Advanced Synthesis of Ursolic Acid Derivatives for High-Potency Anti-Leukemia Applications
Introduction to Novel Ursolic Acid Derivatives
The pharmaceutical landscape is constantly evolving towards more potent and selective therapeutic agents, particularly in the realm of oncology. Patent CN102532245B introduces a significant breakthrough in the field of pentacyclic triterpenoids by disclosing novel ursolic acid derivatives with enhanced anti-leukemic properties. Specifically, the patent details the synthesis and efficacy of ursolic-12-en-28-yl (7'-aminoheptanoic acid) and ursolic-12-en-28-yl (p-aminomethylbenzoic acid). These compounds represent a strategic chemical modification of the natural ursolic acid scaffold, designed to overcome limitations in solubility and bioactivity associated with the parent molecule. The core innovation lies not only in the biological outcome but also in the streamlined synthetic methodology that facilitates cost reduction in API manufacturing. By modifying the 28-position carboxyl group with specific amino acid chains, the resulting derivatives exhibit markedly improved inhibition of human leukemia U937 cell proliferation compared to existing technologies.
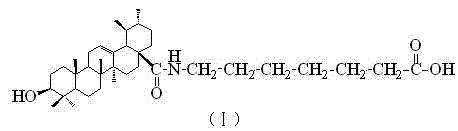
For research and development teams evaluating new leads, the structural integrity of these derivatives is paramount. The preservation of the ursolic acid backbone ensures retention of inherent biological recognition, while the appended side chains introduce new pharmacophoric elements. This dual approach maximizes the therapeutic index, making these compounds highly attractive candidates for further drug development. As a reliable pharmaceutical intermediates supplier, understanding the precise structural nuances depicted in the patent is essential for ensuring batch-to-batch consistency and regulatory compliance. The ability to synthesize these complex molecules efficiently positions manufacturers to support the growing demand for next-generation anti-cancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of ursolic acid amide derivatives involves a rigorous multi-step process that often bottlenecks production efficiency. Conventional methodologies typically require the isolation and purification of the activated acyl chloride intermediate before proceeding to the amidation step. This intermediate, 3-O-acetylarbutane-28-acyl chloride, is chemically reactive and can be prone to degradation or hydrolysis if not handled with extreme care. Furthermore, the purification of such intermediates usually necessitates additional solvent consumption, extended processing time, and specialized equipment, all of which contribute to elevated operational expenditures. In many prior art methods, the focus on high-purity intermediates at every stage leads to cumulative yield losses, making the final product economically challenging to produce at a commercial scale. These inefficiencies create significant hurdles for supply chain heads looking to secure stable and cost-effective sources of high-purity ursolic acid derivatives.
The Novel Approach
The methodology outlined in CN102532245B disrupts this traditional paradigm by introducing a 'telescoped' synthesis strategy that bypasses the purification of the acyl chloride intermediate. Instead of isolating the 3-O-acetylarbutane-28-acyl chloride, the process utilizes the crude reaction mixture directly in the subsequent amidation step. This bold simplification drastically reduces the number of unit operations, minimizes solvent waste, and shortens the overall production cycle. By demonstrating that the crude intermediate is sufficiently pure to drive the amidation reaction to completion without compromising the final product quality, the patent offers a robust solution for cost reduction in pharmaceutical intermediates manufacturing. This approach not only enhances the economic viability of the process but also aligns with green chemistry principles by reducing the environmental footprint associated with excessive purification steps.
Mechanistic Insights into the Three-Step Synthesis Strategy
The synthetic route described in the patent is a masterclass in functional group manipulation, leveraging the distinct reactivity of the ursolic acid scaffold. The first critical transformation involves the protection of the 3-hydroxyl group. Ursolic acid possesses both a carboxylic acid at C-28 and a hydroxyl group at C-3. To achieve selective amidation at the C-28 position, the C-3 hydroxyl must be masked. This is achieved through acetylation using acetic anhydride in pyridine at elevated temperatures (100°C). This step ensures that the hydroxyl group remains inert during the subsequent activation of the carboxylic acid, preventing the formation of unwanted ester byproducts.
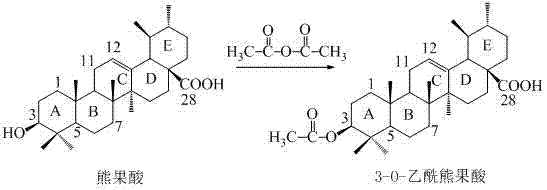
Following protection, the activation of the C-28 carboxyl group is performed using oxalyl chloride in dichloromethane. This reaction converts the carboxylic acid into a highly reactive acyl chloride, which is the key electrophile for the upcoming amidation. The use of oxalyl chloride is preferred due to the volatility of its byproducts (CO and CO2), which simplifies the workup procedure. The patent explicitly notes that the resulting 3-O-acetylarbutane-28-acyl chloride is used as a crude product, a decision that relies on the robustness of the subsequent coupling reaction.
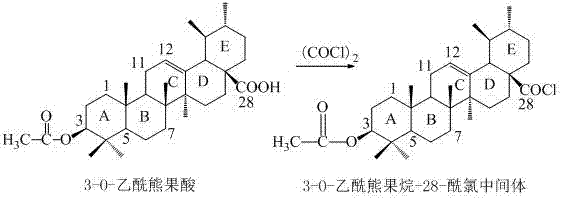
The final and most crucial step is the amidation followed by deprotection. The crude acyl chloride is reacted with amino acid ester hydrochlorides, such as ethyl 7-aminoheptanoate hydrochloride, in the presence of triethylamine. Triethylamine acts as a base to scavenge the HCl generated during the reaction, driving the equilibrium towards the amide product. The reaction is conducted at mild temperatures (20-30°C) to prevent racemization or degradation. After the amide bond is formed, the acetyl protecting group is removed via hydrolysis using aqueous sodium hydroxide. This final step reveals the free 3-hydroxyl group, yielding the target ursolic acid derivative. The entire sequence is designed to maximize yield while minimizing purification burdens, a critical factor for commercial scalability.
