Advanced Manufacturing of Chiral Barosavir Intermediates via Early-Stage Resolution
Advanced Manufacturing of Chiral Barosavir Intermediates via Early-Stage Resolution
The global demand for effective antiviral therapeutics has intensified the search for robust and scalable synthetic routes for key active pharmaceutical ingredients (APIs). Patent CN112266381B, published in early 2022, introduces a transformative methodology for the synthesis of a critical Barosavir intermediate. Barosavir, a novel cap-dependent endonuclease inhibitor, represents a significant leap forward in influenza treatment, offering a mechanism of action distinct from traditional neuraminidase inhibitors. For pharmaceutical manufacturers and procurement strategists, the ability to access high-purity intermediates through a streamlined, cost-effective process is paramount. This technical insight report analyzes the proprietary synthesis route disclosed in the patent, highlighting its potential to redefine supply chain reliability and cost structures in the antiviral sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of morpholine-based antiviral intermediates has been plagued by inefficiencies that hinder large-scale commercial viability. Prior art, such as the methods described in WO 2017221869A1, typically employs a strategy where the morpholine ring is constructed in the final steps, necessitating chiral resolution at a very late stage. This approach is inherently wasteful; resolving a complex, high-molecular-weight final intermediate means that nearly half of the synthesized material—the unwanted enantiomer—is discarded after significant value has already been added through multiple reaction steps. Furthermore, alternative routes like those found in CN110105372 rely on the reduction of oxo-morpholine derivatives using sodium borohydride.
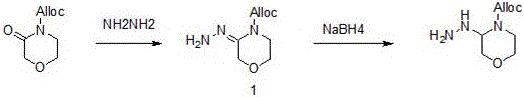
While chemically feasible, the reliance on substantial quantities of sodium borohydride introduces severe safety hazards and cost burdens associated with the handling and disposal of reactive hydride species. Additionally, other documented approaches, such as the one in CN 111018803A, utilize expensive starting materials like 3-(benzyloxy)-4-oxo-4H-pyran-2-carboxylic acid and require specialized hydrogenation equipment. These conventional pathways often mandate column chromatography for purification, a technique that is notoriously difficult to scale and economically prohibitive for multi-kilogram or ton-scale production. The cumulative effect of these limitations is a fragile supply chain characterized by high production costs, extended lead times, and significant environmental waste generation.
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN112266381B proposes a paradigm shift by moving the chiral resolution step to an earlier stage of the synthesis. The novel route begins with the condensation of inexpensive and readily available morpholine with allyl chloroformate. This initial protection step sets the stage for a series of highly controlled transformations. The core innovation lies in the strategic functionalization of the morpholine ring followed by the introduction of the hydrazine moiety prior to the final coupling. By resolving the chirality at the hydrazino-morpholine stage (Compound 5 to Compound 6), the process ensures that only the desired enantiomer proceeds to the final, more resource-intensive coupling reactions.
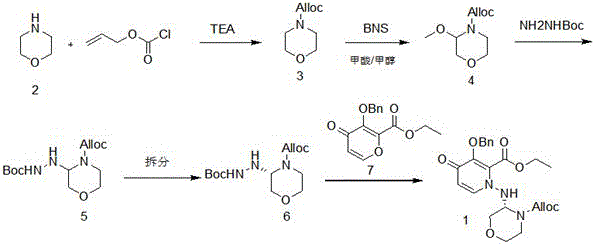
This "early resolution" strategy drastically improves the overall atom economy and reduces the cost of goods sold (COGS). The method avoids the use of dangerous reducing agents and eliminates the need for column chromatography, relying instead on crystallization and standard extraction techniques. The reaction conditions are notably mild, utilizing common solvents like methanol, ethanol, and ethyl acetate, which simplifies solvent recovery and waste management. For a reliable pharmaceutical intermediate supplier, this route offers a clear pathway to consistent quality and supply continuity, decoupling production from the bottlenecks associated with complex purification and hazardous reagent handling.
Mechanistic Insights into Lewis Acid-Catalyzed Hydrazine Coupling
The chemical elegance of this synthesis is best exemplified in the coupling of the methoxylated morpholine derivative (Compound 4) with Boc-hydrazine. This transformation is facilitated by the use of a Lewis acid catalyst, specifically tin tetrachloride (SnCl4), although the patent notes that titanium tetrachloride, aluminum trichloride, or zinc chloride can also serve this function. The mechanism likely involves the coordination of the Lewis acid to the electrophilic center of the morpholine derivative, thereby activating it for nucleophilic attack by the hydrazine nitrogen. This activation allows the reaction to proceed efficiently at low temperatures, ranging from -50°C to 40°C, which is crucial for maintaining stereochemical integrity and preventing side reactions.
