Advanced Manufacturing of Tucatinib Intermediates via Novel Pd-Catalyzed Amination Technology
Introduction to Next-Generation Tucatinib Manufacturing
The global demand for targeted oncology therapies continues to surge, placing immense pressure on the supply chains of critical active pharmaceutical ingredients (APIs) and their precursors. Among these, Tucatinib, a potent small molecule HER2 tyrosine kinase inhibitor, represents a cornerstone in the treatment of metastatic HER2-positive breast cancer. Recent intellectual property developments, specifically patent CN114230568B, have unveiled a transformative preparation method that addresses long-standing inefficiencies in the synthesis of this vital medicine. This patent outlines a concise, four-step synthetic pathway that leverages advanced catalytic chemistry to achieve superior yields and purity profiles. For pharmaceutical manufacturers and procurement strategists, understanding the nuances of this technology is paramount, as it offers a clear pathway to optimizing the cost of goods sold (COGS) while ensuring a reliable supply of high-quality pharmaceutical intermediates. The core of this innovation lies not just in the sequence of reactions, but in the strategic selection of catalysts and purification techniques that eliminate bottlenecks traditionally associated with complex heterocyclic synthesis.
Unlike conventional approaches that often suffer from low atom economy and cumbersome purification requirements, the methodology described in CN114230568B prioritizes industrial practicality without compromising on chemical integrity. The inventors have successfully engineered a route where each transformation is highly selective, minimizing the formation of difficult-to-remove impurities that typically plague late-stage intermediates. By focusing on a strategy that utilizes commercially accessible starting materials and robust reaction conditions, this patent sets a new benchmark for the commercial scale-up of complex oncology intermediates. As we delve deeper into the technical specifics, it becomes evident that this process is not merely an academic exercise but a viable, scalable solution designed to meet the rigorous demands of the global pharmaceutical market. The implications for supply chain stability are profound, offering a potential buffer against the volatility often seen in the sourcing of specialized cancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tucatinib and similar quinazoline-based kinase inhibitors has been fraught with challenges that hinder efficient large-scale production. Traditional routes frequently rely on a sequential strategy where a nitro group is first reduced to an amine, followed by a separate halogenation step to introduce the necessary leaving group for subsequent coupling. This multi-step detour inherently increases the operational complexity, extending the overall cycle time and exposing the intermediate to multiple purification events. Each additional step introduces opportunities for yield loss, impurity generation, and increased solvent consumption, all of which contribute to a bloated manufacturing cost structure. Furthermore, conventional methods often struggle with regioselectivity, particularly when dealing with poly-functionalized aromatic systems where competing reaction sites can lead to isomeric byproducts. These byproducts are notoriously difficult to separate, often necessitating the use of preparative high-performance liquid chromatography (HPLC) or extensive column chromatography, which are economically unfeasible for multi-kilogram or ton-scale production. The reliance on such resource-intensive purification techniques creates a significant bottleneck, limiting the ability of suppliers to respond rapidly to market demand fluctuations.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN114230568B streamlines the synthesis into a highly efficient four-step sequence that maximizes convergence and minimizes waste. The most striking deviation from the status quo occurs in the final coupling stage, where the inventors employ a direct amination strategy rather than the traditional reduction-halogenation-coupling sequence. By utilizing a sophisticated palladium catalyst system, the process achieves direct C-N bond formation on a nitro-substituted intermediate, effectively bypassing the need for prior functional group manipulation. This telescoping of synthetic logic not only reduces the total number of unit operations but also significantly enhances the overall mass balance of the process. The result is a synthetic pathway that is inherently leaner, faster, and more cost-effective. Moreover, the reaction conditions are optimized to favor a single reaction site, thereby suppressing the formation of regioisomers and simplifying the downstream purification landscape. This strategic shift allows manufacturers to achieve high-purity outputs using simple crystallization or slurry techniques, marking a substantial leap forward in the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Amination
