Advanced Manufacturing of p-Acetoxystyrene via Base-Catalyzed Elimination for High-Purity Photoresists
Advanced Manufacturing of p-Acetoxystyrene via Base-Catalyzed Elimination for High-Purity Photoresists
The semiconductor and integrated circuit industries rely heavily on advanced photoresist materials, where poly-p-hydroxystyrene serves as a critical backbone component. The monomer p-acetoxystyrene is the essential precursor for synthesizing this polymer, yet its production has historically faced challenges regarding yield stability and impurity control. Patent CN110655462A introduces a transformative three-step synthetic route that fundamentally shifts the paradigm from traditional acid-catalyzed dehydration to a robust base-catalyzed elimination strategy. This innovation addresses the longstanding issue of double-bond instability during synthesis, offering a pathway to high-purity intermediates essential for next-generation chip manufacturing. By starting from basic raw materials like p-hydroxyacetophenone and employing controlled acetylation and hydrogenation followed by alkaline elimination, the process ensures that the sensitive vinyl group remains intact throughout the reaction lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of styrene derivatives like p-acetoxystyrene has relied heavily on acid-catalyzed thermal elimination or complex multi-step sequences involving Wittig reactions. Traditional acid-catalyzed methods, often utilizing catalysts like potassium bisulfate under high vacuum and elevated temperatures, suffer from significant drawbacks including uncontrollable side reactions. The acidic environment frequently triggers flash side reactions such as double-bond coupling or premature polymerization, leading to substantial product loss and difficult downstream purification. Furthermore, these conventional routes often require expensive reagents or complicated decarboxylation steps that increase the overall cost of goods and complicate the supply chain. The inherent instability of the product under acidic conditions means that yields can vary drastically upon scale-up, posing a severe risk for procurement managers seeking consistent supply continuity for electronic chemical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN110655462A utilizes a mild yet effective base-catalyzed elimination mechanism that preserves the integrity of the vinyl double bond. By shifting the reaction environment to alkaline conditions using inorganic bases like sodium hydroxide or potassium hydroxide in polar aprotic solvents, the process effectively suppresses the acid-catalyzed side reactions that plague older methods. This approach allows the generated double bonds to exist stably for long periods within the reaction system, enabling complete conversion without the risk of runaway polymerization. The result is a streamlined three-step process that starts from readily available p-hydroxyacetophenone, proceeds through a stable acetoxy intermediate, and concludes with a high-yield elimination step. This structural shift in synthetic strategy not only improves the final purity but also drastically simplifies the operational parameters required for commercial production.
Mechanistic Insights into Base-Catalyzed Elimination and Hydrogenation
The core of this synthetic innovation lies in the precise control of reaction conditions across three distinct chemical transformations, beginning with the protection of the phenolic hydroxyl group. In the first step, p-hydroxyacetophenone undergoes acetylation using reagents such as acetyl chloride or acetic anhydride in the presence of an acid-binding agent. The use of bases like triethylamine or sodium carbonate neutralizes the generated acid, driving the equilibrium forward while preventing degradation of the substrate. This step is typically conducted in solvents such as ethyl acetate or dichloromethane at moderate temperatures ranging from 20°C to 50°C, ensuring high conversion rates without thermal stress.
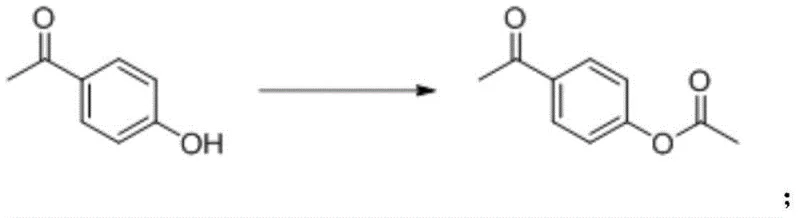
Following acetylation, the ketone functionality is selectively reduced to a secondary alcohol through catalytic hydrogenation, a critical step that sets the stage for the final elimination. This reduction employs heterogeneous catalysts such as palladium on carbon or Raney nickel under a hydrogen atmosphere at pressures between 5 and 10 bar. The reaction is carefully managed in solvents like toluene or methanol at temperatures between 20°C and 50°C to prevent over-reduction or deacetylation. The resulting 1-(4-acetoxyphenyl)ethanol intermediate is isolated with high purity, serving as the direct precursor for the alkene formation.
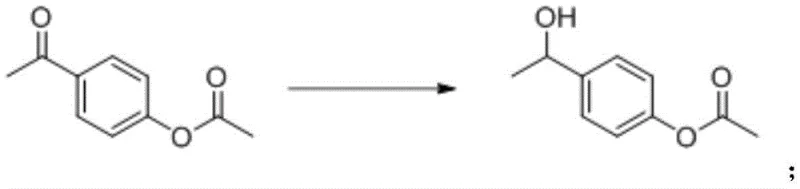
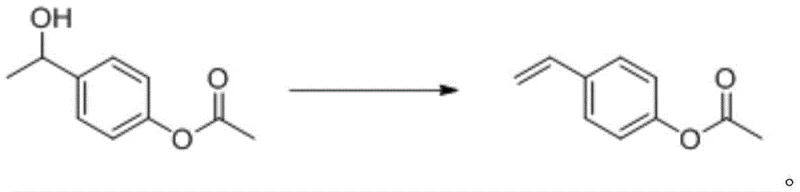
The final and most critical transformation involves the base-catalyzed elimination of water from the alcohol intermediate to form the target styrene derivative. Unlike acid-catalyzed dehydration, this alkaline elimination uses strong inorganic bases such as NaOH or KOH dissolved in polar aprotic solvents like DMAC or DMF. Operating at temperatures between 100°C and 130°C, this step facilitates the removal of the beta-hydrogen without protonating the double bond, thereby avoiding cationic polymerization pathways. The stability of the product under these basic conditions is a key mechanistic advantage, allowing the reaction to proceed to completion with minimal formation of oligomeric byproducts.