Following the coupling, the resolution step utilizes D-tartaric acid to separate the enantiomers of the hydrazino-morpholine intermediate. The formation of the D-tartrate salt is a classic yet highly effective method for chiral purification. The patent specifies that this resolution can be performed in alcohols like methanol or ethanol at temperatures between 20°C and 80°C. The resulting salt precipitates out of the solution, allowing for easy filtration and washing. Subsequent basification liberates the free base of the desired enantiomer (Compound 6) with an exceptional enantiomeric excess (ee) value of 99.5%. This high level of optical purity is critical for the efficacy and safety of the final antiviral drug, ensuring that the biological activity is maximized while minimizing potential toxicity from the opposite enantiomer.
How to Synthesize Chiral Barosavir Intermediate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process is divided into five distinct operational units, beginning with the protection of morpholine and culminating in the final condensation with the pyridone derivative (Compound 7). The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process development teams aiming to replicate or optimize this technology.
- Protect morpholine with allyl chloroformate to form the Alloc-protected intermediate.
- Perform methoxylation using NBS in methanol and formic acid to functionalize the ring.
- Couple with Boc-hydrazine using a Lewis acid catalyst, followed by chiral resolution with D-tartaric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical attributes of this synthesis route translate directly into tangible business value. The shift away from complex, hazardous, and equipment-intensive processes towards a streamlined, crystallization-based workflow addresses several key pain points in the pharmaceutical supply chain. The following analysis details how this technology drives efficiency and risk mitigation.
- Cost Reduction in Manufacturing: The most significant economic driver of this new method is the substitution of expensive and hazardous reagents with commodity chemicals. By eliminating the need for sodium borohydride and specialized hydrogenation catalysts, the direct material costs are substantially lowered. Furthermore, the removal of column chromatography from the purification train represents a massive reduction in operational expenditure. Chromatography is not only slow and labor-intensive but also consumes vast quantities of silica and solvents. Replacing this with crystallization and filtration significantly reduces waste disposal costs and solvent purchase requirements, leading to a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on "cheap and easily-obtained" raw materials, such as morpholine and allyl chloroformate, ensures a stable supply base. Unlike specialized chiral building blocks that may have limited suppliers and long lead times, these starting materials are produced globally at massive scales. Additionally, the early resolution strategy mitigates the risk of batch failure. In late-resolution processes, a failure in the final step results in the loss of all accumulated value. By resolving early, any off-spec material is identified and corrected before expensive reagents are introduced, thereby stabilizing the overall production schedule and ensuring consistent on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The avoidance of heavy metal hydrogenation catalysts and reactive hydrides simplifies the safety profile of the plant. The solvents used—methanol, ethanol, ethyl acetate—are widely accepted in green chemistry initiatives and are easily recovered and recycled. The reduction in "three wastes" (waste water, waste gas, waste residue) aligns with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on the manufacturer and minimizes the risk of production shutdowns due to environmental non-compliance, ensuring long-term business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Barosavir intermediates. These answers are derived directly from the experimental data and claims presented in the patent literature, providing clarity for technical evaluators and sourcing professionals.
Q: What are the primary safety advantages of this new synthesis route?
A: Unlike prior art methods that rely on large quantities of sodium borohydride for reduction, this novel route utilizes mild Lewis acid catalysis and avoids hazardous reducing agents, significantly improving process safety.
Q: How does early-stage resolution impact overall yield?
A: By performing chiral resolution at the hydrazino-morpholine stage rather than the final ring-closure step, the process prevents the accumulation of impurities and avoids wasting expensive downstream reagents on the wrong enantiomer.
Q: Is this method suitable for industrial scale-up?
A: Yes, the method eliminates the need for column chromatography and specialized hydrogenation equipment, relying instead on standard crystallization and extraction techniques that are highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Barosavir Intermediate Supplier
The synthesis route detailed in CN112266381B represents a significant opportunity for optimizing the production of antiviral APIs. However, translating a patent into a robust commercial process requires deep expertise in process chemistry and scale-up engineering. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering comprehensive CDMO services tailored to the complex needs of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest global standards.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis specific to your Barosavir supply chain requirements. By leveraging our optimized synthesis protocols, we can help you achieve significant reductions in COGS while enhancing supply security. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us partner with you to secure a sustainable and cost-effective supply of high-purity pharmaceutical intermediates for the next generation of antiviral therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
