Advanced Synthetic Route for Difluoromethyl Pyrazole Intermediates and Commercial Scalability
Advanced Synthetic Route for Difluoromethyl Pyrazole Intermediates and Commercial Scalability
The chemical landscape for producing high-value agrochemical intermediates has been significantly reshaped by the innovations disclosed in patent CN101535274B, which introduces a robust and economically viable pathway for synthesizing difluoromethyl-substituted pyrazol-4-yl carboxylates. These specific molecular scaffolds serve as critical building blocks for next-generation fungicides, offering superior biological activity and environmental stability compared to their non-fluorinated counterparts. The core breakthrough lies in the strategic avoidance of prohibitively expensive 4,4-difluoroacetoacetate precursors, traditionally a major bottleneck in industrial scale-up. By leveraging readily available trifluoromethyl carbonyl compounds and employing a novel reductive defluorination strategy, this process not only lowers the barrier to entry for manufacturers but also enhances the overall purity profile of the final active pharmaceutical ingredients. For R&D directors and procurement specialists alike, understanding the nuances of this synthetic route is paramount for securing a competitive edge in the global supply chain.
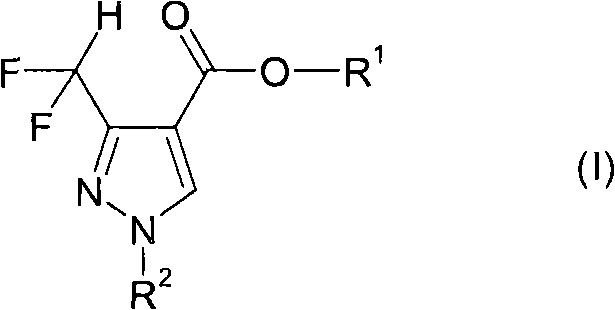
The structural versatility of the target compounds, as depicted in the general formula, allows for extensive derivatization to meet specific agronomic needs. The ability to vary the ester group R1 and the nitrogen substituent R2 provides chemists with the flexibility to tune physicochemical properties such as solubility and metabolic stability. This adaptability is crucial for developing formulations that perform effectively under diverse field conditions. Furthermore, the high yield and regioselectivity reported in the patent data suggest that this methodology can consistently produce material that meets stringent quality specifications required by regulatory bodies. Consequently, adopting this synthetic approach can significantly streamline the development timeline for new fungicidal agents, allowing companies to bring innovative crop protection solutions to market more rapidly than previously possible.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-difluoromethylpyrazole derivatives has been hindered by the reliance on scarce and costly starting materials, specifically 4,4-difluoroacetoacetate, as described in prior art such as WO 92/12970. The preparation of this key precursor often involves complex multi-step sequences that suffer from poor atom economy and generate significant amounts of hazardous waste, thereby inflating the overall production cost. Additionally, the handling of fluorinating agents required in alternative halogen-exchange methods poses serious safety risks and necessitates specialized corrosion-resistant equipment, which further capital expenditure for manufacturing facilities. These economic and operational constraints have long acted as a barrier to the widespread adoption of difluoromethyl-pyrazole based fungicides, limiting their availability to only high-margin niche applications. For supply chain managers, the dependency on such specialized raw materials creates vulnerability to market fluctuations and potential disruptions in the sourcing of critical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN101535274B circumvents these challenges by utilizing abundant and inexpensive trifluoromethyl carbonyl compounds, such as 2,2,2-trifluoroacetoacetate, as the primary feedstock. This strategic shift in raw material selection drastically reduces the cost of goods sold (COGS) while simplifying the procurement process, as these trifluoro-precursors are commodity chemicals available from multiple global suppliers. The process employs a clever reductive defluorination step using common metals like magnesium or zinc in the presence of chlorosilanes, effectively converting the trifluoromethyl group into the desired difluoromethyl moiety in situ. This transformation is not only chemically efficient but also operationally simple, proceeding under mild conditions that do not require extreme temperatures or pressures. By eliminating the need for exotic fluorinating reagents and expensive difluoro-starting materials, this novel approach offers a sustainable and scalable solution for the mass production of high-purity agrochemical intermediates.