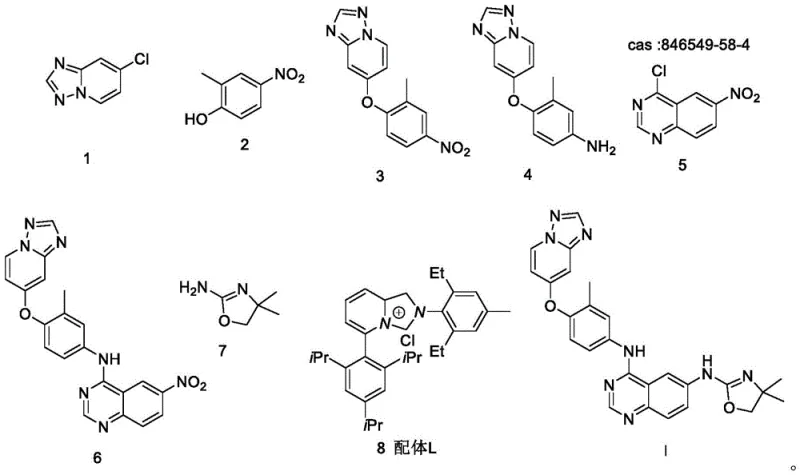
The cornerstone of this innovative synthesis is the fourth step, which employs a palladium-catalyzed amination reaction to couple the key intermediate (Compound 6) with the aminooxazole fragment (Compound 7). Mechanistically, this transformation relies on the synergistic interaction between the palladium source, Pd(acac)2, and a specialized bulky phosphine ligand, referred to as Ligand L in the patent documentation. This catalyst system is meticulously chosen to facilitate the oxidative addition of the aryl chloride bond in Compound 6, even in the presence of other potentially reactive functional groups such as the nitro moiety and the ether linkage. The steric bulk of Ligand L plays a critical role in stabilizing the active palladium species and promoting the reductive elimination step, which forms the final C-N bond with high fidelity. This specific catalytic cycle is engineered to be highly chemoselective, ensuring that the reaction occurs exclusively at the desired chloro-substituted position on the quinazoline ring. Such precision is vital for maintaining the structural integrity of the molecule and preventing the formation of side products that could compromise the safety profile of the final API. The use of Pd(acac)2, a stable and easily handled palladium precursor, further enhances the robustness of the process, making it suitable for diverse manufacturing environments.
Beyond the catalytic cycle itself, the reaction engineering parameters defined in the patent are crucial for controlling the impurity profile. The reaction is conducted in 1,4-dioxane at elevated temperatures (approximately 90°C) in the presence of a mild base, K3PO4·3H2O. These conditions are optimized to drive the reaction to completion while minimizing thermal degradation of the sensitive oxazole ring. A key advantage of this mechanistic approach is the avoidance of harsh reducing agents or strong acids that are often required in alternative synthetic routes. By maintaining a neutral to slightly basic environment throughout the coupling, the process preserves the stability of the intermediate and prevents the hydrolysis of the nitrile or ether groups. Furthermore, the high conversion rates observed (often exceeding 90% yield) indicate that the catalyst turnover number is exceptionally high, allowing for the use of relatively low catalyst loadings. This efficiency translates directly into lower residual metal levels in the crude product, simplifying the metal scavenging steps that are mandatory for pharmaceutical compliance. Ultimately, this mechanistic sophistication ensures that the final product meets stringent purity specifications with minimal downstream processing.
How to Synthesize Tucatinib Efficiently
Implementing this advanced synthetic route requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and optimal yield. The process begins with the preparation of the ether-linked intermediate, followed by a catalytic hydrogenation to generate the aniline precursor. The third step involves a nucleophilic aromatic substitution to install the quinazoline core, setting the stage for the final, critical palladium-catalyzed coupling. Each step has been refined to utilize simple workup procedures, primarily involving filtration and slurry washing, which eliminates the need for complex extraction sequences. For R&D teams looking to adopt this methodology, it is essential to source high-quality starting materials, particularly the specialized Ligand L, to maintain the catalytic efficiency described in the examples. The detailed standardized synthesis steps below provide a roadmap for executing this protocol in a pilot or production setting, ensuring that the theoretical benefits of the patent are realized in practice.
- Perform etherification between Compound 1 and Compound 2 using K2CO3 in DMF at 100°C to obtain Compound 3.
- Reduce the nitro group of Compound 3 using Pd/C and hydrogen in methanol to generate the aniline derivative Compound 4.
- React Compound 4 with Compound 5 in the presence of K2CO3 in DMF at 80°C to form the key intermediate Compound 6.
