Revolutionizing p-Acetoxystyrene Production: A Scalable Route for High-Purity Pharmaceutical Intermediates
Revolutionizing p-Acetoxystyrene Production: A Scalable Route for High-Purity Pharmaceutical Intermediates
In the rapidly evolving landscape of fine chemical synthesis, the demand for high-purity monomers and intermediates continues to surge, driven by the pharmaceutical and advanced materials sectors. Patent CN102795999A introduces a groundbreaking preparation method for p-acetoxystyrene (4-ASM), a critical building block known for its utility in polymer chemistry and drug discovery. This technology addresses long-standing inefficiencies in traditional synthesis pathways by establishing a robust three-step sequence: acetylation, catalytic hydrogenation, and dehydration. By leveraging common starting materials like p-hydroxyacetophenone and avoiding hazardous reagents, this process achieves a remarkable total yield of 72.0% and product purity exceeding 99.5%. For industry leaders seeking a reliable p-acetoxystyrene supplier, understanding the mechanistic depth and commercial viability of this patented route is essential for securing supply chains and optimizing production costs.
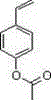
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-acetoxystyrene has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Early methodologies, such as those described by Sovish and later adapted by International Flavors & Fragrances, relied heavily on the condensation of p-hydroxybenzaldehyde with malonic acid or the use of Wittig and Grignard reagents. As illustrated in prior art pathways, these reactions often suffer from abysmal yields, typically ranging between 12% and 42%, primarily due to the formation of complex byproduct mixtures and the sensitivity of the reagents to moisture and oxygen. Furthermore, alternative routes involving the reaction of p-acetoxybenzaldehyde with dibromomethane in the presence of zinc and active chlorine, while offering moderate yields around 80%, introduce severe environmental and safety liabilities. The use of dibromomethane is not only costly but also highly toxic, creating substantial waste disposal challenges that conflict with modern green chemistry principles. Additionally, gas-phase catalytic dehydrogenation of p-ethylphenol requires extreme temperatures exceeding 500°C, necessitating specialized, energy-intensive equipment that is prone to coking and rapid catalyst deactivation, thereby limiting operational continuity.
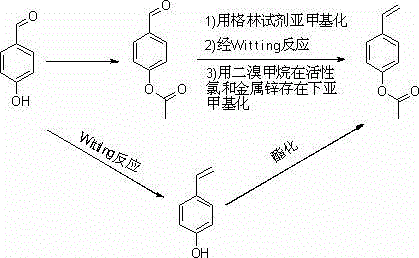
The Novel Approach
The patented methodology presented in CN102795999A offers a transformative solution by shifting the synthetic paradigm towards a liquid-phase, catalytic cascade that prioritizes safety and efficiency. Instead of relying on stoichiometric organometallic reagents or extreme thermal conditions, this novel approach utilizes p-hydroxyacetophenone as a stable and commercially abundant feedstock. The process initiates with a high-efficiency acetylation to protect the phenolic hydroxyl group, followed by a selective catalytic hydrogenation of the ketone moiety to a benzylic alcohol. This intermediate is then subjected to a controlled acid-catalyzed dehydration in the presence of specific polymerization inhibitors. This strategic sequence bypasses the toxicity associated with halogenated methanes and eliminates the need for expensive phosphorus ylides. By operating under milder conditions and utilizing recoverable heterogeneous catalysts, this route significantly enhances the cost reduction in pharmaceutical intermediates manufacturing while ensuring a consistent supply of high-purity p-acetoxystyrene suitable for sensitive downstream applications.
Mechanistic Insights into Catalytic Hydrogenation and Dehydration
The core of this synthesis lies in the precise control of the hydrogenation and dehydration steps, which dictate the overall impurity profile and yield. In the hydrogenation stage, p-acetoxyacetophenone is dissolved in a solvent such as methanol, ethanol, or ethyl acetate and exposed to hydrogen gas under the influence of a transition metal catalyst, such as Raney Nickel, Palladium on Carbon, or Platinum Black. The mechanism involves the adsorption of the carbonyl group onto the metal surface, where it undergoes sequential addition of hydrogen atoms to form the corresponding benzylic alcohol, p-acetoxybenzyl alcohol. The choice of catalyst and solvent polarity is critical; for instance, using a nickel catalyst at moderate pressures ensures high chemoselectivity, preventing the unwanted hydrogenolysis of the acetoxy ester group or the aromatic ring. Following purification via distillation, the alcohol intermediate enters the dehydration phase, where the presence of a strong acid catalyst like sulfuric acid or phosphoric acid facilitates the elimination of water. Crucially, this step generates a reactive vinyl group that is prone to spontaneous polymerization.
To counteract this inherent instability, the process incorporates a sophisticated system of polymerization inhibitors, including phenolic compounds like hydroquinone, quinones, or stable free radicals such as TEMPO derivatives. These inhibitors function by scavenging free radicals generated during the thermal stress of dehydration, effectively terminating chain propagation before high molecular weight polymers can form. The reaction is typically conducted at temperatures between 135°C and 200°C, a range optimized to drive the elimination equilibrium forward without triggering excessive thermal degradation. The resulting crude product is then subjected to rigorous fractional distillation under reduced pressure to separate the target monomer from unreacted alcohol and heavy ends. This meticulous attention to impurity control mechanisms ensures that the final commercial scale-up of complex monomers meets stringent specifications, with gas chromatography analysis confirming purity levels consistently above 99.5%, a benchmark essential for electronic and pharmaceutical grade materials.
