Advanced Synthesis of Roxadustat Intermediates: Scaling High-Purity API Production Efficiently
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for the synthesis of critical therapeutic agents, and the recent disclosure in patent CN114249690A offers a transformative approach to producing Roxadustat, a potent HIF-PH inhibitor. This patent details a refined synthetic methodology that addresses long-standing bottlenecks in the condensation step required to form the core structure of the drug. By shifting away from hazardous high-pressure conditions and complex solvent systems, this innovation utilizes 1,8-diazabicycloundec-7-ene (DBU) as a liquid organic base in a toluene medium. For R&D directors and process chemists, this represents a significant leap forward in reaction engineering, offering a pathway that not only enhances yield but also drastically simplifies the downstream processing requirements. The strategic implementation of this technology allows manufacturers to bypass the need for specialized autoclave reactors, thereby lowering capital expenditure and operational risk while maintaining exceptional product quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat intermediates has been plagued by significant engineering challenges that hinder efficient commercial scale-up. Prior art methods, such as those described in CN108794397 and WO2014114834, typically rely on an organic base in a methanol system, necessitating reaction temperatures between 100°C and 110°C. Since methanol boils well below this range, these reactions mandate the use of sealed pressure vessels or stainless steel autoclaves, which introduces substantial safety hazards and increases the complexity of reactor design. Furthermore, alternative approaches utilizing high-boiling solvents like 1,4-dioxane or ethylene glycol monomethyl ether present their own set of difficulties; these solvents often exhibit poor solubility for reagents like sodium methoxide and require energy-intensive vacuum concentration steps during workup. The thermal instability of ether solvents at high concentrations further exacerbates safety risks, making these traditional routes less desirable for modern, safety-conscious manufacturing facilities aiming for lean operations.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114249690A introduces a paradigm shift by employing toluene as the primary reaction solvent coupled with DBU as the base. This combination allows the condensation reaction between Compound (I) and glycine to proceed efficiently at a much milder temperature range of 80°C to 85°C under atmospheric pressure. The use of toluene not only eliminates the requirement for pressure-rated equipment but also facilitates a remarkably simple workup procedure where the product can be isolated via direct liquid-liquid separation rather than tedious vacuum distillation. This novel approach effectively decouples the reaction efficiency from extreme thermal conditions, resulting in a streamlined process that reduces reaction time to approximately 3 hours while achieving yields as high as 95%. The transition to this solvent system represents a critical optimization for supply chain managers looking to reduce batch cycle times and minimize energy consumption.
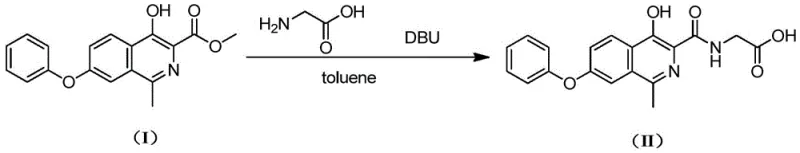
Mechanistic Insights into DBU-Catalyzed Amidation
The success of this synthetic route lies in the unique physicochemical properties of DBU (1,8-diazabicycloundec-7-ene), which acts as a non-nucleophilic, sterically hindered strong base. In the context of this amidation reaction, DBU serves to deprotonate the amino group of glycine, generating a highly nucleophilic species capable of attacking the ester functionality of Compound (I). Unlike smaller amine bases that might participate in unwanted side reactions or form stable salts that complicate isolation, DBU's bulky structure prevents it from acting as a nucleophile itself, ensuring high chemoselectivity for the desired amide bond formation. The mechanism proceeds through a tetrahedral intermediate which collapses to release methanol and form the target amide linkage. The choice of toluene as a solvent is equally critical; its moderate polarity provides an ideal environment for dissolving the organic substrates while allowing the inorganic byproducts or excess reagents to be easily managed during the aqueous workup phase, thus driving the equilibrium towards product formation without requiring excessive thermal energy.
From an impurity control perspective, the lower operating temperature of 80-85°C is instrumental in preserving the integrity of the sensitive isoquinoline scaffold found in Compound (I). High-temperature processes often promote degradation pathways such as hydrolysis of the ester prior to amidation or thermal decomposition of the heterocyclic ring, leading to complex impurity profiles that are difficult to purge. By maintaining the reaction within this optimized thermal window, the formation of thermal degradation byproducts is significantly suppressed. Furthermore, the simplified workup involving pH adjustment to 3.8-4.2 allows for the selective precipitation of the Roxadustat crude product while keeping soluble impurities in the aqueous or organic phase. This precise control over the crystallization event ensures that the resulting crude material possesses a purity of up to 99.7%, drastically reducing the burden on subsequent purification steps and enhancing the overall mass balance of the manufacturing process.
