Advanced Synthesis of Ursolic Acid Ester Derivatives for Oncology Drug Development
Advanced Synthesis of Ursolic Acid Ester Derivatives for Oncology Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds to combat resistant malignancies, and natural product derivatives remain a rich source of inspiration. Patent CN101830961B discloses a significant advancement in the field of triterpenoid chemistry by presenting a series of ursolic acid ester derivatives, specifically ursolic acid 4-[1-acetyl-3-substituted phenyl-4,5-dihydro-2-pyrazolin-5-yl]phenol esters and ursolic acid 4'-substituent-4-chalcone esters. These compounds are engineered to enhance the inherent antitumor properties of ursolic acid, a pentacyclic triterpenoid widely distributed in nature. The structural modifications introduced in this patent aim to improve bioavailability and target specificity, addressing the limitations of the parent compound. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such synthetic pathways is crucial for delivering high-quality materials to R&D teams focused on next-generation anticancer therapies.
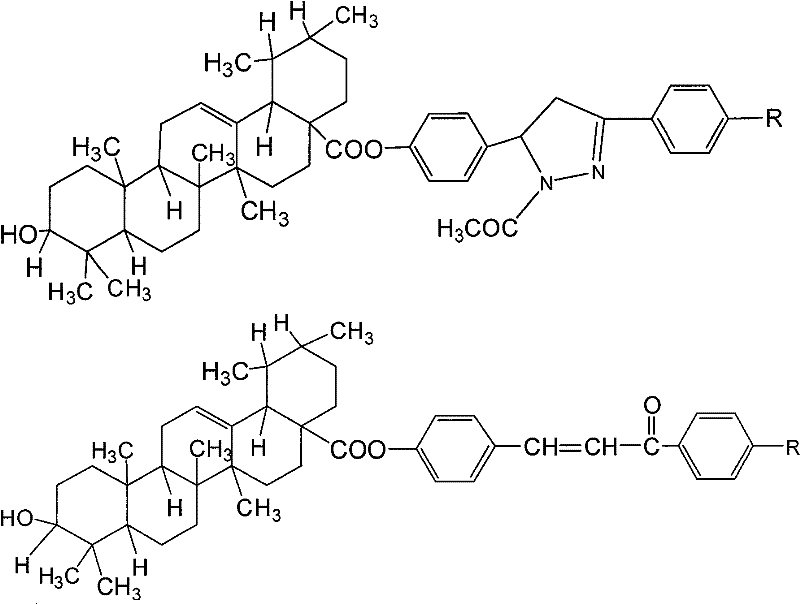
The core innovation lies in the strategic esterification at the C-28 carboxylic acid position of the ursolic acid skeleton with functionalized phenolic moieties. This approach not only preserves the bioactive triterpene core but also introduces diverse aromatic systems known for their own biological activities, such as chalcones and pyrazolines. The synergy between the triterpene backbone and the aromatic side chains creates a hybrid molecule with potentially superior pharmacokinetic profiles. For procurement managers and supply chain heads, the feasibility of synthesizing these complex molecules relies heavily on the robustness of the chemical transformations involved, which we will analyze in depth below.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for modifying ursolic acid often involve harsh conditions that can compromise the integrity of the sensitive triterpene skeleton or result in poor regioselectivity. Direct esterification using strong acid catalysts can lead to dehydration or rearrangement of the double bond at the C-12 position, generating unwanted impurities that are difficult to remove. Furthermore, many conventional routes rely on expensive coupling reagents or protecting group strategies that add unnecessary steps, increasing both the cost of goods sold (COGS) and the environmental footprint. The lack of modularity in older synthesis designs means that creating a library of analogs for structure-activity relationship (SAR) studies is time-consuming and resource-intensive, slowing down the drug discovery pipeline.
The Novel Approach
The methodology outlined in CN101830961B offers a streamlined and modular alternative that overcomes these historical bottlenecks. By decoupling the synthesis of the aromatic side chain from the final coupling step, the process allows for the rapid generation of diverse derivatives. The use of a mixed anhydride activation strategy for the ursolic acid carboxylic group ensures mild reaction conditions that preserve the stereochemistry of the pentacyclic ring system. This novel approach significantly reduces the formation of side products, thereby simplifying downstream purification. For a reliable pharmaceutical intermediate supplier, this translates to higher yields and consistent quality, which are critical metrics for maintaining supply chain reliability and meeting the stringent purity specifications required by global regulatory bodies.
Mechanistic Insights into the Multi-Step Synthesis Strategy
The synthetic route is elegantly divided into three distinct phases, each optimized for efficiency and scalability. The first phase involves the Claisen-Schmidt condensation between p-hydroxybenzaldehyde and various substituted acetophenones. This reaction is conducted in ethanol with sodium hydroxide at controlled low temperatures of 0°C to 5°C. Maintaining this thermal window is critical to prevent polymerization of the aldehyde and ensure the formation of the trans-chalcone geometry, which is thermodynamically more stable. The resulting 4'-substituted-4-hydroxychalcones serve as versatile building blocks. In the second phase, for the pyrazoline series, these chalcones undergo a 1,4-Michael addition with hydrazine hydrate in glacial acetic acid. This step involves a cascade of cyclization and intramolecular dehydration to form the dihydropyrazole ring, a heterocyclic motif frequently associated with anti-inflammatory and anticancer activities.
