Scalable Production of Barosavir Intermediates via Safe TEMPO-Catalyzed Oxidation and Benzylation
Scalable Production of Barosavir Intermediates via Safe TEMPO-Catalyzed Oxidation and Benzylation
The rapid global demand for effective antiviral therapies has placed significant pressure on the supply chains of critical pharmaceutical ingredients, particularly for next-generation influenza treatments like Barosavir (Baloxavir marboxil). As a cap-dependent endonuclease inhibitor, Barosavir represents a paradigm shift in influenza management, necessitating robust and scalable synthetic routes for its key building blocks. Patent CN112159380B, published in early 2022, introduces a groundbreaking methodology for preparing essential Barosavir intermediates, specifically targeting the synthesis of 3-benzyloxy-4-oxo-4H-pyran-2-formic acid. This technical disclosure addresses long-standing industry pain points regarding toxicity, operational complexity, and thermal safety, offering a viable pathway for commercial scale-up of complex heterocycles. By leveraging a TEMPO-catalyzed oxidation strategy combined with mild benzylation conditions, this process eliminates the need for hazardous reagents such as selenium dioxide and methylsulfonyl chloride, thereby aligning with modern green chemistry principles while ensuring high purity standards required for active pharmaceutical ingredient (API) production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyranone-based intermediates for antiviral drugs has been plagued by severe safety hazards and operational inefficiencies that hinder large-scale manufacturing. Prior art, such as the route described in the Journal of the American Chemical Society (2006), relies heavily on selenium dioxide (SeO2) for oxidation steps, which mandates extremely harsh reaction temperatures reaching 155°C. Such high-temperature conditions are not only energy-intensive but also difficult to maintain safely using standard steam heating, often requiring dangerous high-temperature oil baths that increase the risk of thermal runaway. Furthermore, selenium dioxide is a highly toxic, controlled chemical known to cause severe respiratory issues, including chemical bronchitis and pulmonary edema, posing unacceptable occupational health risks in an industrial setting. Another documented approach, disclosed in WO2010/11816a1, utilizes methylsulfonyl chloride and expensive ruthenium catalysts, introducing both significant environmental pollution burdens and exorbitant raw material costs that render the process economically unfeasible for generic production.
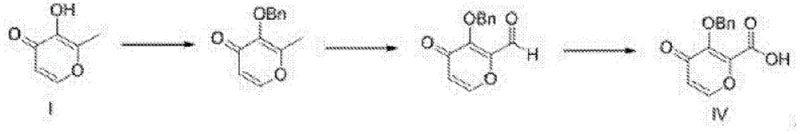
The Novel Approach
In stark contrast to these hazardous legacy methods, the novel process outlined in patent CN112159380B employs a streamlined three-step sequence that prioritizes safety and cost-efficiency without compromising yield. The core innovation lies in the substitution of toxic oxidants with a TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) catalytic system, which facilitates the conversion of the starting alcohol to the corresponding carboxylic acid under remarkably mild conditions. The entire synthetic pathway is conducted at temperatures strictly controlled below 60°C, effectively eliminating the need for high-temperature heating equipment and drastically reducing energy consumption. By utilizing cheap and easily obtained starting materials like 3-hydroxy-2-(hydroxymethyl)-4H-pyran-4-one, the process ensures a stable supply chain foundation. The reaction scheme demonstrates a logical progression from oxidation to benzyl protection and finally hydrolysis, achieving high conversion rates while avoiding the generation of hazardous waste streams associated with heavy metals or sulfonyl chlorides.
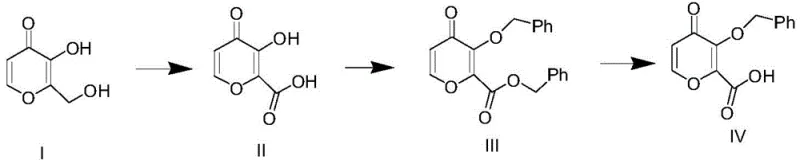
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Benzylation
The mechanistic elegance of this process is rooted in the selective oxidation capabilities of the TEMPO radical system, which operates efficiently in biphasic or mixed solvent systems. In the initial oxidation step, the primary alcohol of the starting material is converted to a carboxylic acid using sodium hypochlorite as the terminal oxidant, mediated by catalytic amounts of TEMPO and potassium bromide. Crucially, the patent specifies the use of aprotic solvents such as acetonitrile or dichloromethane in this stage to prevent the competitive oxidation of protic solvents like methanol, which would otherwise consume the oxidant and generate acidic impurities that complicate downstream purification. The reaction is maintained at 0-10°C, a temperature range that suppresses side reactions and ensures the stability of the sensitive pyranone ring structure. Following oxidation, the benzylation step utilizes benzyl chloride in the presence of a strong base like sodium hydroxide or potassium carbonate. The choice of base is critical; strong alkalinity is required to deprotonate the phenolic hydroxyl group effectively, ensuring complete conversion to the benzyl ether without requiring prolonged reaction times or excessive heating that could degrade the product.
