Advanced Manufacturing of Difluoromethylpyrazolyl Carboxylates for Global Agrochemical Supply Chains
Advanced Manufacturing of Difluoromethylpyrazolyl Carboxylates for Global Agrochemical Supply Chains
The development of efficient synthetic routes for fluorinated heterocycles remains a critical priority for the global agrochemical industry, particularly for the production of next-generation fungicides. Patent CN101535274A discloses a groundbreaking process for preparing difluoromethyl-substituted pyrazol-4-yl carboxylates of Formula (I), which serve as pivotal intermediates in the synthesis of potent mycocides. The introduction of the difluoromethyl group is strategically significant, as it enhances the lipophilicity and metabolic stability of the final active pharmaceutical ingredients without the environmental persistence associated with trifluoromethyl groups. This technical insight report analyzes the novel magnesium-mediated reductive cyclization strategy that bypasses traditional bottlenecks, offering a robust pathway for reliable agrochemical intermediate suppliers to secure their supply chains against raw material volatility.
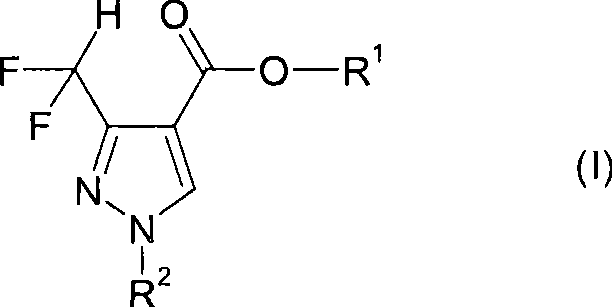
The core innovation lies in the structural versatility of Formula (I), where R1 can range from simple C1-C8 alkyl groups to complex benzyl or alkenyl substituents, allowing for extensive downstream derivatization. For R&D directors focusing on impurity profiles, the ability to tune R2 from hydrogen to methyl or phenyl groups provides a powerful handle for optimizing biological activity and solubility parameters. The patent explicitly demonstrates that this methodology is not limited to laboratory scales but is designed with industrial feasibility in mind, utilizing reagents that are commercially abundant and cost-effective. By establishing a reliable source for these high-purity intermediates, manufacturers can accelerate the development pipeline for new crop protection agents while adhering to stringent regulatory standards regarding fluorine content and residual metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-difluoromethyl-pyrazole-4-carboxylic acid derivatives has been hindered by the reliance on scarce and prohibitively expensive starting materials. Prior art, such as WO 92/12970, describes routes initiating from 4,4-difluoroacetoacetic ester, a compound that is notoriously difficult to procure in bulk quantities and commands a premium price due to complex fluorination steps required for its manufacture. Furthermore, alternative methods involving halogen exchange on heteroaromatic carboxylates, as seen in WO 2005/044804, necessitate the use of hazardous and costly fluorinating agents. These conventional approaches impose severe constraints on cost reduction in fungicide manufacturing, as the high input cost of the difluoro-building block propagates through the entire value chain. Additionally, these older methods often suffer from poor regioselectivity, generating mixtures of isomers that require energy-intensive and yield-reducing purification steps to isolate the biologically active 4-carboxylate isomer.
The Novel Approach
The process disclosed in CN101535274A fundamentally disrupts this status quo by utilizing readily available 4,4,4-trihaloacetoacetate derivatives, specifically those where the trihalo group is trifluoromethyl or chlorodifluoromethyl. As illustrated in the synthesis of the key intermediate Formula (II), the process begins with the condensation of trifluoroacetoacetic ester (VII) with trialkyl orthoformates (VIII), a reaction that is well-established and highly efficient. 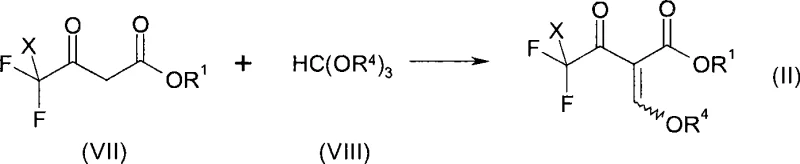
This strategic shift allows manufacturers to leverage the commodity pricing of trifluoroacetoacetates, effectively decoupling the production cost from the scarcity of difluoro-precursors. The subsequent transformation involves a sophisticated magnesium-mediated reduction in the presence of chlorosilanes, which selectively reduces one of the fluorine atoms to generate the difluoromethyl moiety in situ. This approach not only simplifies the supply chain by relying on stable, shelf-stable reagents but also dramatically improves the atom economy of the process. For procurement managers, this translates to a significant mitigation of supply risk, as the raw materials are produced by multiple global vendors, ensuring continuity of supply even during market fluctuations.
