Advanced Synthesis of Ursolic Acid Ester Derivatives for Enhanced Antitumor Drug Development
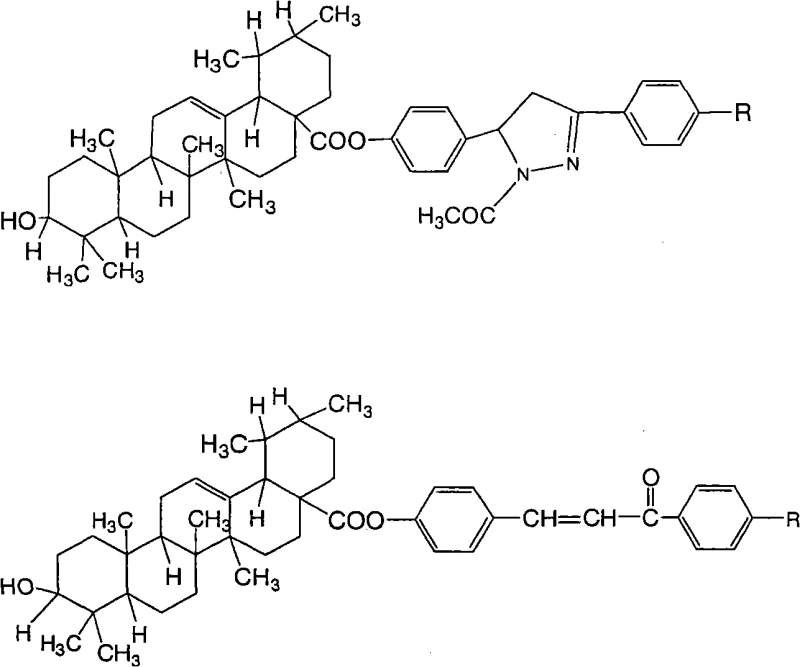
The pharmaceutical industry continuously seeks novel scaffolds to overcome drug resistance and improve therapeutic indices in oncology. Patent CN101830961A introduces a significant advancement in this domain by disclosing a series of Ursolic Acid (UA) ester derivatives, specifically Ursolic Acid 1-acetyl-3-substituted phenyl-4,5-dihydro-5-phenol esters and Ursolic Acid 4'-substituent-4-chalcone esters. These compounds represent a strategic structural modification of the naturally occurring pentacyclic triterpenoid, Ursolic Acid, which is known for its broad biological activities but often limited by poor water solubility and moderate potency. The patent outlines a robust synthetic methodology that not only enhances the antitumor profile of the parent molecule but also offers a scalable route for industrial production. By attaching diverse aromatic side chains through an ester linkage at the C28 position, the inventors have created a library of high-purity pharmaceutical intermediates with tunable pharmacological properties. This technical breakthrough provides a reliable foundation for developing next-generation anticancer agents, addressing the critical need for more effective treatments in the global healthcare market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying natural triterpenoids like Ursolic Acid often suffer from significant drawbacks that hinder their commercial viability and clinical application. Conventional esterification methods frequently employ strong acid catalysts or high-temperature conditions that can lead to the degradation of the sensitive olefinic bond at the C12-C13 position or cause skeletal rearrangements, resulting in complex impurity profiles that are difficult to separate. Furthermore, direct coupling reactions often exhibit low regioselectivity, potentially reacting with the C3 hydroxyl group instead of the desired C28 carboxylic acid, necessitating extensive protection and deprotection steps that drastically reduce overall yield and increase production costs. Additionally, many existing derivatives fail to demonstrate a substantial improvement in cytotoxicity compared to the native compound, limiting their value as lead candidates for drug development. The lack of a modular synthesis strategy in older methods also restricts the ability to rapidly explore structure-activity relationships (SAR) by introducing diverse functional groups efficiently.
