Advanced Manufacturing Route for Gemcitabine Hydrochloride via Optimized Reformatsky Chemistry
Advanced Manufacturing Route for Gemcitabine Hydrochloride via Optimized Reformatsky Chemistry
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology therapeutics, and the methodology disclosed in patent CN102617677A represents a significant advancement in the production of Gemcitabine Hydrochloride. This specific intellectual property outlines a streamlined process for preparing 2-deoxidized-2,2-difluoro Deoxyribose cytidine compounds, addressing long-standing challenges in yield and purity that have plagued earlier generations of synthesis. By leveraging a modified Reformatsky reaction followed by a carefully controlled lactonization sequence, this route offers a compelling alternative to traditional methods that often rely on cumbersome purification techniques. For R&D directors and process chemists, the elimination of column chromatography and the use of milder reaction conditions signal a pathway that is not only chemically elegant but also inherently more scalable for commercial manufacturing environments.
From a strategic supply chain perspective, the adoption of such optimized routes is critical for ensuring the continuity of supply for life-saving cancer treatments. The patent emphasizes that the method is simple in process and quite suitable for industrial production, which directly correlates to reduced operational complexity and lower risk of batch failure. As a reliable API intermediate supplier, understanding these nuances allows us to align our manufacturing capabilities with the most efficient technologies available. The ability to produce high-purity intermediates without harsh reaction conditions translates to better impurity profiles and reduced downstream processing costs, ultimately benefiting the entire value chain from raw material sourcing to final drug product formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Gemcitabine precursors have been fraught with inefficiencies that hinder cost-effective mass production. Early methodologies, such as those described by L.W. Hertel, relied on asymmetric Reformatsky reactions that suffered from poor selectivity, necessitating further purification via column chromatography to achieve acceptable chiral purity. This reliance on chromatographic separation is a major deterrent for industrial scale-up, as it introduces significant solvent consumption, time delays, and yield losses that accumulate over multiple steps. Furthermore, other attempts to improve selectivity, such as those by T.S. Chou, resulted in overall yields as low as 25%, rendering the process economically unviable for large-scale commercial application.
Additionally, prior art often utilized expensive and hazardous reagents to drive selectivity. For instance, methods employing Titanocene catalysts or iodo difluoro acetate with trialkylchlorosilane additives introduced severe reaction conditions and high material costs. These factors combined to create a synthetic landscape where the production of high-quality Gemcitabine intermediates was both capital-intensive and operationally risky. The need for harsh processing conditions not only increased safety hazards but also complicated waste management and environmental compliance, making these older routes less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the novel approach detailed in CN102617677A simplifies the reaction mechanism and significantly reduces the number of processing steps required to reach the target molecule. By utilizing a specific Reformatsky reaction with zinc and ethyl bromodifluoroacetate in an inert solvent, the process achieves high conversion rates without the need for exotic catalysts. The subsequent deprotection and lactonization steps are conducted under mild acidic conditions using trifluoroacetic acid, which allows for precise control over the cyclization process. This streamlined workflow eliminates the bottlenecks associated with column chromatography, replacing them with standard unit operations like extraction and crystallization that are easily automated and scaled.
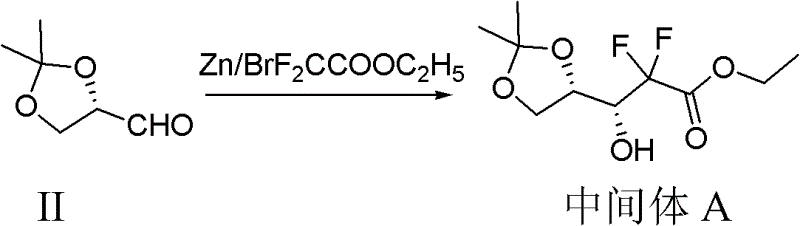
The operational simplicity of this new route is further enhanced by the use of readily available reagents and solvents. The reduction step utilizes lithium aluminum hydride under controlled low temperatures, while the final condensation employs sodium methylate in methanol, a system that is easier to operate and yields superior results compared to previous methods. By avoiding harsh processing conditions and optimizing reaction parameters, this methodology ensures that productive rates are greatly improved. For procurement managers, this translates to a more predictable supply of intermediates with consistent quality, reducing the need for extensive rework or rejection of off-spec batches.
Mechanistic Insights into Reformatsky Reaction and Lactonization
The core of this synthetic strategy lies in the initial Reformatsky reaction, where zinc metal activates ethyl bromodifluoroacetate to form an organozinc intermediate that attacks the aldehyde group of the protected glyceraldehyde derivative. This step is critical for establishing the carbon framework containing the gem-difluoro motif, which is essential for the biological activity of the final drug. The reaction is performed in tetrahydrofuran (THF) with trimethylchlorosilane as an activator, ensuring that the zinc surface remains active throughout the process. Careful control of the addition rate and temperature prevents runaway exotherms and ensures high regioselectivity, resulting in the formation of Intermediate A with minimal byproduct formation.