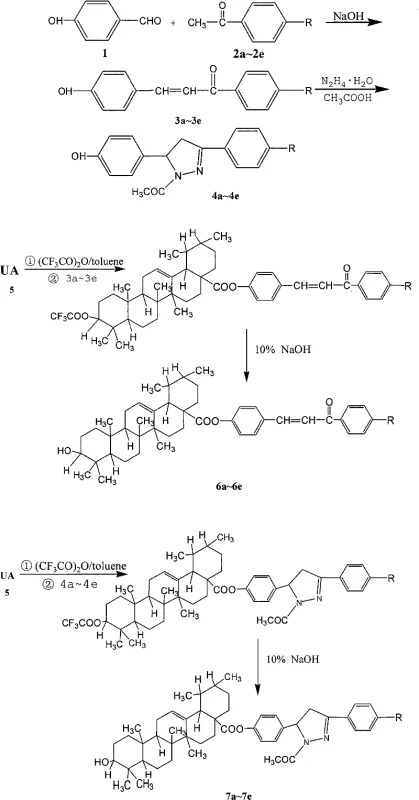
The final esterification step represents the convergence of the triterpene and aromatic domains. Ursolic acid is activated in situ using trifluoroacetic anhydride (TFAA) in toluene to form a reactive mixed anhydride intermediate. This activation method is superior to using thionyl chloride or oxalyl chloride as it generates fewer corrosive byproducts and operates under milder conditions. The phenolic intermediates (either the chalcone or the pyrazoline derivative) then attack this activated species to form the ester bond. A subsequent hydrolysis step with 10% sodium hydroxide solution removes any trifluoroacetyl protecting groups that may have formed on the phenolic hydroxyls during the coupling, ensuring the final product matches the target structure. This mechanistic precision ensures high purity, a key requirement for cost reduction in API manufacturing where impurity profiles dictate the viability of a drug candidate.
How to Synthesize Ursolic Acid Ester Derivatives Efficiently
Executing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the initial condensation and the final coupling stages. The patent provides detailed embodiments that serve as a robust foundation for process development. The following guide summarizes the critical operational parameters derived from the intellectual property, offering a roadmap for technical teams aiming to replicate or scale this chemistry. For detailed standardized synthesis steps, please refer to the guide below.
- Perform Claisen-Schmidt condensation between p-hydroxybenzaldehyde and substituted acetophenones using NaOH in ethanol at 0°C to 5°C to form 4'-substituted-4-hydroxychalcones.
- Execute 1,4-Michael addition and cyclization of the chalcone intermediates with hydrazine hydrate in glacial acetic acid under reflux to generate 4-(1-acetyl-3-substituted phenyl-4,5-dihydro-2-pyrazolin-5-yl)phenols.
- Activate ursolic acid with trifluoroacetic anhydride in toluene, followed by coupling with the phenolic intermediates and subsequent hydrolysis with 10% NaOH to yield the final ester derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in CN101830961B presents compelling advantages for procurement and supply chain optimization. The reliance on commodity chemicals such as acetophenones, benzaldehydes, and hydrazine hydrate ensures that raw material sourcing is straightforward and less susceptible to market volatility compared to specialized reagents. The use of common solvents like ethanol, toluene, and acetic acid further simplifies logistics, as these are widely available in bulk quantities globally. This accessibility directly contributes to substantial cost savings in the supply chain, reducing the risk of production delays caused by material shortages. Additionally, the moderate reaction temperatures and atmospheric pressure conditions eliminate the need for specialized high-pressure reactors or cryogenic equipment, lowering capital expenditure requirements for manufacturing facilities.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts or complex protecting group manipulations that are often required in similar triterpenoid modifications. By utilizing a direct activation strategy with TFAA and simple base-mediated condensations, the number of unit operations is minimized. This streamlining reduces labor costs, energy consumption, and waste disposal fees. The high yields reported in the patent embodiments, ranging significantly across different substituents, indicate a material-efficient process that maximizes the output per batch, driving down the unit cost of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch reproducibility, which is vital for maintaining a steady supply of intermediates for clinical trials or commercial production. The simplicity of the workup procedures, involving standard extraction and recrystallization techniques, reduces the likelihood of processing errors that could lead to batch failures. This reliability allows supply chain managers to forecast inventory needs with greater accuracy and reduces the need for excessive safety stock, thereby optimizing working capital.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram or ton-scale production without fundamental changes to the reaction engineering. The solvents used are recoverable and recyclable, aligning with green chemistry principles and reducing the environmental impact of the manufacturing process. This compliance with environmental standards is increasingly important for pharmaceutical companies aiming to meet sustainability goals and regulatory requirements regarding solvent emissions and waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these ursolic acid derivatives. The answers are derived from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating these intermediates into your drug development pipeline.
Q: What are the key advantages of this synthetic route for ursolic acid derivatives?
A: The process utilizes mild reaction conditions, such as low-temperature condensation (0°C-5°C) and standard reflux, avoiding extreme pressures or temperatures. It employs readily available commodity chemicals like acetophenones and hydrazine hydrate, significantly simplifying raw material procurement and reducing overall production costs compared to complex multi-step organometallic routes.
Q: How does the biological activity of these derivatives compare to native ursolic acid?
A: According to patent data, specific derivatives such as the 4'-chloro and 4'-fluoro substituted esters demonstrate enhanced inhibitory activity against A549 lung cancer and SKOV-3 ovarian cancer cell lines compared to unmodified ursolic acid. This structural modification improves the pharmacological profile, making them promising candidates for further oncology drug development.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the methodology is designed for scalability. The reactions use common solvents like ethanol, toluene, and acetic acid, and the workup procedures involve standard techniques like recrystallization and column chromatography. The absence of sensitive catalysts or cryogenic requirements facilitates easier scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursolic Acid Derivatives Supplier
The synthesis of complex triterpenoid esters requires a partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the preclinical or commercial phase. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of ursolic acid derivatives meets the highest industry standards, minimizing the risk of downstream processing issues for our clients.
We invite you to collaborate with us to leverage this innovative technology for your oncology drug programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and bring life-saving therapies to market faster.