Impurity control is meticulously managed through solvent selection and pH regulation throughout the synthesis. During the workup of the oxidation step, the aqueous phase is carefully acidified to pH 1-2 using hydrochloric acid to precipitate the crude acid, allowing for the removal of organic-soluble impurities via dichloromethane extraction. This liquid-liquid extraction strategy is far superior to traditional chromatographic purification for bulk manufacturing, as it significantly reduces solvent usage and processing time. In the subsequent hydrolysis step, the ester moiety is cleaved under mild alkaline conditions at room temperature, avoiding the harsh acidic or basic conditions that might open the lactone ring or cause decarboxylation. The final product is isolated by adjusting the pH to induce precipitation, followed by simple filtration and drying. This sequence of unit operations—oxidation, extraction, benzylation, and hydrolysis—is designed to maximize throughput while maintaining a tight impurity profile, which is essential for meeting the stringent quality specifications of pharmaceutical intermediates.
How to Synthesize 3-benzyloxy-4-oxo-4H-pyran-2-formic acid Efficiently
The practical implementation of this synthesis requires precise control over stoichiometry and temperature to replicate the high yields reported in the patent examples. The process begins with the dissolution of the starting pyranone in a mixture of acetonitrile and water, followed by the addition of the TEMPO catalyst and bromide promoter. Sodium hypochlorite is then added dropwise while maintaining the internal temperature between 0-10°C to manage the exotherm and ensure selective oxidation. Once the oxidation is complete, the intermediate acid is isolated and subjected to benzylation using benzyl chloride and a base in a methanol-water system at 45-60°C. The final step involves hydrolyzing the ethyl ester (if formed) or directly processing the protected acid to the final target using sodium hydroxide at room temperature. Detailed standardized operating procedures for each unit operation, including specific quenching and isolation protocols, are essential for technology transfer.
- Oxidize 3-hydroxy-2-(hydroxymethyl)-4H-pyran-4-one using TEMPO/NaOCl system at 0-10°C to form the carboxylic acid intermediate.
- Perform benzylation on the acid intermediate using benzyl chloride and a base at 45-60°C to protect the hydroxyl group.
- Conduct hydrolysis of the ester group using an alkaline reagent at room temperature to yield the final target acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple laboratory yield improvements. The elimination of selenium dioxide and methylsulfonyl chloride removes the need for specialized handling protocols, expensive personal protective equipment, and complex waste disposal procedures associated with highly toxic and controlled substances. This simplification of the safety profile directly translates to substantial cost savings in operational overhead and regulatory compliance, as facilities no longer require dedicated containment systems for hazardous reagents. Furthermore, the reliance on commodity chemicals such as sodium hypochlorite, benzyl chloride, and common organic solvents ensures a resilient supply chain that is less susceptible to market volatility or geopolitical disruptions affecting specialty reagent availability. The ability to source raw materials from multiple vendors reduces single-source dependency risks, enhancing the overall reliability of the manufacturing schedule.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive noble metal catalysts, such as ruthenium, with inexpensive organocatalysts like TEMPO, which can be used in low loading ratios. Additionally, the avoidance of high-temperature reactions (155°C) significantly lowers energy consumption costs, as standard cooling water and low-pressure steam suffice for temperature control instead of high-temperature oil baths. The simplified workup procedures, which rely on pH-adjusted precipitation rather than column chromatography, drastically reduce solvent consumption and processing time, leading to a lower cost of goods sold (COGS) per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: By utilizing reagents that are widely available in the global chemical market, manufacturers can secure long-term supply contracts with favorable pricing, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, specifically the tolerance for slight variations in temperature and mixing within the 0-60°C range, ensures consistent batch-to-batch quality even when scaling up from pilot plants to multi-ton reactors. This process stability minimizes the likelihood of batch failures, thereby guaranteeing a steady flow of high-purity Barosavir intermediates to downstream API manufacturers and protecting the continuity of the finished drug supply.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction steps that generate minimal hazardous waste and avoid the formation of persistent organic pollutants. The absence of heavy metals simplifies the purification of the final product, reducing the burden on analytical quality control labs to test for residual metal contaminants. From an environmental perspective, the use of aqueous workups and the recycling of common solvents align with green chemistry initiatives, potentially qualifying the manufacturing site for environmental incentives and reducing the carbon footprint of the production facility. This sustainability profile is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional approaches in terms of safety, efficiency, and scalability. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this process for commercial production.
Q: Why is the TEMPO oxidation method preferred over Selenium Dioxide oxidation for Barosavir intermediates?
A: The TEMPO oxidation method operates at low temperatures (0-10°C) and avoids highly toxic selenium dioxide, which requires dangerous high-temperature conditions (155°C) and poses severe health risks like chemical pneumonia.
Q: How does this new process improve supply chain reliability compared to previous methods?
A: By eliminating controlled chemicals like methylsulfonyl chloride and expensive ruthenium catalysts, the process relies on common, easily purchased reagents, ensuring consistent raw material availability and reducing regulatory hurdles.
Q: What are the safety advantages of keeping reaction temperatures below 60°C?
A: Maintaining temperatures below 60°C prevents thermal runaway risks associated with high-temperature oil baths, simplifies cooling requirements, and significantly enhances the overall safety coefficient for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Barosavir Intermediate Supplier
As the pharmaceutical industry continues to evolve towards safer and more sustainable manufacturing practices, the ability to execute complex synthetic routes with precision and reliability becomes a key differentiator for CDMO partners. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific temperature controls and solvent management required for TEMPO-catalyzed reactions, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We understand that the production of antiviral intermediates demands not just chemical expertise but also a commitment to supply security and regulatory compliance, which forms the cornerstone of our service offering to global pharmaceutical clients.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to drive value. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements and regional logistics. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your competitive position in the antiviral market while ensuring a reliable supply of high-quality intermediates.