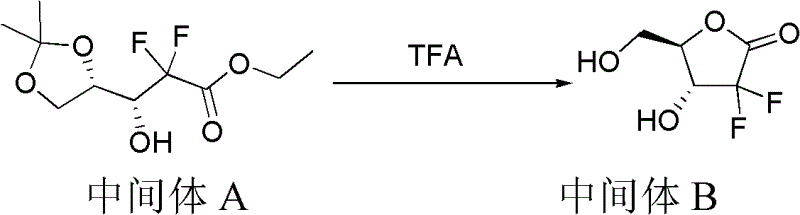
Following the Reformatsky step, the process moves to a crucial lactonization phase mediated by trifluoroacetic acid (TFA) in the presence of water and acetonitrile. This step involves the removal of the acetonide protecting group and the simultaneous cyclization of the hydroxy ester to form the difluoro lactone ring. Maintaining the temperature at 50°C for 3 to 5 hours is vital to drive this equilibrium towards the desired lactone product while minimizing hydrolysis of the ester moiety. The mechanistic precision here ensures that the stereochemistry at the adjacent centers is preserved, which is paramount for the subsequent nucleoside coupling steps.
Impurity control is rigorously managed through temperature regulation during the reduction and mesylation stages. The patent specifies that these reactions must be controlled below 15°C, preferably between 5°C and 10°C, to prevent the formation of unwanted isomers or degradation products. This low-temperature protocol stabilizes the reactive intermediates and ensures that the mesylate group is installed with high fidelity at the anomeric position. Such attention to thermal parameters demonstrates a deep understanding of the reaction kinetics, allowing for the production of intermediates that meet stringent purity specifications required for pharmaceutical applications.
How to Synthesize Gemcitabine Hydrochloride Efficiently
The synthesis of this potent oncology agent requires a disciplined approach to reaction engineering, starting with the activation of zinc and proceeding through a series of protection and coupling steps. The patent outlines a clear trajectory from simple starting materials to the complex nucleoside structure, emphasizing the importance of anhydrous conditions and precise stoichiometry. Detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Perform Reformatsky reaction using Zn and ethyl bromodifluoroacetate with protected glyceraldehyde, followed by deprotection and lactonization to form the difluoro lactone intermediate.
- Execute reduction of the lactone to the hemiacetal, followed by mesylation to activate the anomeric position for coupling.
- Condense the activated sugar with silylated cytosine, followed by deprotection, salification with HCl, and crystallization to obtain the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the transition to this optimized synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification workflow, which directly impacts the cost of goods sold (COGS). By eliminating the need for silica gel column chromatography, the process removes a major source of solvent waste and labor intensity, leading to significant cost reduction in pharmaceutical manufacturing. This efficiency gain allows for tighter margins and more competitive pricing in the global market for generic oncology drugs.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps results in a leaner manufacturing process that consumes fewer resources and generates less hazardous waste. Without the need for large columns and vast quantities of elution solvents, the facility overhead associated with solvent recovery and disposal is markedly decreased. Furthermore, the use of common reagents like zinc powder and ethyl bromodifluoroacetate ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic catalysts.
- Enhanced Supply Chain Reliability: The robustness of this method enhances supply chain reliability by reducing the likelihood of batch failures due to purification issues. Since the process relies on crystallization and extraction rather than delicate chromatographic separations, it is more forgiving of minor variations in feedstock quality. This resilience ensures a steady flow of high-purity intermediates, reducing lead time for high-purity pharmaceutical intermediates and allowing manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures make this route highly scalable from pilot plant to commercial production volumes. The avoidance of heavy metal catalysts like titanium simplifies the regulatory burden regarding residual metal limits in the final API. Consequently, the environmental footprint of the manufacturing process is reduced, aligning with modern sustainability goals and facilitating easier permitting for expansion projects in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications to provide clarity on process capabilities and limitations. Understanding these details is crucial for partners evaluating the feasibility of technology transfer or contract manufacturing agreements.
Q: How does this patent improve upon conventional Gemcitabine synthesis routes?
A: This method eliminates the need for column chromatography, which is a major bottleneck in prior art methods like the Hertel route. By optimizing the Reformatsky reaction conditions and utilizing a simplified lactonization step, the process achieves higher overall yields and avoids the use of expensive catalysts like Titanocene.
Q: What are the critical temperature controls required for this synthesis?
A: Strict temperature control is essential during the reduction and mesylation steps, where reactions must be maintained below 15°C (preferably 5-10°C) to prevent side reactions and ensure stereochemical integrity. Additionally, the lactonization step requires heating to 50°C for 3-5 hours to ensure complete cyclization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its simple operation, mild reaction conditions, and avoidance of harsh processing. The use of standard solvents like THF, methanol, and ethyl acetate, along with straightforward workup procedures like extraction and crystallization, facilitates easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patents like CN102617677A are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Gemcitabine Hydrochloride meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this methodology. We encourage you to contact us for specific COA data and route feasibility assessments to ensure that your project timelines and quality targets are met with precision and reliability.
