Advanced Refinement of Sugammadex Sodium for Commercial Scale API Manufacturing
Advanced Refinement of Sugammadex Sodium for Commercial Scale API Manufacturing
The pharmaceutical landscape for neuromuscular blockade reversal agents has been significantly advanced by the innovations detailed in patent CN110627925B, which introduces a robust method for refining Sugammadex Sodium. This specific chemical entity, known commercially as Bridion, serves as a critical selective muscle relaxant antagonist, and its therapeutic efficacy is intrinsically linked to its chemical purity. The patent addresses a longstanding challenge in the industry: the removal of structurally similar dimer impurities that persist through conventional purification workflows. By leveraging a sophisticated poor solvent and water system, this technology enables the production of high-purity active pharmaceutical ingredients (APIs) that meet rigorous global safety standards. For procurement and supply chain leaders, understanding this refinement capability is essential for securing a reliable Sugammadex Sodium supplier who can guarantee consistency and quality in large-volume orders.
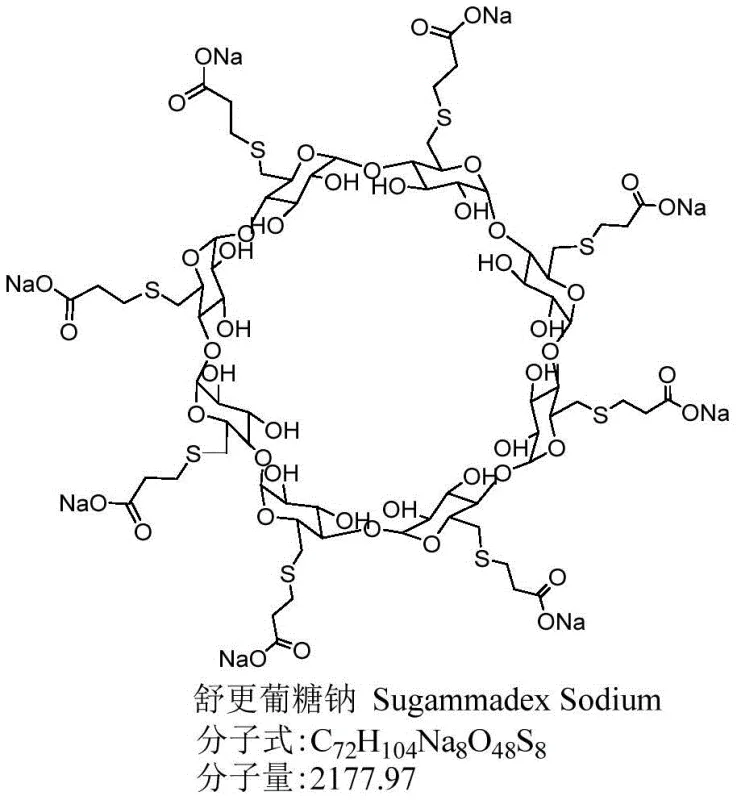
Traditionally, the purification of Sugammadex Sodium has relied on methods that are either resource-intensive or insufficiently selective. Conventional approaches often involve prolonged dialysis processes lasting up to 36 hours, which consume vast quantities of water and generate substantial waste liquid, creating environmental and cost burdens. Alternatively, column chromatography using silica gel or Sephadex G25 often fails to achieve the necessary purity levels, while adsorption methods using specific activated carbons can lead to significant product loss and rely on single-source suppliers for critical materials. Furthermore, methods involving strong inorganic acids pose stability risks to the cyclodextrin substrate, potentially generating acid degradation impurities that compromise patient safety. These limitations highlight the urgent need for a more efficient, scalable, and chemically gentle purification strategy that can be implemented in standard industrial facilities without specialized equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on dialysis and adsorption techniques presents significant bottlenecks for commercial scale-up of complex pharmaceutical intermediates. Dialysis, while effective at removing small molecules, is notoriously slow and inefficient for separating large molecular weight impurities like dimers that share similar physicochemical properties with the target molecule. This inefficiency translates directly into extended production cycles and increased utility costs. Moreover, adsorption-based purification often requires proprietary adsorbents, such as specific grades of activated carbon from limited international manufacturers, introducing supply chain vulnerabilities. When these adsorbents are used in large excess, as seen in some prior art requiring 20-150% w/w ratios, the physical entrapment of the product leads to reduced yields. Additionally, the use of strong acidic conditions in certain ion-exchange protocols can destabilize the sensitive thioether linkages in Sugammadex, leading to the formation of degradation products that are difficult to remove in subsequent steps.