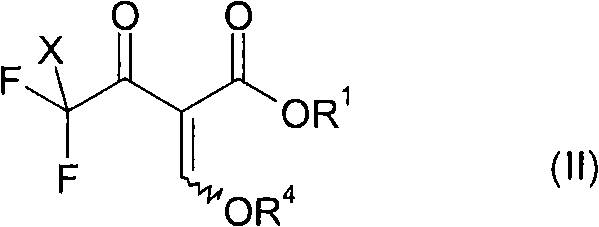
Mechanistic Insights into Reductive Silylation and Cyclization
The heart of this innovative process lies in the initial reductive silylation step, where a 4,4,4-trihalogen substituted acetoacetate derivative reacts with a chlorosilane and a reducing metal to generate a reactive silylated enol intermediate. Mechanistically, the metal, typically magnesium or zinc with a redox potential less than -0.7 V, facilitates the single-electron transfer required to cleave the carbon-halogen bond, initiating the defluorination sequence. The presence of the chlorosilane serves a dual purpose: it traps the resulting enolate species to prevent unwanted side reactions and stabilizes the intermediate as a silyl ether, which is crucial for controlling the subsequent regioselectivity. This careful manipulation of reaction intermediates ensures that the difluoromethyl group is positioned correctly at the 3-position of the pyrazole ring, minimizing the formation of the undesired 5-isomer. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters such as stoichiometry and addition rates to maximize yield and purity.
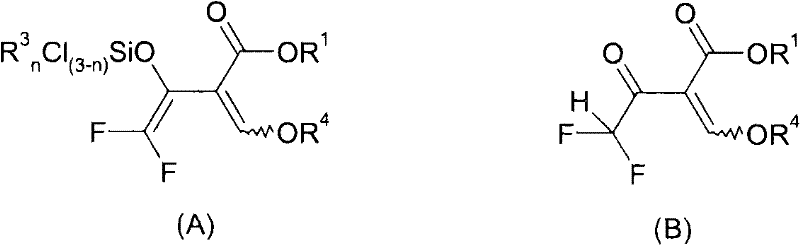
Following the formation of the silylated intermediate, the subsequent cyclization with hydrazines proceeds with remarkable efficiency, driven by the nucleophilic attack of the hydrazine nitrogen on the activated carbonyl system. The patent data indicates that this step can be performed in a one-pot manner without isolating the sensitive silylated species, thereby reducing processing time and solvent consumption. The choice of solvent plays a critical role in this stage, with protic solvents like methanol or ethanol facilitating the hydrolysis of the silyl group and promoting ring closure. Furthermore, the reaction conditions allow for the use of various substituted hydrazines, enabling the synthesis of a diverse library of N-substituted pyrazoles tailored for specific biological targets. This mechanistic robustness ensures that the process remains reliable even when scaling up from laboratory benchtop to multi-ton commercial production, providing a consistent supply of high-quality intermediates for downstream formulation.
How to Synthesize Difluoromethylpyrazolyl Carboxylates Efficiently
To implement this synthesis effectively, manufacturers must adhere to strict control over reaction parameters, particularly regarding the activation of the metal reductant and the moisture content of the solvent system. The initial step requires anhydrous conditions to prevent premature hydrolysis of the chlorosilane, ensuring that the silylated enol forms quantitatively before the introduction of the hydrazine. Detailed protocols for activating magnesium turnings, such as ultrasonic treatment or iodine etching, are essential to initiate the reaction smoothly and avoid induction periods that can lead to runaway exotherms. Once the intermediate is formed, the careful addition of the hydrazine component at controlled temperatures ensures high regioselectivity, favoring the desired 1,3-substitution pattern over the 1,5-isomer. For a comprehensive guide on the specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis steps provided below.
- React a 4,4,4-trihalogen substituted acetoacetate derivative with a chlorosilane and a reducing metal (Mg, Zn) in an aprotic solvent to form a silylated enol intermediate.
- Without isolating the intermediate, react the mixture with a hydrazine or hydrazine derivative in a protic solvent to effect cyclization and formation of the pyrazole ring.