The Novel Approach
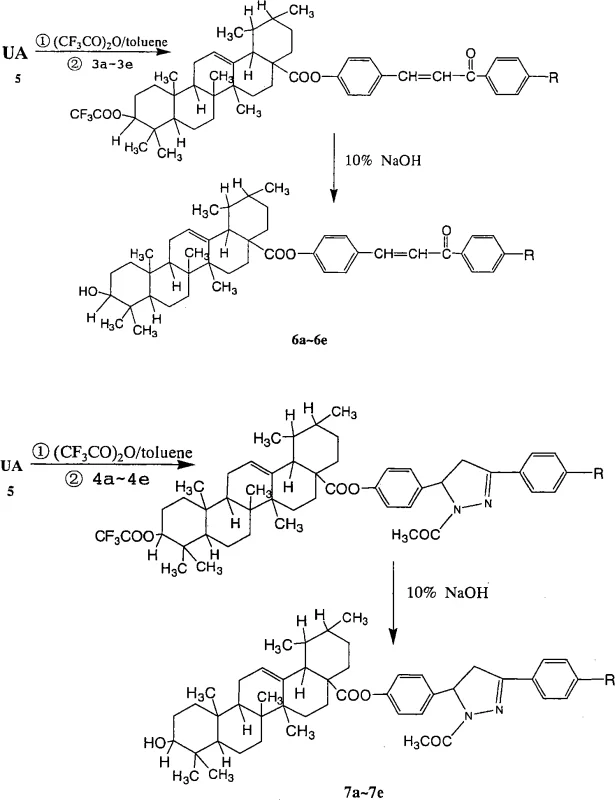
The methodology described in CN101830961A overcomes these historical challenges through a cleverly designed modular synthesis strategy that separates the construction of the side chain from the final coupling step. This approach utilizes a mild activation protocol involving trifluoroacetic anhydride (TFAA) in toluene, which selectively activates the C28 carboxylic acid of Ursolic Acid under controlled conditions, preserving the integrity of the triterpenoid core. By pre-synthesizing the phenolic side chains—either chalcones or pyrazolines—with high purity prior to the esterification step, the process ensures that the final coupling reaction proceeds with high efficiency and minimal byproduct formation. This decoupling of synthetic steps allows for precise control over the substitution pattern on the aromatic ring (R = H, Me, OMe, Cl, F), enabling the rapid generation of a focused library of derivatives. The result is a streamlined process that delivers high-purity intermediates suitable for rigorous biological evaluation, significantly reducing the time and resources required for lead optimization in antitumor drug discovery programs.
Mechanistic Insights into TFAA-Mediated Esterification and Side-Chain Construction
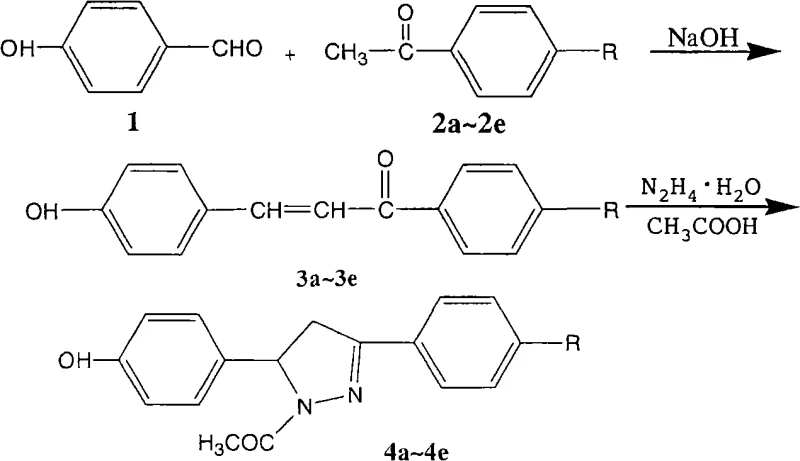
The core of this synthetic innovation lies in the two distinct pathways for constructing the side chains and their subsequent conjugation to the Ursolic Acid backbone. For the chalcone series (Compounds 6a-6e), the process initiates with a Claisen-Schmidt condensation between p-hydroxybenzaldehyde and various substituted acetophenones. This reaction is catalyzed by sodium hydroxide in ethanol at low temperatures (0°C to 5°C), promoting the formation of the α,β-unsaturated ketone moiety with high stereoselectivity for the trans-isomer. For the pyrazoline series (Compounds 7a-7e), these chalcone intermediates undergo a subsequent 1,4-Michael addition with hydrazine hydrate in glacial acetic acid. This cyclization step forms the dihydropyrazole ring, introducing a nitrogen heterocycle that can enhance binding interactions with biological targets. The final esterification step is mechanistically driven by the formation of a mixed anhydride intermediate between Ursolic Acid and TFAA. This highly reactive species is then attacked by the phenolic hydroxyl group of the side chain, forming the stable ester bond. This mechanism avoids the use of harsh coupling reagents like DCC/DMAP which can leave difficult-to-remove urea byproducts, thereby simplifying downstream purification.
How to Synthesize Ursolic Acid Ester Derivatives Efficiently
The synthesis of these high-value intermediates requires strict adherence to the reaction parameters outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of the phenolic precursors, where temperature control during the base-catalyzed condensation is critical to prevent polymerization or side reactions. Following the isolation of the side chains, the esterification with Ursolic Acid must be performed under anhydrous conditions to prevent the hydrolysis of the activated anhydride intermediate. The reaction mixture is typically heated to reflux in toluene to drive the equilibrium towards product formation. Post-reaction workup involves a careful pH adjustment using dilute sodium hydroxide to remove excess acid and byproducts, followed by extraction and column chromatography. Detailed standardized synthetic steps for reproducing these results in a GMP-compliant environment are provided in the guide below.
