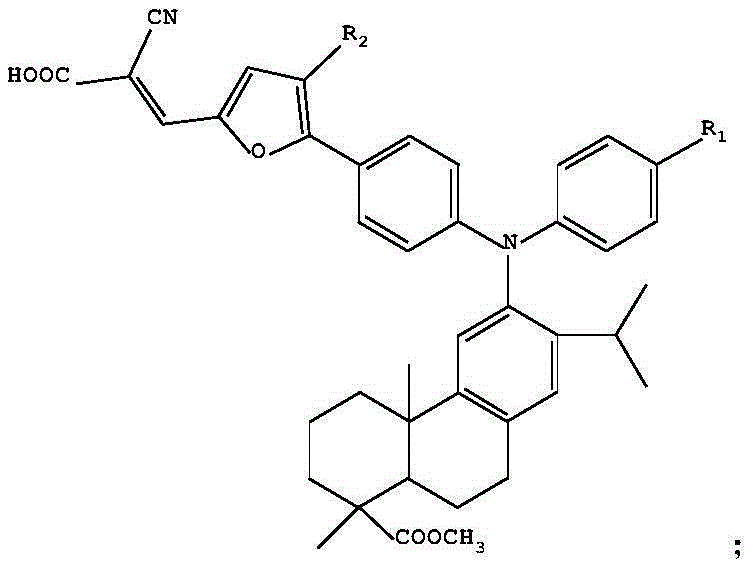 This structural modification not only enhances charge transfer capabilities but also mitigates the energy loss typically caused by molecular stacking, thereby offering a robust solution for high-efficiency photovoltaic and display technologies.
This structural modification not only enhances charge transfer capabilities but also mitigates the energy loss typically caused by molecular stacking, thereby offering a robust solution for high-efficiency photovoltaic and display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of dye sensitizers relied heavily on simpler D-A (Donor-Acceptor) structural motifs which, while functional, often failed to meet the stringent spectral requirements of modern high-efficiency devices. Traditional dehydroabietic acid triarylamine compounds exhibited ultraviolet absorption spectra that were critically short, typically confined within the 400nm range, rendering them inadequate for maximizing photon capture in solar cell applications. Furthermore, the planar nature of many conventional acceptors often led to undesirable aggregation on photoelectric electrode interfaces, causing significant energy loss through non-radiative decay pathways. These structural deficiencies necessitated complex and costly post-synthesis modifications or the use of expensive co-adsorbents to prevent stacking, thereby inflating the overall cost reduction in electronic chemical manufacturing. The inability to independently tune the donor, bridge, and acceptor units in older architectures limited the fine-tuning of HOMO-LUMO energy levels, restricting the versatility of these materials across different device configurations.
The Novel Approach
The synthesis method disclosed in the patent revolutionizes this landscape by employing a modular D-pi-A architecture that allows for independent optimization of each functional unit. The core innovation lies in the utilization of a furan derivative as the pi-conjugated bridge, which effectively extends the conjugation length and facilitates superior intramolecular charge transfer. As illustrated in the comprehensive reaction scheme, the process begins with the bromination of the triarylamine precursor, followed by a palladium-catalyzed cross-coupling with a furan boronic acid, and concludes with a Knoevenagel condensation. 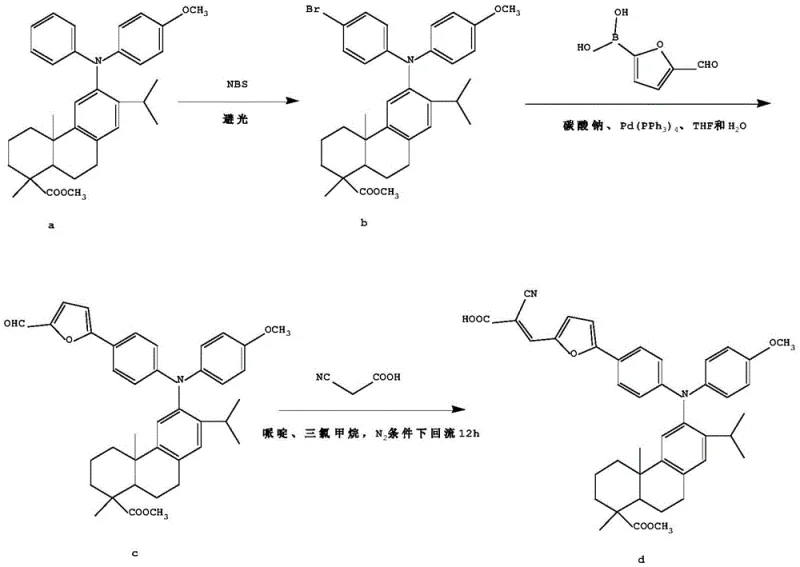 This stepwise approach ensures high regioselectivity and purity, crucial for a reliable agrochemical intermediate supplier or electronic material provider. The resulting compounds demonstrate a dramatic red-shift in both absorption and emission spectra compared to their predecessors, with the maximum fluorescence emission wavelength shifting from 374nm to 595nm. This substantial improvement validates the efficacy of the furan bridge in enhancing the photoelectric performance, making these compounds ideal candidates for commercial scale-up of complex polymer additives and optoelectronic materials.