Mechanistic Insights into Magnesium-Mediated Reductive Cyclization
The heart of this technology is the unique reactivity observed in Step A, where the trihalo-beta-keto ester derivative (Formula II) undergoes reductive silylation. When reacted with a metal having a redox potential of less than -0.7 V, such as magnesium, zinc, or sodium, in the presence of a chlorosilane like trimethylsilyl chloride, the system generates a silylated enol intermediate (Compound A). 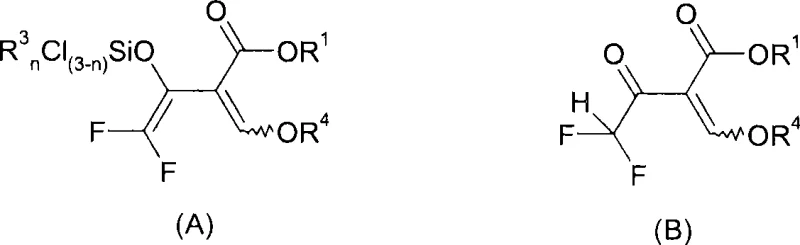
Mechanistically, the metal inserts into the carbon-halogen bond, facilitating the elimination of a halide ion and the concurrent formation of a carbon-silicon bond at the enol oxygen. This silyl protection is crucial as it stabilizes the enol form and directs the subsequent nucleophilic attack. The patent data suggests that this intermediate can exist in equilibrium with its hydrolyzed form (Compound B) depending on the water content of the reaction mixture. However, maintaining anhydrous conditions during this phase is preferred to prevent premature hydrolysis, which could lead to side reactions. The choice of solvent plays a pivotal role here; polar aprotic solvents like N-methylpyrrolidone (NMP) or dimethylformamide (DMF) are essential to solvate the magnesium cations and facilitate the electron transfer process, ensuring high conversion rates of the starting material.
Following the formation of the silylated intermediate, the addition of a hydrazine derivative (Formula III) triggers the cyclization event that constructs the pyrazole ring. The regioselectivity of this cyclization is the defining feature of this process, heavily favoring the formation of the 3-difluoromethyl-4-carboxylate isomer (Isomer A) over the 3-carboxylate isomer (Isomer B). This selectivity is believed to be driven by the electronic influence of the difluoromethyl group and the steric environment created by the silyl group during the transition state. For quality control teams, this high intrinsic selectivity (often exceeding 80:20 ratios) minimizes the burden on downstream purification, allowing for the production of high-purity API intermediates with reduced solvent consumption and waste generation. The ability to fine-tune this ratio by adjusting reaction temperatures between -10°C and 60°C provides process engineers with additional levers to optimize the impurity profile for specific regulatory requirements.
How to Synthesize Difluoromethylpyrazolyl Carboxylates Efficiently
The operational protocol for this synthesis is designed to balance reaction kinetics with thermal safety, making it amenable to both batch and continuous processing environments. The initial step requires the activation of the metal surface, typically magnesium turnings, using agents like iodine or 1,2-dibromoethane to ensure rapid initiation of the reduction. Once activated, the trihaloacetoacetate derivative is added slowly to the mixture of metal and chlorosilane in a dry aprotic solvent, maintaining the temperature below 30°C to manage the exotherm. This controlled addition prevents the accumulation of unreacted reagents and ensures the steady formation of the silylated enol. Subsequently, the hydrazine component is introduced, often as an aqueous solution or in a protic solvent like ethanol, which facilitates the cyclization while simultaneously hydrolyzing the silyl group to reveal the final pyrazole structure.
- React a 4,4,4-trihaloacetoacetate derivative (Formula II) with a chlorosilane and a reactive metal like magnesium in an aprotic solvent to form a silylated enol intermediate.