- Synthesize 4'-substituted-4-hydroxychalcone intermediates via Claisen-Schmidt condensation of p-hydroxybenzaldehyde and substituted acetophenones in ethanol with NaOH at 0-5°C.
- Convert chalcone intermediates into 4-(1-acetyl-3-substituted phenyl-4,5-dihydro-2-pyrazolin-5-yl)phenol derivatives using hydrazine hydrate in glacial acetic acid under reflux.
- Perform final esterification by activating Ursolic Acid with trifluoroacetic anhydride in toluene, followed by coupling with the phenolic intermediates and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route disclosed in this patent offers substantial advantages that translate directly into cost efficiency and supply reliability for pharmaceutical manufacturers. The reliance on commodity chemicals such as acetophenones, benzaldehydes, and common solvents like ethanol and toluene ensures that raw material sourcing is straightforward and not subject to the volatility associated with exotic reagents. This accessibility significantly mitigates supply chain risks, allowing for consistent production scheduling and inventory management. Furthermore, the elimination of expensive transition metal catalysts or complex protecting group strategies reduces the overall bill of materials, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The simplicity of the workup procedures, which primarily involve filtration and standard solvent extraction, minimizes waste generation and lowers the environmental compliance burden, making the process highly attractive for large-scale industrial adoption.
- Cost Reduction in Manufacturing: The process achieves cost efficiency by utilizing inexpensive, readily available starting materials and avoiding the need for costly chromatographic separations at every step. The high yields reported for the intermediate steps (often exceeding 80%) mean that less raw material is wasted, directly lowering the cost per kilogram of the final active pharmaceutical ingredient (API) precursor. Additionally, the use of TFAA as an activator is more economical than many peptide coupling reagents, and the byproducts formed are easily removed during the aqueous workup, reducing solvent consumption and disposal costs.
- Enhanced Supply Chain Reliability: Because the synthesis does not depend on single-source specialty reagents or unstable catalysts, the supply chain is inherently more robust. The modular nature of the synthesis allows manufacturers to stockpile key intermediates, such as the chalcone derivatives, which can be stored stably and coupled with Ursolic Acid on demand. This flexibility enables rapid response to market fluctuations and ensures continuous availability of the final product, which is crucial for maintaining uninterrupted drug production lines and meeting regulatory delivery commitments.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-atmospheric pressure, making the transition from laboratory scale to multi-ton commercial production seamless without the need for specialized high-pressure equipment. The use of ethanol and toluene, which are well-understood solvents in the chemical industry, facilitates efficient solvent recovery and recycling systems. This aligns with modern green chemistry principles by minimizing hazardous waste and energy consumption, ensuring that the manufacturing process meets stringent environmental regulations while maintaining high throughput capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these Ursolic Acid derivatives. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines. The answers are derived directly from the experimental data and process descriptions found in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of the TFAA activation method for Ursolic Acid esterification?
A: The use of trifluoroacetic anhydride (TFAA) allows for mild activation of the C28 carboxylic acid group of Ursolic Acid without requiring harsh acidic conditions that might degrade the sensitive triterpenoid skeleton, ensuring higher purity and yield.
Q: How does the structural modification affect the antitumor activity compared to native Ursolic Acid?
A: According to patent data, specific derivatives such as the 4'-chloro and 4'-fluoro chalcone esters demonstrate significantly enhanced inhibitory activity against A549 lung cancer and SKOV-3 ovarian cancer cell lines compared to the parent Ursolic Acid compound.
Q: Are the raw materials for this synthesis readily available for commercial production?
A: Yes, the synthesis relies on commodity chemicals such as p-hydroxybenzaldehyde, various acetophenones, and standard solvents like ethanol and toluene, ensuring a stable and cost-effective supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursolic Acid Derivative Supplier
As the demand for potent antitumor agents continues to rise, partnering with an experienced CDMO is essential for translating promising patent technologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for the TFAA-mediated esterification process described in CN101830961A. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ursolic Acid derivatives meets the highest international standards for pharmaceutical intermediates, providing you with a solid foundation for your clinical and commercial programs.
We invite you to collaborate with us to optimize this synthetic route for your specific requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume, helping you identify further efficiencies in the supply chain. Please contact our technical procurement team today to request specific COA data for our available batches and to discuss detailed route feasibility assessments. Let us help you accelerate your drug development timeline with our reliable supply of high-quality Ursolic Acid ester derivatives.
