Advanced Manufacturing of Barosavir Intermediate IV via Safe Enamine Oxidation Technology
Advanced Manufacturing of Barosavir Intermediate IV via Safe Enamine Oxidation Technology
The pharmaceutical landscape for antiviral treatments has shifted dramatically with the approval of Baloxavir marboxil, marking a new era in influenza management. As global demand for this potent cap-dependent endonucase inhibitor surges, the supply chain for its critical precursors faces intense scrutiny regarding safety, cost, and scalability. Patent CN109879843B introduces a transformative synthetic methodology for producing the key Barosavir intermediate, specifically Compound IV, addressing the severe limitations of prior art. This novel approach leverages a strategic enamine oxidation pathway that bypasses the use of hypertoxic selenium dioxide and expensive ruthenium catalysts, offering a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their manufacturing portfolios. By transitioning from hazardous high-temperature oxidations to mild, aqueous-compatible conditions, this technology not only enhances operator safety but also significantly reduces the environmental footprint associated with heavy metal waste disposal.
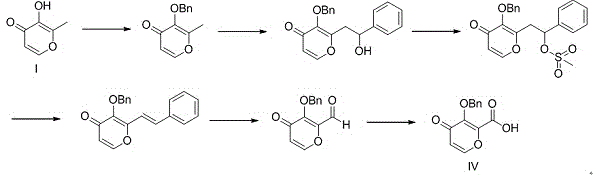
The core innovation lies in the ingenious design of converting a methyl group indirectly into an aldehyde via an enamine intermediate, rather than attempting direct oxidation which often suffers from poor selectivity and harsh conditions. This method utilizes cheap and easily obtained Compound I as the starting material, subjecting it to benzyl protection followed by a condensation reaction to form the novel Compound III. The subsequent oxidation of Compound III is achieved through a controlled two-stage process, first yielding an aldehyde and finally the target carboxylic acid (Compound IV). This sequence represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, as it simplifies the operational complexity while maintaining high yields. For procurement and technical teams, understanding the mechanistic advantages of this route is essential for evaluating long-term supply stability and regulatory compliance in the production of high-value antiviral agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical scaffold has been plagued by significant safety and efficiency hurdles that hinder industrial adoption. One prominent prior art route, documented in the Journal of the American Chemical Society, relies on the direct oxidation of a methyl group using selenium dioxide. While chemically feasible on a small scale, this method requires extreme reaction temperatures reaching 155°C, necessitating specialized high-temperature oil bath heating that is difficult to replicate safely in standard steam-heated workshops. Furthermore, selenium dioxide is a controlled, hypertoxic chemical known to cause severe irritation to skin, mucous membranes, and the respiratory tract, with inhalation risks leading to chemical pneumonia or pulmonary edema. Another existing pathway disclosed in WO2010/11816 A1 attempts to circumvent some issues but introduces a lengthy multi-step sequence involving sulfonylation and elimination.
This alternative route is operationally cumbersome and relies on methylsulfonyl chloride, another hypertoxic reagent with strong irritant properties that can cause laryngeal spasms and burns upon contact. Moreover, the oxidation steps in this legacy process depend on expensive ruthenium catalysts combined with sodium periodate, driving up the raw material costs substantially. The accumulation of heavy metal waste and the need for rigorous purification to remove trace ruthenium from the final API intermediate create significant bottlenecks for commercial scale-up of complex pharmaceutical intermediates. These factors collectively render conventional methods economically unviable and environmentally unsustainable for modern large-scale production requirements.
The Novel Approach
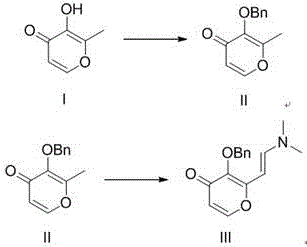
In stark contrast, the novel methodology presented in CN109879843B offers a streamlined, three-step trajectory that prioritizes safety and economic efficiency without compromising yield. The process initiates with the benzyl protection of Compound I, utilizing benign reagents like benzyl bromide and potassium carbonate in polar aprotic solvents such as DMF at moderate temperatures of 45-80°C. This is followed by a condensation reaction with N,N-dimethylformamide dimethyl acetal to generate the key enamine intermediate, Compound III. The true brilliance of this route is manifested in the final oxidation stage, where the enamine is converted to the target acid through a mild, two-phase oxidation system. By avoiding direct methyl oxidation, the process sidesteps the need for toxic selenium reagents entirely.
The oxidation is executed in two distinct stages within a potentially one-pot framework: first, the enamine is oxidized to an aldehyde using sodium periodate at ambient temperatures (10-25°C), and subsequently, the aldehyde is oxidized to the carboxylic acid using sodium chlorite systems. This approach effectively eliminates the use of expensive ruthenium catalysts and toxic selenium compounds, replacing them with cost-effective, readily available oxidants. The reaction conditions are remarkably mild, operating between 0°C and 25°C, which drastically reduces energy consumption and eliminates the need for specialized high-temperature equipment. This novel pathway stands as a superior alternative for reducing lead time for high-purity pharmaceutical intermediates, offering a cleaner, safer, and more economically attractive solution for global supply chains.
Mechanistic Insights into Enamine-Mediated Oxidation Strategy
The mechanistic cornerstone of this invention is the strategic transformation of the methyl substituent into an enamine functionality prior to oxidation. In traditional organic synthesis, the direct oxidation of an aromatic methyl group to a carboxylic acid often requires harsh conditions that can degrade sensitive heterocyclic scaffolds, such as the pyranone ring present in this molecule. By first converting the methyl group into an electron-rich enamine (Compound III) via condensation with DMF-DMA, the molecule becomes primed for oxidative cleavage under much milder conditions. The enamine double bond acts as a reactive handle that is susceptible to attack by periodate ions, facilitating a smooth conversion to the aldehyde intermediate without the radical pathways typically associated with high-temperature oxidations. This indirect conversion strategy effectively decouples the harshness of the oxidation from the stability of the core ring system.
