Advanced Manufacturing of Difluoromethyl Pyrazole Intermediates for Global Agrochemical Supply Chains
Advanced Manufacturing of Difluoromethyl Pyrazole Intermediates for Global Agrochemical Supply Chains
The global demand for high-performance agrochemicals, particularly fungicides containing difluoromethyl pyrazole motifs, necessitates robust and economically viable synthetic routes. Patent CN101535274A introduces a groundbreaking methodology for preparing difluoromethyl-substituted pyrazol-4-yl carboxylates of formula (I), which serve as critical building blocks in the synthesis of modern crop protection agents. This technology addresses long-standing challenges in fluorine chemistry by enabling the use of abundant trifluoro-methyl precursors instead of scarce difluoro-analogues. The core innovation lies in a clever reductive silylation strategy that effectively converts a trifluoromethyl group into a difluoromethyl moiety during the construction of the heterocyclic ring. 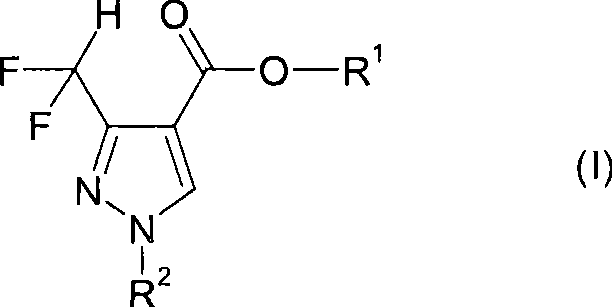 For procurement specialists and R&D directors alike, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms. By circumventing the need for specialized fluorinating agents and expensive starting materials, the process outlined in CN101535274A offers a reliable pathway for securing high-purity intermediates essential for the next generation of agricultural solutions.
For procurement specialists and R&D directors alike, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms. By circumventing the need for specialized fluorinating agents and expensive starting materials, the process outlined in CN101535274A offers a reliable pathway for securing high-purity intermediates essential for the next generation of agricultural solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-difluoromethyl-1-methyl-pyrazol-4-yl carboxylic acid derivatives has been hindered by the prohibitive cost and limited availability of key starting materials. Traditional routes, such as those described in WO 92/12970, typically rely on 4,4-difluoroacetoacetic ester as the primary feedstock. This specific fluorinated beta-keto ester is not only expensive to produce but also presents significant supply chain vulnerabilities due to the complex multi-step fluorination processes required for its manufacture. Furthermore, alternative strategies involving halogen exchange on heteroaromatic carboxylates, as seen in WO 2005/044804, often necessitate the use of hazardous and costly fluorinating agents. These reagents demand stringent safety measures, specialized equipment, and rigorous waste management protocols, all of which inflate the overall production cost and extend the lead time for high-purity agrochemical intermediates. Consequently, manufacturers have faced a bottleneck where the economic feasibility of producing these valuable fungicide precursors was compromised by the upstream complexity of raw material sourcing.
The Novel Approach
The methodology disclosed in CN101535274A elegantly bypasses these obstacles by utilizing readily available and inexpensive trifluoromethyl carbonyl compounds, such as 2,2,2-trifluoroacetyl acetic ester or 2-chloro-2,2-difluoro acetylacetic ester, as the starting point. The process employs a reductive silylation step using base metals like magnesium or zinc in the presence of chlorosilanes, which facilitates the selective removal of one fluorine atom while simultaneously protecting the enol functionality. This transformation generates a reactive silylated intermediate that can be directly cyclized with hydrazines to form the target pyrazole ring. 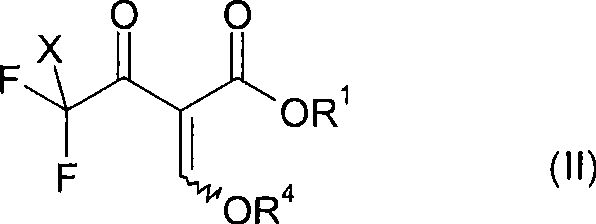 This approach not only drastically simplifies the synthetic sequence but also eliminates the dependency on rare difluoro-precursors. The ability to start from commodity chemicals translates directly into substantial cost savings and enhanced supply chain reliability, making this novel route highly attractive for large-scale commercial production of complex polymer additives and agrochemical intermediates.
This approach not only drastically simplifies the synthetic sequence but also eliminates the dependency on rare difluoro-precursors. The ability to start from commodity chemicals translates directly into substantial cost savings and enhanced supply chain reliability, making this novel route highly attractive for large-scale commercial production of complex polymer additives and agrochemical intermediates.
Mechanistic Insights into Reductive Silylation and Cyclization
The heart of this innovative process lies in the mechanistic interplay between the metal reductant, the silane protecting group, and the fluorinated substrate. In the first critical step, a compound of general formula (II), which contains a trifluoromethyl or chlorodifluoromethyl group, is reacted with a silane compound such as trimethylsilyl chloride in the presence of a metal with a redox potential of less than -0.7 V. Magnesium is particularly preferred for this transformation. The metal acts as a single-electron donor, initiating a reductive cleavage of the carbon-fluorine bond. Concurrently, the silyl group traps the resulting enolate, forming a silylated enol ether intermediate (Compound A). This intermediate is crucial because it stabilizes the reactive species and prevents unwanted side reactions that could lead to impurities. The reaction is typically conducted in aprotic polar solvents like N-Methyl pyrrolidone (NMP) or Dimethylformamide (DMF) at controlled temperatures ranging from -10°C to +60°C to ensure optimal conversion and selectivity.