The Novel Approach
The methodology disclosed in patent CN110627925B circumvents these issues by utilizing a controlled crystallization technique within a poor solvent and water system. This approach exploits the subtle solubility differences between the monomeric Sugammadex Sodium and its dimeric impurities, specifically Impurity A and Impurity B. By carefully tuning the ratio of water to organic poor solvents such as DMF, ethanol, or acetonitrile, the process preferentially precipitates the dimer impurities into a solid phase during the initial separation steps. This allows the target molecule to remain in the mother liquor, effectively enriching the solution with the desired product while discarding the contaminants. The subsequent crystallization step from this purified mother liquor yields a product with exceptional purity, eliminating the need for time-consuming dialysis or expensive, loss-prone adsorption columns. This represents a significant technological leap in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Solvent-Induced Impurity Separation
The core mechanism driving this purification success lies in the differential solubility behavior of the cyclodextrin derivatives in mixed solvent systems. Sugammadex Sodium, being a highly polar molecule due to its multiple carboxylate groups, exhibits specific solubility characteristics in water-organic mixtures. The dimer impurities, formed through side reactions during the initial synthesis involving octaiodo-gamma-cyclodextrin and 3-mercaptopropionic acid, possess different hydrophobic-hydrophilic balances. When a poor solvent is introduced to the aqueous solution of the crude product, the solubility of the dimeric species drops more precipitously than that of the monomer. This phenomenon allows for a liquid-liquid or solid-liquid separation where the impurities are physically segregated. The process avoids harsh chemical transformations, relying instead on physical chemistry principles to achieve separation, thereby preserving the structural integrity of the API.
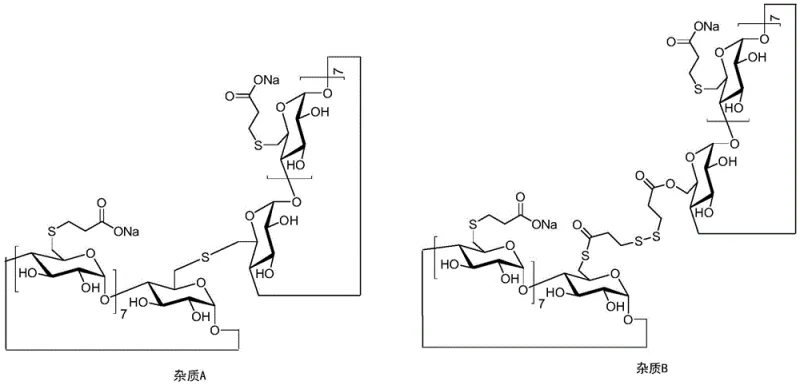
Furthermore, the control of impurity profiles is critical for regulatory compliance, particularly for genotoxic or pharmacologically active related substances. The patent details the identification and structural elucidation of key impurities, including Impurity A and Impurity B, which are dimeric in nature. Understanding their formation pathways, often stemming from incomplete substitution or intermolecular coupling during the thiolation reaction, allows for better upstream control. However, even with optimized synthesis, trace amounts persist. The refinement method described ensures that these specific dimers are reduced to below detection limits or less than 0.1%. This level of control is achieved without the use of protective agents like glutathione or cysteine, which were previously thought necessary to prevent oxidation but often complicated the purification matrix. The simplicity of the solvent system ensures that no new extraneous impurities are introduced during the refining stage.
