Advanced Synthesis of p-Acetoxystyrene for High-Performance Photoresist Manufacturing
Advanced Synthesis of p-Acetoxystyrene for High-Performance Photoresist Manufacturing
The semiconductor and microelectronics industries rely heavily on advanced photoresist materials to manufacture integrated circuits with ever-shrinking node sizes. Central to the production of 248nm deep ultraviolet photoresists is p-acetoxystyrene, a critical monomer that serves as the precursor for poly-p-hydroxystyrene resins. Patent CN112409176A introduces a transformative synthesis method that addresses the longstanding inefficiencies of traditional manufacturing routes. By shifting from a reduction-dehydration pathway to a direct palladium-catalyzed cross-coupling strategy, this technology offers a robust solution for producing high-purity intermediates. The innovation lies in the strategic use of 4-acetoxybromobenzene as a key coupling partner, which streamlines the synthetic sequence and drastically improves overall process efficiency. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to optimize supply chains for electronic chemical manufacturing while adhering to stricter environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of p-acetoxystyrene has been plagued by complex multi-step sequences that start from p-hydroxyacetophenone. These conventional routes typically involve acetylation followed by catalytic hydrogenation and a final dehydration step. A major drawback of this approach is the reliance on strong alkalis during the dehydration phase to achieve acceptable conversion rates. The use of such harsh reagents not only complicates the workup procedure but also generates substantial amounts of hazardous waste, posing significant environmental compliance challenges. Furthermore, literature data indicates that these traditional methods often suffer from mediocre total yields, frequently hovering around 68.5 percent, which translates to higher raw material consumption and increased production costs. The necessity for rigorous purification to remove side products generated under strong alkaline conditions further erodes the economic viability of these legacy processes.
The Novel Approach
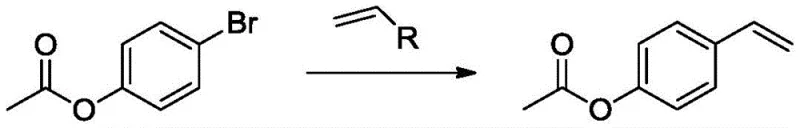
In stark contrast, the methodology disclosed in the patent utilizes a convergent synthesis strategy centered on a palladium-catalyzed coupling reaction. The process begins with the acetylation of p-bromophenol to generate 4-acetoxybromobenzene, which is then directly coupled with a vinyl boron compound. This route elegantly bypasses the need for harsh dehydration steps and strong alkalis, thereby minimizing waste generation and simplifying downstream processing. The reaction conditions are notably mild, typically operating between 50-100°C, which enhances safety and reduces energy consumption. By employing readily available starting materials like p-bromophenol and vinyl boronic acids, the method achieves impressive yields of up to 91 percent with high purity. This shift not only improves the atom economy of the synthesis but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for sustainable industrial scale-up.
Mechanistic Insights into Palladium-Catalyzed Vinyl Coupling
The core of this innovative synthesis is a palladium-catalyzed cross-coupling reaction, chemically akin to the Suzuki-Miyaura protocol, although the patent text broadly references Heck-type conditions. The mechanism involves the oxidative addition of the aryl bromide (4-acetoxybromobenzene) to the active palladium(0) species, forming an aryl-palladium intermediate. This is followed by transmetallation with the vinyl boron species, a step that is facilitated by the presence of a base such as sodium phosphate or potassium carbonate. The choice of base is critical, as it activates the boron reagent for transfer without degrading the sensitive acetate protecting group. Finally, reductive elimination releases the desired p-acetoxystyrene product and regenerates the palladium catalyst for the next cycle. The versatility of this mechanism is highlighted by its compatibility with various vinyl boron reagents, allowing manufacturers to select the most cost-effective or stable boron source for their specific supply chain needs.