How to Synthesize Roxadustat Efficiently
The execution of this synthesis requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the DBU/toluene system. The process begins with the charging of Compound (I) and glycine into a reactor containing toluene, followed by the controlled addition of DBU to manage the exotherm. Maintaining the reaction temperature between 80°C and 85°C for a duration of 3 hours is essential to ensure complete conversion while avoiding thermal stress on the product. Following the reaction, the mixture is cooled, and water is added to facilitate phase separation, a step that replaces the energy-intensive concentration steps of older methods. For a comprehensive understanding of the specific operational parameters, including exact molar ratios and drying conditions, please refer to the standardized synthesis protocol detailed below.
- Charge Compound (I) and Glycine into a reactor with Toluene solvent, then add DBU dropwise below 45°C.
- Heat the mixture to 80-85°C and maintain for 3 hours to complete the condensation reaction.
- Cool, separate phases, adjust pH to 4.0 with acetic acid to precipitate the product, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis method described in CN114249690A offers tangible strategic advantages that extend beyond mere chemical yield. The elimination of high-pressure reactors translates directly into reduced capital expenditure (CAPEX) for manufacturing facilities, as standard glass-lined or stainless steel reactors can be utilized without the need for expensive pressure ratings. Additionally, the switch to toluene, a ubiquitous and cost-effective industrial solvent, mitigates supply chain risks associated with specialized or hazardous solvents. The simplified workup procedure, which avoids vacuum concentration, significantly shortens the batch cycle time, allowing for increased throughput and faster response to market demand fluctuations. These factors collectively contribute to a more resilient and cost-efficient supply chain for this critical pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The transition to a DBU/toluene system fundamentally alters the cost structure of Roxadustat production by removing the necessity for specialized high-pressure equipment. Traditional methods requiring autoclaves involve higher maintenance costs, stricter safety inspections, and greater energy inputs to maintain pressure. By operating at atmospheric pressure, this new method allows manufacturers to utilize standard reactor infrastructure, thereby lowering both fixed and variable production costs. Furthermore, the avoidance of vacuum concentration steps reduces energy consumption and equipment wear, leading to substantial operational savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The reliance on common industrial solvents like toluene and readily available reagents like glycine and DBU ensures a stable supply chain不受 limited by niche chemical availability. Unlike processes dependent on specific high-boiling ethers or pressurized methanol systems which may face logistical bottlenecks, this route utilizes materials that are globally sourced and consistently available. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment failure or safety shutdowns, ensuring a continuous and reliable flow of high-purity intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The simplicity of the post-treatment process, involving direct liquid separation and pH-adjusted precipitation, makes this route exceptionally scalable from pilot plant to commercial tonnage. The reduction in solvent handling steps and the elimination of high-temperature concentration minimize the generation of volatile organic compound (VOC) emissions, aligning with increasingly stringent environmental regulations. This green chemistry approach not only simplifies waste management but also enhances the sustainability profile of the manufacturing process, a key consideration for modern pharmaceutical supply chains aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on how this method compares to established industry standards. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this process for large-scale production.
Q: Why is the DBU/Toluene system superior to traditional Methanol methods for Roxadustat synthesis?
A: Traditional methods often require high-pressure equipment due to methanol's boiling point limitations at reaction temperatures (100-110°C). The DBU/Toluene system operates effectively at atmospheric pressure (80-85°C), significantly reducing equipment costs and safety risks associated with high-pressure vessels.
Q: How does this process improve impurity control compared to ether-based solvents?
A: Unlike high-boiling ether solvents which require difficult vacuum concentration and pose thermal risks, the toluene system allows for direct liquid-liquid separation. This simplifies workup, minimizes thermal degradation of the product, and ensures higher crude purity (up to 99.7%).
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scale-up. It eliminates the need for specialized pressure reactors, uses common industrial solvents like toluene, and features a simplified isolation procedure that avoids time-consuming vacuum concentration steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN114249690A can be fully realized in an industrial setting. We are committed to delivering high-purity Roxadustat intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Our facility is equipped to handle the specific requirements of the DBU/toluene process, guaranteeing consistent quality and supply continuity for your global operations.
We invite you to collaborate with us to optimize your supply chain for Roxadustat and related pharmaceutical intermediates. By leveraging our technical proficiency, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and efficiency for your organization.