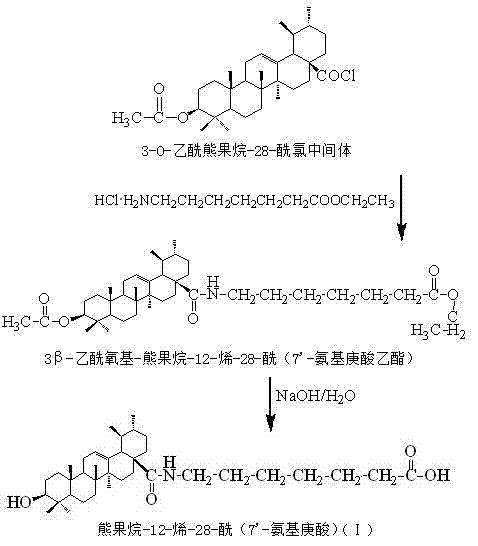
How to Synthesize Ursolic Acid Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters to ensure high purity and yield. The process begins with the dissolution of ursolic acid in pyridine, followed by the dropwise addition of acetic anhydride. Maintaining the temperature at 100°C for 4 to 5 hours is essential for complete acetylation. Once the 3-O-acetyl ursolic acid is isolated, it is dissolved in dichloromethane and treated with excess oxalyl chloride at room temperature. The removal of solvent and excess reagent yields the crude acyl chloride, which is immediately redissolved for the next step without further purification. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the guide below.
- Protect the 3-hydroxyl group of ursolic acid via acetylation using acetic anhydride and pyridine at 100°C.
- Activate the 28-carboxyl group by reacting with oxalyl chloride in dichloromethane to form the crude acyl chloride intermediate.
- React the crude intermediate with ethyl 7-aminoheptanoate hydrochloride using triethylamine, followed by silica gel purification and NaOH hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical innovations in CN102532245B translate directly into tangible business benefits. The primary advantage lies in the significant simplification of the manufacturing process. By eliminating the purification step for the acyl chloride intermediate, manufacturers can reduce the consumption of expensive solvents like dichloromethane and silica gel. This reduction in material usage leads to substantial cost savings in raw materials and waste disposal. Furthermore, the shortened process flow means that production cycles are faster, allowing for increased throughput and better responsiveness to market demand. This efficiency is crucial for maintaining a competitive edge in the supply of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The decision to use the crude acyl chloride intermediate is a major driver for cost optimization. In traditional synthesis, purifying reactive intermediates often requires column chromatography or recrystallization, which are labor-intensive and result in material loss. By skipping this step, the process inherently improves the overall mass balance. Additionally, the reagents used, such as oxalyl chloride and triethylamine, are commodity chemicals available from multiple global suppliers, ensuring price stability and reducing the risk of supply chain disruptions. This strategic sourcing flexibility allows for better negotiation power and long-term cost predictability.
- Enhanced Supply Chain Reliability: A simpler synthesis route is inherently more robust and less prone to failure. Complex purification steps often introduce variability; by removing them, the process becomes more reproducible. This reliability is vital for securing long-term supply contracts with pharmaceutical companies. The use of standard reaction conditions (e.g., room temperature for acyl chloride formation, mild heating for acetylation) means that the process can be easily transferred between different manufacturing sites without requiring specialized equipment. This flexibility ensures continuity of supply even in the face of regional logistical challenges.
- Scalability and Environmental Compliance: As the demand for anti-cancer agents grows, the ability to scale production is paramount. This synthetic route is highly amenable to scale-up, moving seamlessly from laboratory grams to multi-ton commercial production. The reduction in solvent usage and waste generation also aligns with increasingly stringent environmental regulations. Minimizing the use of silica gel and organic solvents reduces the burden on waste treatment facilities, lowering the environmental compliance costs associated with manufacturing. This sustainability aspect is becoming a key differentiator for suppliers seeking partnerships with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these ursolic acid derivatives. The answers are derived directly from the experimental data and claims presented in patent CN102532245B, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the crude acyl chloride intermediate used directly in this synthesis?
A: Using the crude 3-O-acetylarbutane-28-acyl chloride intermediate eliminates a costly and time-consuming purification step. The subsequent amidation and hydrolysis steps are robust enough to tolerate minor impurities, which are removed during the final silica gel column chromatography, significantly reducing production costs.
Q: What is the biological advantage of these specific ursolic acid derivatives?
A: Unlike unmodified ursolic acid, these derivatives (specifically ursolic-12-en-28-yl (7'-aminoheptanoic acid)) demonstrate superior inhibitory effects on human leukemia U937 cell proliferation, particularly at lower concentrations, reducing potential toxicity while maintaining efficacy.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process utilizes standard reagents like oxalyl chloride and triethylamine under mild conditions (20-30°C for amidation). The elimination of intermediate purification makes it highly suitable for large-scale commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursolic Acid Derivatives Supplier
The development of potent anti-leukemia agents requires a manufacturing partner who understands both the chemical complexity and the commercial imperatives of the pharmaceutical industry. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in triterpenoid chemistry to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of ursolic acid derivatives meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for these critical oncology intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Whether you require specific COA data for regulatory filings or detailed route feasibility assessments for process optimization, we are committed to supporting your success. Contact us today to discuss how our advanced synthesis capabilities can accelerate your drug development timeline and enhance your market competitiveness.