Following the formation of the silylated intermediate, the second step involves cyclization with a hydrazine derivative of formula (III). Interestingly, the process does not require the isolation of the silylated intermediate; the reaction mixture from step (a) can be treated directly with the hydrazine. Upon addition of the hydrazine, often in an alcoholic solvent like ethanol or methanol, the silyl group is hydrolyzed or displaced, and the nucleophilic nitrogen attacks the carbonyl carbon. This triggers a cascade of condensation and cyclization events that construct the pyrazole ring with remarkable regioselectivity. The mechanism favors the formation of the 4-carboxylate isomer (Isomer A) over the 3-carboxylate isomer (Isomer B), with experimental data showing isomer ratios as high as 90:10 under optimized conditions. This high degree of regiocontrol is vital for pharmaceutical and agrochemical applications where specific isomeric purity is mandated by regulatory standards.
How to Synthesize Difluoromethyl Pyrazole Carboxylates Efficiently
To implement this synthesis effectively, one must first prepare the key enol ether intermediate from cheap trifluoroacetoacetate derivatives. As illustrated in the reaction scheme below, trifluoroacetyl acetic ester reacts with triethyl orthoformate and acetic anhydride to yield the necessary precursor. 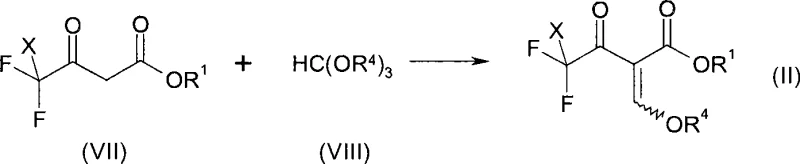 This precursor is then subjected to the reductive silylation conditions described previously. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are provided in the guide below to ensure reproducibility and safety in your pilot plant operations.
This precursor is then subjected to the reductive silylation conditions described previously. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are provided in the guide below to ensure reproducibility and safety in your pilot plant operations.
- Prepare the enol ether intermediate (Formula II) by reacting trifluoroacetoacetate with trialkyl orthoformate and acetic anhydride at elevated temperatures.
- Perform reductive silylation using magnesium or zinc metal and a chlorosilane (e.g., TMSCl) in an aprotic polar solvent like DMF or NMP to generate the silylated enol.
- React the crude silylated intermediate directly with a hydrazine derivative (Formula III) in an alcoholic solvent to form the final pyrazole ring with high regioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101535274A offers transformative benefits that extend far beyond simple chemical curiosity. The primary advantage is the decoupling of production costs from the volatile market prices of specialized fluorinated reagents. By shifting the feedstock base to ubiquitous trifluoromethyl compounds, manufacturers can achieve significant cost reduction in agrochemical intermediate manufacturing. This strategic pivot mitigates the risk of supply disruptions associated with niche fluorine chemistry suppliers and allows for more accurate long-term budget forecasting. Furthermore, the use of common base metals like magnesium and zinc, rather than precious metal catalysts, simplifies the procurement of reagents and reduces the environmental burden associated with heavy metal waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive 4,4-difluoroacetoacetic ester from the bill of materials results in a drastic decrease in raw material expenditure. Since trifluoroacetoacetate is produced on a much larger industrial scale, it benefits from economies of scale that difluoro-analogues do not. Additionally, the process avoids the need for costly fluorinating agents and the specialized containment equipment they require. The one-pot nature of the cyclization step, where the intermediate is not isolated, further reduces operational costs by saving on solvent usage, filtration time, and drying energy. These cumulative efficiencies translate into a highly competitive cost structure for the final active ingredient.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals ensures a stable and continuous supply of raw materials. Unlike specialized fluorinated building blocks which may have long lead times and limited supplier bases, trifluoroacetoacetate and chlorosilanes are available from multiple global vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions—operating at near-atmospheric pressure and moderate temperatures—means that the process can be easily transferred between different manufacturing sites without extensive requalification, thereby enhancing overall supply chain resilience and flexibility.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred gram batches with high yields. The use of magnesium or zinc generates metal salts as byproducts, which are generally easier to handle and dispose of compared to the complex organic waste streams generated by traditional fluorination methods. The high regioselectivity of the reaction minimizes the formation of difficult-to-separate isomers, reducing the need for resource-intensive purification steps like chromatography. This aligns well with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding waste discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoromethyl pyrazole synthesis technology. These answers are derived directly from the experimental data and claims within patent CN101535274A, providing a factual basis for your evaluation of this process. Understanding these nuances is critical for R&D teams planning pilot runs and for quality assurance teams establishing specification limits.
Q: How does this process improve cost efficiency compared to conventional methods?
A: The process replaces expensive 4,4-difluoroacetoacetate starting materials with significantly cheaper trifluoroacetoacetate derivatives, utilizing a reductive defluorination strategy that lowers raw material costs substantially.
Q: What is the regioselectivity of the pyrazole formation?
A: The method achieves high regioselectivity for the 4-carboxylate isomer (Isomer A) over the 3-carboxylate isomer (Isomer B), with ratios often exceeding 80:20 or even 90:10 depending on solvent and conditions.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes common industrial metals like magnesium and zinc and standard solvents like DMF and ethanol, making it highly suitable for commercial scale-up without requiring exotic catalysts or extreme pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylpyrazolyl carboxylates Supplier
The technological breakthroughs detailed in CN101535274A underscore the immense potential of difluoromethyl pyrazole derivatives in modern agrochemistry. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of intermediate meets the exacting standards required for global regulatory filings.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods sold.