How to Synthesize p-Acetoxystyrene Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and safety. The process begins with the acetylation of p-hydroxyacetophenone using acetic anhydride, typically at temperatures between 100°C and 140°C, followed by vacuum distillation to isolate the ketone intermediate. The subsequent hydrogenation step demands precise control over hydrogen pressure and catalyst loading to ensure complete conversion while maintaining the integrity of the ester functionality. Finally, the dehydration step must be managed with adequate inhibitor loading to prevent reactor fouling due to polymerization. For detailed operational protocols, safety data sheets, and specific catalyst recommendations, please refer to the standardized synthesis guide below which outlines the critical process parameters validated across multiple pilot scales.
- Acetylate p-hydroxyacetophenone with acetic anhydride to form p-acetoxyacetophenone.
- Perform catalytic hydrogenation on p-acetoxyacetophenone using Ni, Pd, or Pt catalysts to obtain p-acetoxybenzyl alcohol.
- Dehydrate p-acetoxybenzyl alcohol using an acid catalyst and polymerization inhibitor to yield p-acetoxystyrene.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond simple yield metrics. Traditional methods relying on Wittig reagents or dibromomethane create volatile cost structures due to the fluctuating prices of specialized organophosphorus compounds and the regulatory burdens associated with handling toxic halogenated solvents. By transitioning to a route based on p-hydroxyacetophenone and acetic anhydride, manufacturers can leverage a supply chain built on commodity chemicals that are produced in massive global volumes, ensuring price stability and availability. This shift fundamentally alters the cost basis of production, allowing for cost reduction in pharmaceutical intermediates manufacturing through the elimination of expensive stoichiometric reagents and the simplification of waste treatment protocols. Furthermore, the avoidance of highly toxic substances reduces the regulatory compliance overhead and insurance costs associated with hazardous material storage and transport.
- Cost Reduction in Manufacturing: The elimination of expensive Grignard or Wittig reagents, which are often single-use and generate significant phosphine oxide waste, drastically lowers the variable cost per kilogram of the final product. Additionally, the use of heterogeneous catalysts like Raney Nickel or Pd/C allows for potential recovery and reuse, further amortizing capital expenditure over larger production batches. The process operates in the liquid phase at moderate temperatures, avoiding the extreme energy consumption required for gas-phase dehydrogenation processes that operate above 500°C. This energy efficiency translates directly into lower utility costs and a reduced carbon footprint, aligning with corporate sustainability goals while enhancing overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is significantly more robust compared to niche reagents required for alternative pathways. p-Hydroxyacetophenone and acetic anhydride are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks that often plague specialized chemical supply chains. The simplified reaction sequence, consisting of only three main unit operations, reduces the complexity of the manufacturing schedule and minimizes the potential for equipment downtime caused by fouling or catalyst poisoning. This operational simplicity ensures reducing lead time for high-purity specialty chemicals, enabling faster response to market demand fluctuations and more reliable delivery commitments to downstream customers in the API and polymer sectors.
- Scalability and Environmental Compliance: The liquid-phase nature of this process makes it inherently easier to scale from pilot plant to multi-ton commercial production compared to gas-phase technologies that require specialized high-temperature reactors. The absence of toxic dibromomethane and heavy metal waste streams simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations such as REACH and TSCA. The high selectivity of the catalytic steps minimizes the formation of difficult-to-separate byproducts, reducing the load on purification units and solvent recovery systems. This environmental compatibility not only future-proofs the manufacturing asset against regulatory changes but also enhances the brand reputation of the supplier as a responsible partner in the global value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of p-acetoxystyrene using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing supplier qualifications. We encourage stakeholders to review these points to gain a comprehensive understanding of how this synthesis route compares to legacy technologies in terms of performance and reliability.
Q: What are the advantages of this synthesis route over the Wittig reaction?
A: Unlike the Wittig reaction which suffers from low yields (12-42%) and expensive reagents, this patented route utilizes catalytic hydrogenation and dehydration, achieving a total yield of 72.0% with significantly lower raw material costs.
Q: How is polymerization prevented during the dehydration step?
A: The process employs specific polymerization inhibitors such as hydroquinone, phenothiazine, or stable free radicals during the dehydration reaction at 135-200°C to ensure the stability of the vinyl group.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method avoids extreme conditions like the 500°C required for gas-phase dehydrogenation and eliminates toxic reagents like dibromomethane, making it safer and more feasible for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Acetoxystyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patented processes like CN102795999A are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch of p-acetoxystyrene meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality assurance means that clients receive not just a chemical product, but a guaranteed level of performance that supports their own downstream synthesis and formulation efforts.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain resilience and cost efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your long-term strategic goals. Let us be your trusted partner in delivering high-performance chemical solutions that drive innovation and growth in your organization.