This stepwise approach ensures high regioselectivity and purity, crucial for a reliable agrochemical intermediate supplier or electronic material provider. The resulting compounds demonstrate a dramatic red-shift in both absorption and emission spectra compared to their predecessors, with the maximum fluorescence emission wavelength shifting from 374nm to 595nm. This substantial improvement validates the efficacy of the furan bridge in enhancing the photoelectric performance, making these compounds ideal candidates for commercial scale-up of complex polymer additives and optoelectronic materials.
Mechanistic Insights into Suzuki-Miyaura Coupling and Knoevenagel Condensation
The heart of this synthetic strategy relies on two pivotal reactions: the Suzuki-Miyaura cross-coupling and the Knoevenagel condensation, both of which are executed with high precision to ensure product integrity. The Suzuki coupling step utilizes a palladium catalyst, such as tetrakis(triphenylphosphine)palladium, in conjunction with an inorganic carbonate base to facilitate the formation of the carbon-carbon bond between the brominated triarylamine and the furan boronic acid. This mechanism proceeds through a catalytic cycle involving oxidative addition, transmetallation, and reductive elimination, ensuring that the sensitive furan ring remains intact while forming the critical linkage. The use of a mixed solvent system comprising organic solvents like THF or toluene and water enhances the solubility of the inorganic base, thereby accelerating the reaction kinetics without compromising the stability of the intermediates. 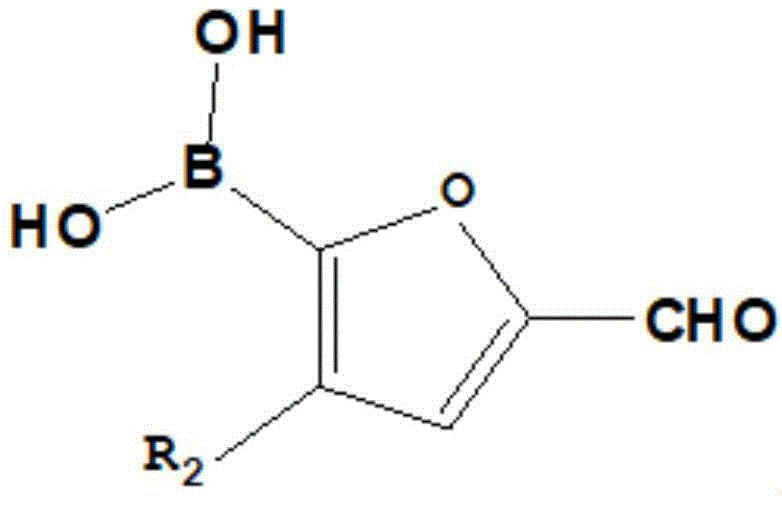 Following the coupling, the resulting aldehyde intermediate undergoes Knoevenagel condensation with cyanoacetic acid in the presence of piperidine. This reaction creates the final electron-withdrawing anchor group, completing the D-pi-A system and establishing the push-pull electronic character essential for efficient charge separation.
Following the coupling, the resulting aldehyde intermediate undergoes Knoevenagel condensation with cyanoacetic acid in the presence of piperidine. This reaction creates the final electron-withdrawing anchor group, completing the D-pi-A system and establishing the push-pull electronic character essential for efficient charge separation.
Controlling impurities in this multi-step synthesis is paramount, particularly given the sensitivity of the conjugated system to side reactions. The patent specifies rigorous purification protocols, including column chromatography with specific eluent ratios (e.g., petroleum ether:ethyl acetate), to remove unreacted starting materials and palladium residues. The choice of mild reaction conditions, such as maintaining temperatures between 50-200°C and utilizing nitrogen atmospheres, minimizes the formation of oxidation byproducts and polymerization of the furan moiety. Furthermore, the stoichiometric control of reagents, such as using a 10:1 molar ratio of cyanoacetic acid to the aldehyde intermediate, drives the condensation equilibrium towards completion, ensuring high conversion rates. This meticulous attention to reaction parameters guarantees the production of high-purity OLED material with consistent batch-to-batch reproducibility, a key requirement for industrial adoption.