- Purify the resulting difluoromethylpyrazolyl carboxylate through crystallization or extraction to achieve high regioselectivity and purity suitable for agrochemical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement teams seeking to optimize the cost structure of agrochemical manufacturing. The substitution of expensive difluoro-precursors with commodity trifluoro-compounds results in a substantial reduction in raw material costs, directly improving the gross margin of the final active ingredient. Moreover, the use of common reagents like magnesium and trimethylsilyl chloride eliminates the need for specialized supply chains dedicated to hazardous fluorinating agents, thereby simplifying vendor management and reducing logistical complexity. This shift towards more accessible chemistry also mitigates the risk of supply disruptions, as the key inputs are produced by a wide range of chemical manufacturers globally. For supply chain heads, this translates into enhanced reliability and continuity of supply, ensuring that production schedules can be met consistently without the fear of raw material shortages impacting delivery timelines to customers.
- Cost Reduction in Manufacturing: The elimination of costly 4,4-difluoroacetoacetate and hazardous fluorinating agents significantly lowers the direct material costs associated with production. By utilizing widely available trifluoroacetoacetates and standard reducing metals, the process avoids the premium pricing often attached to specialized fluorine chemistry reagents. This fundamental change in the bill of materials allows for a more competitive pricing strategy in the global agrochemical market. Additionally, the simplified workup procedures reduce solvent usage and energy consumption, further contributing to overall operational expense savings. These cumulative cost benefits make the technology highly attractive for large-scale commercial operations aiming to maximize profitability.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals rather than bespoke intermediates greatly strengthens the resilience of the supply chain against market volatility. Since trifluoroacetoacetates and chlorosilanes are produced in high volumes for various industries, the risk of supply bottlenecks is minimized compared to niche fluorinated building blocks. This abundance ensures that manufacturers can secure long-term contracts with multiple suppliers, fostering a stable procurement environment. Furthermore, the reduced safety hazards associated with the reagents simplify transportation and storage requirements, lowering insurance and compliance costs. This reliability is crucial for maintaining uninterrupted production flows and meeting the just-in-time delivery expectations of major agrochemical clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation under atmospheric pressure and moderate temperatures, removing the need for expensive high-pressure reactors or cryogenic cooling systems. This ease of scale-up facilitates rapid technology transfer from pilot plants to full commercial production facilities, accelerating time-to-market for new products. From an environmental standpoint, the avoidance of toxic fluorinating agents and the potential for solvent recycling align with increasingly stringent green chemistry regulations. The generation of less hazardous waste streams simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. These factors collectively enhance the sustainability profile of the production process, appealing to environmentally conscious stakeholders and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the process. Understanding these details is essential for technical teams evaluating the integration of this route into existing manufacturing portfolios. The responses cover aspects ranging from raw material selection to impurity control, ensuring a comprehensive overview of the technology's capabilities. Stakeholders are encouraged to review these points to assess the alignment of this method with their specific production goals and quality standards.
Q: What is the primary cost advantage of the process in CN101535274B?
A: The process eliminates the need for expensive 4,4-difluoroacetoacetate starting materials, substituting them with much cheaper and readily available trifluoromethyl carbonyl compounds like 2,2,2-trifluoroacetoacetate.
Q: How does this method improve regioselectivity compared to conventional routes?
A: By utilizing a specific reductive silylation step followed by controlled cyclization with substituted hydrazines, the method achieves high regioselectivity for the 3-difluoromethyl-1-substituted pyrazole isomer, minimizing difficult-to-separate byproducts.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions utilize standard atmospheric pressure and moderate temperatures with common solvents like DMF or alcohols, making it highly adaptable for commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylpyrazolyl Carboxylates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN101535274B for the agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including rigorous QC labs that enforce stringent purity specifications to meet global regulatory standards. We understand that the transition from laboratory innovation to commercial reality requires a partner who can navigate the complexities of process optimization and scale-up with precision and expertise. Our team is dedicated to delivering value through technical excellence and operational efficiency, making us the ideal choice for your difluoromethylpyrazole manufacturing needs.
We invite you to engage with our technical procurement team to discuss how we can tailor this advanced synthesis to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel route for your supply chain. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver superior results. Let us collaborate to optimize your production costs and secure a competitive advantage in the dynamic agrochemical market. Contact us today to initiate a dialogue about your project requirements and discover how NINGBO INNO PHARMCHEM can support your growth objectives.