How to Synthesize p-Acetoxystyrene Efficiently
The synthesis of p-acetoxystyrene via this patented route requires strict adherence to the specified reaction parameters to maximize yield and minimize impurities. The process begins with the acetylation of the starting material, followed by a controlled hydrogenation step, and concludes with the thermal elimination in a basic medium. Each step demands precise stoichiometric control, particularly the ratio of base to substrate in the final elimination step, which should ideally range from 1:1 to 1:3 to ensure complete conversion. Detailed standard operating procedures regarding solvent recovery, catalyst filtration, and crystallization protocols are essential for replicating the high purity levels demonstrated in the patent examples.
- Acetylate p-hydroxyacetophenone using acetyl chloride or anhydride with a base scavenger in organic solvent to form p-acetoxyacetophenone.
- Hydrogenate p-acetoxyacetophenone using Pd/C or Raney Nickel catalyst under 5-10 bar hydrogen pressure to obtain 1-(4-acetoxyphenyl)ethanol.
- Perform base-catalyzed elimination on the alcohol intermediate using NaOH or KOH in polar aprotic solvent at 100-130°C to yield p-acetoxystyrene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-catalyzed synthesis route offers profound advantages in terms of cost structure and supply reliability. By eliminating the need for expensive phosphorus ylides used in Wittig reactions or the complex vacuum distillation setups required for acid-catalyzed dehydration, the overall capital expenditure and operational complexity are significantly reduced. The use of commodity chemicals such as sodium hydroxide, acetyl chloride, and common organic solvents ensures that raw material sourcing remains stable and unaffected by niche market fluctuations. Furthermore, the robustness of the reaction system means that production schedules are less likely to be disrupted by batch failures or unpredictable yield drops, providing a more dependable supply chain for critical electronic materials.
- Cost Reduction in Manufacturing: The transition to a base-catalyzed elimination process removes the necessity for costly acid catalysts and the associated neutralization waste streams, leading to substantial cost savings in waste treatment. Additionally, the higher selectivity of the reaction reduces the burden on downstream purification units, lowering energy consumption and solvent usage per kilogram of product. The ability to use standard stainless steel reactors without specialized acid-resistant linings further contributes to a lower total cost of ownership for the manufacturing facility.
- Enhanced Supply Chain Reliability: Since the process relies on widely available bulk chemicals rather than specialized reagents, the risk of supply disruption is drastically minimized. The stability of the intermediate and final product under the reaction conditions allows for flexible batch scheduling and inventory management, ensuring that delivery timelines can be met consistently. This reliability is crucial for downstream customers in the semiconductor industry who require uninterrupted access to high-purity monomers for their photoresist formulations.
- Scalability and Environmental Compliance: The method exhibits a small amplification effect, meaning that yields observed in the laboratory are maintained during commercial scale-up, reducing the financial risk of technology transfer. The avoidance of heavy metal catalysts in the final step and the use of recyclable solvents align with increasingly stringent environmental regulations, simplifying the permitting process for new production lines. This environmental compatibility enhances the long-term viability of the supply source and reduces the regulatory burden on the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on reaction mechanisms and process advantages. Understanding these details is vital for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: Why is base-catalyzed elimination preferred over acid catalysis for p-acetoxystyrene?
A: Traditional acid-catalyzed dehydration often leads to uncontrolled side reactions, such as double-bond polymerization or coupling, resulting in low yields and difficult purification. The novel base-catalyzed method avoids these acidic side reactions, ensuring the generated double bond remains stable for extended periods within the reaction system.
Q: What catalysts are suitable for the reduction step in this synthesis?
A: The process utilizes heterogeneous hydrogenation catalysts, specifically palladium carbon (Pd/C) or nickel-based catalysts like Raney Nickel. These catalysts operate effectively under moderate hydrogen pressures of 5 to 10 bar and temperatures between 20°C and 50°C, offering high selectivity for the alcohol intermediate.
Q: How does this method improve scalability for electronic chemical manufacturing?
A: The method demonstrates a small amplification effect, meaning reaction yields remain consistent when scaling from laboratory to industrial production. The use of common solvents like DMAC and inorganic bases simplifies post-treatment and waste management, facilitating reliable commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Acetoxystyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity p-acetoxystyrene plays in the advancement of semiconductor lithography and electronic materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade chemicals, guaranteeing that every batch performs consistently in your photoresist applications.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can enhance your supply chain resilience and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next generation of electronic material development.