Furthermore, the two-stage oxidation protocol demonstrates exceptional control over impurity profiles, a critical factor for R&D directors focused on purity specifications. The first stage utilizes sodium periodate to selectively cleave the enamine to an aldehyde, a reaction that proceeds rapidly at room temperature with high specificity. The second stage employs a sodium chlorite-based system (often buffered with sulfamic acid or hydrogen peroxide) to oxidize the aldehyde to the carboxylic acid. This specific combination of oxidants minimizes over-oxidation or side reactions on the benzyl protecting group or the pyranone ring. The ability to potentially telescope these steps into a one-pot procedure, where the aldehyde intermediate is not isolated but directly subjected to the second oxidation, significantly reduces solvent usage and processing time. This mechanistic elegance ensures that the final high-purity pharmaceutical intermediate is obtained with minimal downstream purification burden, directly impacting the overall cost of goods sold.
How to Synthesize Barosavir Intermediate IV Efficiently
The synthesis of Compound IV via this novel route is designed for operational simplicity and robustness, making it highly amenable to technology transfer and scale-up. The process begins with the dissolution of the starting material in a polar solvent, followed by the addition of the benzylating agent and base under controlled heating. Once the protected intermediate is secured, it undergoes condensation to form the enamine, which serves as the pivotal precursor for the final oxidation. The detailed standardized synthetic steps, including precise molar ratios, temperature gradients, and workup procedures, are outlined below to guide process engineers in replicating this high-yielding pathway.
- Perform benzyl protection on Compound I using benzyl bromide and potassium carbonate in DMF at 80°C to obtain Compound II.
- Conduct a condensation reaction on Compound II with N,N-dimethylformamide dimethyl acetal at reflux to generate the enamine intermediate (Compound III).
- Execute a two-stage oxidation: first oxidize the enamine to an aldehyde using sodium periodate, then oxidize the aldehyde to the carboxylic acid (Compound IV) using sodium chlorite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic advantages that extend beyond simple unit price reductions. The elimination of controlled, hypertoxic chemicals like selenium dioxide removes a significant regulatory and logistical burden, as sourcing and disposing of such materials require specialized licenses and costly waste treatment protocols. By shifting to common oxidants like sodium periodate and sodium chlorite, the supply chain becomes more resilient and less prone to disruptions caused by the scarcity of exotic reagents. Furthermore, the mild reaction conditions (0-25°C) imply lower energy consumption for heating and cooling, contributing to a leaner manufacturing cost structure that can be passed down as substantial cost savings in the final pricing of the intermediate.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the complete removal of expensive transition metal catalysts, specifically ruthenium, which is a precious metal with volatile pricing and high recovery costs. In conventional routes, the necessity to scavenge trace ruthenium from the final product adds multiple purification steps, increasing solvent usage and processing time. By replacing this with a base-metal-free oxidation system, the new route drastically simplifies the downstream processing. Additionally, the avoidance of selenium dioxide eliminates the高昂 costs associated with hazardous waste disposal and environmental compliance, leading to a significantly optimized cost of goods without compromising yield.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium chlorite, sodium periodate, and benzyl bromide ensures a stable and diversified supply base. Unlike specialized catalysts that may have single-source suppliers or long lead times, these reagents are globally available in bulk quantities, mitigating the risk of raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility supplies (e.g., steam pressure), ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines required by major pharmaceutical clients.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is inherently safer and more scalable. The absence of toxic gas evolution (common with selenium reagents) and the use of aqueous-compatible oxidation systems reduce the risk of workplace exposure incidents. The process generates significantly less hazardous waste, aligning with green chemistry principles and simplifying the permitting process for manufacturing facilities. The ability to run the oxidation in a telescoped manner further reduces the volumetric throughput required, allowing existing reactor trains to produce higher volumes of the intermediate, thereby enhancing overall plant capacity and efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Barosavir intermediate synthesis. These insights are derived directly from the patent data and are intended to clarify the operational benefits and safety improvements offered by this technology. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing frameworks.
Q: Why is the new enamine oxidation route safer than traditional methods?
A: Traditional methods often utilize highly toxic selenium dioxide (SeO2) which poses severe respiratory and skin hazards, or expensive ruthenium catalysts. The new route replaces these with safer oxidants like sodium periodate and sodium chlorite under mild conditions (0-25°C), significantly reducing environmental pollution and operational risk.
Q: What are the key cost drivers eliminated in this synthesis?
A: The process eliminates the need for expensive transition metal catalysts (such as ruthenium) and avoids the complex waste treatment costs associated with heavy metals and toxic selenium byproducts. Additionally, the use of cheap, commercially available starting materials like Compound I further drives down raw material costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights suitability for large-scale production due to mild reaction temperatures (avoiding the 155°C required in older methods), short reaction times, and the ability to perform the oxidation in a one-pot manner without isolating unstable intermediates, which streamlines the workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Barosavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term viability of antiviral drug production. Our team of expert process chemists has extensively analyzed the methodology described in CN109879843B and is fully prepared to execute this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this enamine oxidation strategy are realized in practical, GMP-compliant manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Barosavir intermediate delivered meets the highest global standards for API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this ruthenium-free process for your organization. We encourage you to contact us today to discuss your specific volume requirements,索取 specific COA data, and review our comprehensive route feasibility assessments. Together, we can secure a sustainable and cost-effective supply of this critical influenza medication intermediate.