- Without isolating the intermediate, react the mixture with a hydrazine compound (Formula III) under controlled temperature conditions to effect cyclization.
- Purify the resulting difluoromethylpyrazole carboxylate (Formula I) via crystallization or extraction, achieving high regioselectivity for the 4-position isomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers a compelling value proposition for stakeholders focused on margin improvement and supply chain resilience. The elimination of expensive difluoroacetoacetate starting materials represents a direct and substantial cost saving, as the process substitutes them with commodity-grade trifluoroacetoacetates and orthoformates. This raw material substitution does not merely lower the bill of materials; it fundamentally alters the risk profile of the supply chain by removing dependence on niche fluorination specialists. Furthermore, the use of magnesium and chlorosilanes, which are produced at massive scales for the silicone and metallurgical industries, ensures that the reagents are subject to stable pricing dynamics rather than the volatility often seen in specialized fine chemical markets.
- Cost Reduction in Manufacturing: The process achieves cost optimization through multiple vectors, primarily by avoiding the multi-step synthesis of specialized difluoro-building blocks. By performing the defluorination in situ during the cyclization sequence, the number of unit operations is reduced, leading to lower labor and utility costs. Additionally, the high regioselectivity minimizes the loss of valuable material to isomeric byproducts, thereby improving the overall yield per kilogram of input. The ability to use technical grade solvents and reagents without compromising the purity of the final crystal further contributes to a leaner cost structure, making the final intermediate highly competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain heads will find significant value in the robustness of the reagent list. Magnesium, trimethylsilyl chloride, and hydrazine hydrate are all high-volume industrial chemicals with multiple qualified suppliers globally, reducing the risk of single-source bottlenecks. The process tolerance to various R1 and R2 substituents means that a single manufacturing line can be adapted to produce a wide portfolio of analogues, enhancing asset utilization. This flexibility allows manufacturers to respond rapidly to changing market demands for different fungicide variants without requiring extensive retooling or new vendor qualification processes, ensuring consistent delivery performance to downstream formulators.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be safely scaled from pilot plants to multi-ton commercial reactors without requiring exotic high-pressure or cryogenic equipment. The exothermic nature of the magnesium reduction is manageable with standard cooling jackets, and the subsequent cyclization proceeds efficiently at near-ambient temperatures. From an environmental standpoint, the process avoids the use of heavy metal catalysts or stoichiometric amounts of toxic fluorinating agents, simplifying waste treatment and disposal. The aqueous workup procedures described allow for the efficient recovery of solvents and the safe neutralization of acidic byproducts, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoromethylpyrazole synthesis technology. These insights are derived directly from the experimental data and claims within the patent specification, providing a factual basis for feasibility assessments. Understanding these nuances is essential for technical teams evaluating the integration of this route into existing production schedules or for R&D groups designing new molecular entities around this scaffold.
Q: What is the primary advantage of this magnesium-mediated process over conventional methods?
A: The primary advantage is the avoidance of expensive and difficult-to-source 4,4-difluoroacetoacetate starting materials. Instead, this process utilizes readily available and cost-effective 4,4,4-trifluoroacetoacetate derivatives, significantly reducing raw material costs while maintaining high regioselectivity for the desired pyrazole isomer.
Q: How does the process control regioselectivity during pyrazole ring formation?
A: The process achieves high regioselectivity through the specific formation of a silylated enol intermediate (Compound A) using magnesium and chlorosilanes. This intermediate directs the subsequent cyclization with hydrazines to favor the formation of the 3-difluoromethyl-4-carboxylate isomer over the 2-difluoromethyl-3-carboxylate isomer, often achieving ratios exceeding 80:20.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly scalable. It operates under moderate temperatures (-10°C to 60°C) and uses common industrial reagents like magnesium turnings and trimethylsilyl chloride. The elimination of complex purification steps for intermediates and the use of robust solvents like DMF or NMP facilitate safe and efficient scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylpyrazolyl Carboxylates Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this magnesium-mediated process are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex fluorine-containing impurity profiles, guaranteeing that every batch of difluoromethylpyrazolyl carboxylates meets the exacting standards required for agrochemical registration.
We invite global partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and cost targets. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your competitive position in the fungicide market.
