Revolutionizing Shikimic Acid Production: A Scalable 4-Step Synthetic Route for Global Pharma Supply Chains
Introduction: Breaking the Supply Chain Bottleneck for Antiviral Intermediates
The global demand for Shikimic Acid, a critical precursor for antiviral medications such as Oseltamivir (Tamiflu), has historically been constrained by reliance on botanical extraction from star anise, a process plagued by seasonal variability and geopolitical supply risks. Patent CN102633629B introduces a transformative synthetic methodology that shifts production from agriculture-dependent extraction to a robust, four-step chemical synthesis starting from the ubiquitous and low-cost feedstock, crotonaldehyde. This technological breakthrough addresses the urgent need for a reliable shikimic acid supplier capable of delivering high-purity intermediates independent of harvest cycles. By leveraging advanced catalytic systems including Sharpless asymmetric epoxidation and efficient one-pot cascade reactions, this route achieves a total yield of 22% with final product purity exceeding 98% ee. For pharmaceutical manufacturers, this represents a paradigm shift towards supply chain resilience, offering a scalable alternative that mitigates the volatility associated with natural product sourcing while maintaining the rigorous stereochemical integrity required for downstream drug synthesis.
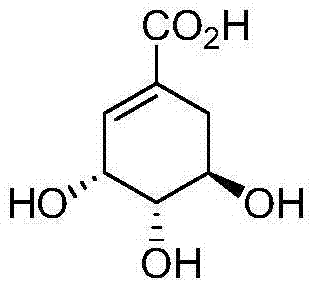
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Shikimic Acid has been dominated by extraction from Illicium verum (star anise), a method inherently fraught with logistical vulnerabilities and economic inefficiencies. The concentration of Shikimic Acid in plant material is naturally low, necessitating massive volumes of biomass and complex fractional extraction processes that drive up the cost of goods significantly. Furthermore, earlier synthetic attempts, such as those reported by Smissman in 1959 or Bestmann in 1971, often relied on hazardous reagents like osmium tetroxide or required prohibitively long synthetic sequences extending up to 17 steps, resulting in dismal overall yields and unacceptable impurity profiles for GMP manufacturing. These conventional pathways fail to meet the modern demands for cost reduction in pharmaceutical intermediate manufacturing, as they generate excessive chemical waste and require extensive purification protocols that erode profit margins. The dependency on agricultural cycles also introduces unpredictable lead times, making it difficult for supply chain heads to guarantee continuous production of life-saving antiviral therapies during pandemic outbreaks.
The Novel Approach
In stark contrast, the methodology disclosed in CN102633629B presents a streamlined, four-step synthetic architecture designed for industrial scalability and environmental compliance. The route initiates with the silylation of crotonaldehyde, followed by a highly efficient one-pot transformation involving a Diels-Alder cycloaddition, desilylation, and nitro elimination to construct the cyclohexene core. This consolidation of three distinct chemical transformations into a single reactor operation drastically reduces solvent usage and processing time, embodying the principles of green chemistry. The subsequent installation of chirality is achieved through a kinetically controlled Sharpless asymmetric epoxidation, which, when coupled with a final one-pot ring-opening and hydrolysis sequence, delivers the target molecule with exceptional stereocontrol. This novel approach not only simplifies the operational complexity but also ensures that the commercial scale-up of complex organic intermediates can be executed with minimal equipment footprint and maximum throughput efficiency.