- Execute the final amination of Compound 6 with Compound 7 using Pd(acac)2 and Ligand L in dioxane at 90°C to yield Tucatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN114230568B offers compelling economic and operational advantages that extend far beyond simple yield improvements. The primary value proposition lies in the drastic simplification of the purification workflow. By replacing expensive and time-consuming column chromatography with simple slurry purification using common solvent systems like petroleum ether and ethyl acetate, the process significantly reduces both solvent consumption and labor hours. This shift not only lowers the direct variable costs associated with manufacturing but also accelerates the batch cycle time, allowing facilities to increase throughput without capital expansion. Furthermore, the use of commercially available starting materials mitigates supply risk, as there is no dependency on bespoke, hard-to-source reagents that could lead to production delays. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor process deviations, which is a critical factor in maintaining supply continuity for high-demand oncology drugs.
- Cost Reduction in Manufacturing: The economic impact of this novel route is driven by the elimination of costly purification steps and the optimization of raw material utilization. Traditional methods often incur significant expenses related to silica gel, large volumes of elution solvents, and the disposal of hazardous waste generated during chromatography. By transitioning to a precipitation and slurry-based purification strategy, manufacturers can achieve substantial cost savings in both material procurement and waste management. Additionally, the high yields observed in each of the four steps mean that less starting material is required to produce a kilogram of the final intermediate, directly improving the atom economy and reducing the cost per unit. The use of a highly efficient catalyst system also minimizes the amount of precious metal required, further contributing to a leaner cost structure. These cumulative efficiencies result in a significantly more competitive pricing model for the final API, providing a strategic advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the simplicity and robustness of the manufacturing process. This patented method enhances reliability by utilizing reagents that are readily available on the global chemical market, reducing the lead time associated with sourcing specialized precursors. The four-step linear sequence is straightforward to scale, with each reaction having been demonstrated to proceed with high consistency and minimal sensitivity to environmental variables. This predictability allows supply chain planners to forecast production output with greater accuracy, ensuring that inventory levels can be maintained to meet fluctuating market demands. Moreover, the reduced complexity of the process lowers the barrier for technology transfer between different manufacturing sites, enabling a more diversified and geographically distributed supply base. This flexibility is crucial for mitigating risks associated with regional disruptions or regulatory changes in specific jurisdictions.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns well with modern green chemistry principles and regulatory expectations. The avoidance of column chromatography significantly reduces the volume of organic solvent waste, lowering the environmental footprint of the manufacturing process and simplifying compliance with increasingly strict environmental regulations. The solvents used, such as methanol, DMF, and dioxane, are well-understood industrially, and their recovery and recycling are standard practices in modern chemical plants. The high purity of the crude product obtained after slurry purification reduces the burden on wastewater treatment facilities and minimizes the generation of solid waste. Furthermore, the process is inherently scalable; the reaction conditions do not rely on exotic equipment or extreme pressures and temperatures, making it adaptable to standard stainless steel reactors found in most multipurpose pharmaceutical facilities. This ease of scale-up ensures that production can be ramped up quickly from pilot batches to commercial tonnage without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tucatinib synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in patent CN114230568B, providing clarity on the practical aspects of the technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios. The answers highlight the specific advantages of the catalytic system and the purification strategy, offering a transparent view of the process capabilities.
Q: What is the key innovation in the Tucatinib synthesis described in CN114230568B?
A: The key innovation lies in Step 4, where a specific catalyst system comprising Pd(acac)2 and Ligand L is used for direct amination. This avoids the conventional multi-step reduction-halogenation sequence, resulting in a single reaction site, higher yield (over 90%), and simplified purification.
Q: How does this method improve purification compared to traditional routes?
A: Traditional methods often require expensive and time-consuming column chromatography. This patented process utilizes simple slurry purification (beating) with petroleum ether and ethyl acetate mixtures after filtration, significantly reducing solvent costs and processing time while maintaining high purity (99.7%).
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for industrial operability. It uses commercially available starting materials, avoids hazardous reagents where possible, and features robust reaction conditions (e.g., 90°C in dioxane) with high yields across all four steps, making it ideal for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tucatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medicines like Tucatinib depends on a partnership grounded in technical excellence and supply chain reliability. Our team of expert process chemists has thoroughly analyzed the methodology presented in CN114230568B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific catalytic requirements of this synthesis, including the safe management of palladium catalysts and the precise control of reaction parameters necessary to achieve the reported high yields. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch of intermediate meets the highest international standards. We understand the critical nature of oncology supply chains and are dedicated to providing a seamless, compliant, and efficient manufacturing service that supports your drug development and commercialization goals.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and timeline requirements. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of adopting this technology for your project. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Together, we can accelerate the availability of high-quality Tucatinib intermediates, ensuring that patients worldwide have timely access to this vital therapy while optimizing your manufacturing economics.