How to Synthesize Dehydroabietic Acid Triarylamine D-pi-A Compounds Efficiently
The synthesis of these advanced materials follows a logical three-step progression designed for scalability and ease of execution in a standard chemical laboratory or pilot plant. The process begins with the activation of the dehydroabietic acid triarylamine core via bromination, followed by the construction of the pi-bridge through palladium catalysis, and finishes with the installation of the acceptor group. Each step has been optimized to balance yield and purity, utilizing readily available reagents and standard workup procedures. For detailed operational parameters, safety considerations, and specific stoichiometric calculations required for GMP-compliant production, please refer to the standardized protocol below.
- Bromination of the dehydroabietic acid triarylamine precursor using NBS in acetonitrile under dark conditions to form the bromo-intermediate.
- Suzuki-Miyaura cross-coupling reaction between the bromo-intermediate and furan boronic acid derivatives using a Palladium catalyst and carbonate base.
- Knoevenagel condensation of the resulting aldehyde intermediate with cyanoacetic acid in chloroform using piperidine as a catalyst to finalize the D-pi-A structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical performance. The methodology eliminates the need for exotic or highly unstable reagents, relying instead on commodity chemicals like N-bromosuccinimide, cyanoacetic acid, and standard palladium catalysts. This reliance on established supply chains significantly reduces the risk of raw material shortages and price volatility, ensuring a steady flow of production inputs. Moreover, the reaction conditions are relatively mild, avoiding the need for extreme cryogenic temperatures or ultra-high pressures, which translates to lower energy consumption and reduced wear on reactor equipment. These factors collectively contribute to a more resilient and cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The streamlined three-step sequence minimizes the number of unit operations required, directly lowering labor and overhead costs associated with prolonged processing times. By avoiding the use of expensive transition metals other than standard palladium catalysts, which can be recovered and recycled, the process eliminates the need for costly heavy metal scavenging steps often required in pharmaceutical synthesis. The high selectivity of the Suzuki coupling reduces the formation of difficult-to-separate isomers, thereby simplifying downstream purification and increasing the overall yield of the desired product. Consequently, the total cost of goods sold (COGS) is significantly optimized, providing a competitive edge in the pricing of high-value electronic intermediates.
- Enhanced Supply Chain Reliability: The starting materials, including dehydroabietic acid derivatives and furan boronic acids, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demands without extensive re-validation. Additionally, the stability of the intermediates permits safe storage and transport, facilitating a decentralized production model if necessary. This flexibility ensures reducing lead time for high-purity electronic chemical deliveries, keeping downstream device fabrication lines running smoothly without interruption.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, having been demonstrated effectively from gram-scale laboratory experiments to potential multi-kilogram production runs. The use of common organic solvents like THF, toluene, and chloroform allows for straightforward solvent recovery and recycling systems, aligning with modern green chemistry principles and environmental regulations. The absence of highly toxic reagents or hazardous byproducts simplifies waste treatment protocols, lowering the environmental compliance burden on the facility. This eco-friendly profile not only meets the stringent sustainability criteria of major multinational corporations but also future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its integration into their existing portfolios. The following questions address common inquiries regarding the scalability, purity, and application scope of these dehydroabietic acid triarylamine derivatives. The answers provided are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and reliability for decision-making purposes.
Q: What is the primary advantage of introducing a furan derivative as a pi-bridge in these compounds?
A: The introduction of a furan derivative as a pi-bridge significantly extends the conjugation system, resulting in a red-shift of the ultraviolet absorption spectrum (new peak at 381-591nm) and fluorescence emission (max 595nm), which is critical for improving photoelectric conversion efficiency in DSSCs and OLED applications.
Q: Which catalysts are suitable for the coupling reaction in this synthesis?
A: The patent specifies several effective palladium catalysts including tetrakis(triphenylphosphine)palladium, bis(triphenylphosphine)palladium dichloride, palladium acetate, and 1,1'-bisdiphenylphosphinoferrocene palladium dichloride, allowing flexibility in catalyst selection based on availability and cost.
Q: How does this synthesis method address the limitations of previous dehydroabietic acid triarylamine compounds?
A: Previous compounds suffered from short ultraviolet absorption wavelengths (within 400nm), limiting their utility in solar cells. This novel D-pi-A architecture effectively shifts absorption towards longer wavelengths, meeting the rigorous spectral requirements for advanced optoelectronic devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroabietic Acid Triarylamine Supplier
As the demand for high-performance optoelectronic materials continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of dehydroabietic acid triarylamine intermediate meets the exacting standards required for OLED and DSSC applications. We understand the critical nature of material consistency in device performance and are committed to delivering products that drive your technological advancements.
We invite you to collaborate with our technical team to explore how this novel synthetic route can be adapted to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this furan-bridged architecture. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project timelines. Contact our technical procurement team today to initiate a dialogue on securing a sustainable and high-quality supply of these next-generation electronic materials.