How to Synthesize Sugammadex Sodium Efficiently
The synthesis and subsequent refinement of Sugammadex Sodium involve a streamlined sequence designed for maximum recovery and purity. The process begins with the preparation of the crude material, followed by the critical three-step refinement protocol. This protocol is designed to be robust against variations in crude quality, making it ideal for processing batches from different synthesis runs. The operational simplicity means that it can be transferred easily between manufacturing sites, ensuring consistent quality regardless of production location. For technical teams looking to implement this, the focus should be on precise control of solvent ratios and temperature gradients during the crystallization phases to maximize the exclusion of dimeric contaminants.
- Dissolve crude Sugammadex Sodium in water under inert gas, add a poor solvent like DMF or ethanol at room temperature, allow stratification, and collect the upper mother liquor.
- Treat the lower organic phase with additional water and poor solvent to precipitate solid impurities, filter the solids, and combine the filtrate with the previously collected mother liquor.
- Heat the combined mother liquor, add further poor solvent, cool the mixture to induce crystallization of high-purity Sugammadex Sodium, then filter and dry the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refinement technology offers tangible strategic benefits beyond mere chemical purity. The elimination of dependency on specialized, single-source adsorbents significantly de-risks the supply chain, ensuring continuity of supply even during global material shortages. The reduction in processing time compared to dialysis translates to faster turnaround times for batch completion, allowing for more responsive inventory management. Furthermore, the use of common industrial solvents like ethanol or DMF simplifies logistics and reduces raw material costs compared to proprietary reagents. This efficiency drives substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing structures in the final API market.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive, specialized activated carbon adsorbents that were previously required to achieve high purity levels. By replacing these consumables with standard organic solvents, the variable cost per kilogram of production is significantly lowered. Additionally, the avoidance of long-duration dialysis reduces energy consumption and labor hours associated with monitoring extended processes. The higher yield resulting from reduced product entrapment in adsorbents further contributes to overall economic efficiency, ensuring that more of the synthesized material ends up as saleable product.
- Enhanced Supply Chain Reliability: Relying on conventional solvents such as DMF, ethanol, and water removes the bottleneck of sourcing niche purification media from limited vendors. This diversification of raw material sources enhances the resilience of the supply chain against geopolitical or logistical disruptions. The scalability of the liquid-liquid separation and crystallization steps means that production volume can be increased simply by scaling up vessel sizes, without the need for complex parallel column setups. This flexibility ensures that suppliers can meet sudden spikes in demand for this critical reversal agent without compromising on quality or lead times.
- Scalability and Environmental Compliance: The method significantly reduces water consumption compared to the 36-hour dialysis processes of the past, aligning with modern green chemistry initiatives and reducing wastewater treatment loads. The solvents used are recoverable and recyclable through standard distillation processes, minimizing hazardous waste generation. This environmental stewardship not only lowers disposal costs but also ensures compliance with increasingly stringent environmental regulations in major pharmaceutical manufacturing hubs. The robustness of the process under conventional production conditions makes it highly suitable for multi-ton scale manufacturing, facilitating the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality of Sugammadex Sodium refined via this advanced method. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and quality assurance protocols.
Q: How does this refinement method improve upon traditional dialysis?
A: Unlike traditional dialysis which requires up to 36 hours and generates massive wastewater, this crystallization method is rapid, uses conventional solvents, and effectively removes difficult dimer impurities without significant product loss.
Q: What is the achieved purity level for Impurity A and B?
A: The process achieves a total purity exceeding 99.5%, with Impurity A being undetectable and Impurity B reduced to less than 0.1%, meeting stringent pharmacopoeia standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids specialized adsorbents and complex column chromatography, relying instead on scalable liquid-liquid separation and crystallization steps that are easily adapted for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity Sugammadex Sodium in ensuring patient safety and therapeutic efficacy. Our technical team has extensively analyzed the pathways described in patent CN110627925B and integrated similar advanced refinement strategies into our own manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial supplies and full-scale commercial launches. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Sugammadex Sodium we deliver meets or exceeds the 99.5% purity benchmark with minimal dimer impurities.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how our refined Sugammadex Sodium can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our high-efficiency manufacturing route. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner in the neuromuscular blockade reversal market. Contact us today to secure a reliable supply of this vital API intermediate.