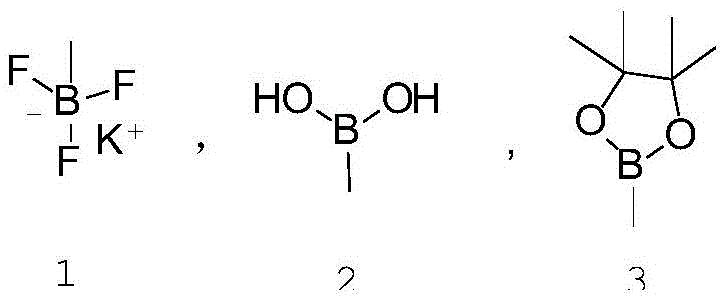
Impurity control is another critical aspect where this mechanistic approach excels. Traditional dehydration routes often lead to polymerization of the styrene double bond or incomplete elimination, resulting in difficult-to-remove impurities. In the coupling method, the formation of the vinyl group occurs in the final step under controlled thermal conditions, minimizing the risk of premature polymerization. The use of specific ligands on the palladium catalyst, such as triphenylphosphine or ferrocene-based ligands, further enhances selectivity, ensuring that the coupling occurs exclusively at the bromine position without affecting the ester moiety. This high chemoselectivity results in a cleaner crude product profile, which significantly reduces the burden on purification units like distillation columns. For quality control teams, this means a more consistent impurity profile and easier validation of the final API intermediate or electronic material.
How to Synthesize p-Acetoxystyrene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into two distinct stages: the protection of the phenol group via acetylation and the subsequent carbon-carbon bond formation. The acetylation step can be performed using either acetyl chloride or acetic anhydride, offering flexibility depending on reagent availability. Following the isolation of the intermediate 4-acetoxybromobenzene, the coupling reaction is conducted in common organic solvents such as ethanol, dichloromethane, or dimethylformamide. The detailed standardized synthesis steps below outline the specific molar ratios, temperature profiles, and workup procedures required to replicate the high yields reported in the examples.
- Acetylate p-bromophenol using acetyl chloride or acetic anhydride in the presence of a base to form 4-acetoxybromobenzene.
- React the resulting 4-acetoxybromobenzene with a vinyl boron compound (such as vinylboronic acid or potassium vinyltrifluoroborate) using a palladium catalyst.
- Maintain reaction temperatures between 50-100°C in a suitable organic solvent to achieve high conversion and yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for high-pressure hydrogenation equipment and corrosive strong alkali handling facilities, capital investment requirements for new production lines are significantly lowered. Furthermore, the mild reaction conditions reduce the energy load on the plant, contributing to lower utility costs per kilogram of product. The robustness of the reaction also implies fewer batch failures and less downtime, ensuring a more reliable supply of this critical photoresist intermediate to downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the high atom efficiency of the coupling reaction, leads to substantial cost savings. Unlike traditional methods that suffer from yield losses during multiple purification steps, this one-pot coupling approach maximizes the conversion of raw materials into the final product. The ability to use diverse vinyl boron sources allows procurement teams to leverage market fluctuations and source the most economical boron reagent at any given time. Additionally, the reduced generation of chemical waste lowers the costs associated with waste treatment and disposal, further enhancing the overall profit margin of the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically p-bromophenol and various vinyl boron compounds, are commodity chemicals with well-established global supply chains. This reduces the risk of raw material shortages that can plague more exotic synthetic routes. The stability of the intermediate 4-acetoxybromobenzene also allows for potential stockpiling, decoupling the two reaction steps and providing greater flexibility in production scheduling. For supply chain planners, this means shorter lead times and the ability to respond more agilely to sudden spikes in demand from the semiconductor or coating industries without compromising on quality.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, this method offers a clear path to compliance. The avoidance of strong alkalis and the reduction of solvent usage due to higher concentrations and yields make the process inherently greener. Scaling this reaction from 100 kgs to multi-ton batches is straightforward because the exotherm is manageable and the reaction does not require extreme temperatures or pressures. This ease of scale-up ensures that manufacturers can rapidly expand capacity to meet the growing demand for advanced photoresists driven by the expansion of the integrated circuit industry, all while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production facilities or for procurement officers negotiating supply contracts.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The new method avoids the use of strong alkalis required in traditional dehydration steps, significantly reducing three-waste generation. It also achieves higher yields (up to 91%) compared to the 68.5% typical of older multi-step reductions.
Q: Which vinyl boron reagents are compatible with this coupling process?
A: The process is versatile and supports various vinyl boron species, including vinylboronic acid, potassium vinyltrifluoroborate, and pinacol vinylboronate, allowing for flexibility in raw material sourcing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (50-100°C) and utilize standard organic solvents like ethanol or dichloromethane, making it highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Acetoxystyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity p-acetoxystyrene plays in the advancement of electronic materials and photoresist technologies. Our team of expert chemists has extensively analyzed the pathway described in CN112409176A and possesses the capability to execute this palladium-catalyzed coupling with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for semiconductor applications.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value for your organization. Let us be your partner in delivering high-performance chemical solutions for the next generation of electronic devices.