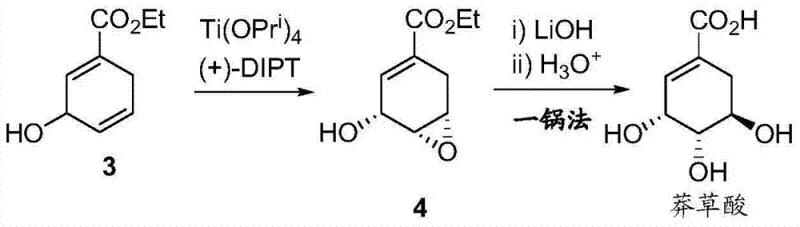
Mechanistic Insights into Sharpless Asymmetric Epoxidation and Kinetic Resolution
The cornerstone of achieving high optical purity in this synthesis lies in the strategic application of the Sharpless asymmetric epoxidation in Step 3, which functions as a powerful kinetic resolution tool. In this critical stage, the racemic allylic alcohol intermediate (Structure 3) is subjected to oxidation using titanium tetraisopropoxide and (+)-diisopropyl tartrate (DIPT) as a chiral ligand. The mechanism relies on the differential reaction rates of the enantiomers; the catalyst system selectively oxidizes one enantiomer much faster than the other. By meticulously monitoring the reaction progress via NMR and terminating the process precisely at 58% conversion, the unreacted starting material is enriched in the desired stereoisomer, while the epoxide product (Structure 4) is formed with high enantiomeric excess (89% ee). This precise control over reaction kinetics is vital for R&D directors focused on impurity control, as it prevents the formation of diastereomeric byproducts that would be difficult to separate in later stages. The use of dry dichloromethane as the solvent further safeguards the moisture-sensitive titanium catalyst, ensuring consistent catalytic turnover and reproducible stereochemical outcomes across different batch sizes.
Following the establishment of chirality, the final transformation involves a tandem epoxy ring-opening and ester hydrolysis, a dual-function reaction that elegantly installs the remaining hydroxyl groups while revealing the free carboxylic acid. The use of lithium hydroxide (LiOH) at a controlled pH of 11.5 is preferred over sodium hydroxide, as empirical data within the patent indicates superior yields (88% vs 84%) and cleaner reaction profiles. The nucleophilic attack of the hydroxide ion occurs regioselectively at the allylic position of the epoxide, driven by electronic factors and the adjacent ester group, leading to the characteristic triol arrangement of Shikimic Acid. This step exemplifies process intensification, where two chemical objectives are met simultaneously in a single pot, thereby minimizing the exposure of the sensitive polyhydroxylated intermediate to potentially degrading conditions. The final acidification to pH < 3 ensures the product precipitates as the free acid rather than a salt, facilitating isolation via simple filtration or extraction, which is a crucial consideration for designing cost-effective downstream processing units.
How to Synthesize Shikimic Acid Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters, particularly regarding the stoichiometry of the silylation step and the temperature control during the asymmetric epoxidation. The initial conversion of crotonaldehyde to the silyl ether must be conducted under anhydrous conditions with zinc chloride acting as a Lewis acid promoter to ensure high conversion rates exceeding 90%. Subsequent steps leverage the reactivity of the diene system to build molecular complexity rapidly, avoiding the need for protecting group manipulations that typically elongate synthetic timelines. For process chemists looking to adopt this technology, the detailed standardized synthesis steps provided below outline the specific molar ratios, solvent choices, and workup procedures necessary to replicate the patent's success metrics in a pilot or production environment.
- Silylation of crotonaldehyde with TMSCl using ZnCl2/Et3N to form 1,3-butadienyl trimethylsilyl ether.
- One-pot Diels-Alder reaction with ethyl 3-nitroacrylate followed by deprotection and nitro elimination to yield ethyl 3-hydroxy-1,4-cyclohexadiene-1-carboxylate.
- Sharpless asymmetric epoxidation using Ti(OPri)4 and (+)-DIPT with kinetic resolution control at 58% conversion.
- Base-mediated epoxy ring opening and simultaneous ester hydrolysis using LiOH, followed by acidification to isolate Shikimic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the transition from extraction-based sourcing to this synthetic methodology offers profound strategic benefits that extend beyond simple unit price considerations. The primary advantage lies in the decoupling of raw material supply from agricultural variables; crotonaldehyde is a commodity chemical produced continuously by the petrochemical industry, ensuring stable pricing and uninterrupted availability regardless of weather patterns or crop failures. This stability allows for long-term contracting and accurate budget forecasting, eliminating the speculative risks associated with botanical raw materials. Furthermore, the implementation of one-pot reaction sequences significantly reduces the volume of solvents and reagents required per kilogram of product, directly translating to lower waste disposal costs and a smaller environmental footprint. This alignment with green chemistry principles not only reduces operational expenditures but also enhances the sustainability profile of the supply chain, a factor increasingly weighted in vendor selection criteria by major multinational pharmaceutical corporations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the consolidation of multiple reaction steps into single vessels, which dramatically lowers capital expenditure on reactor hardware and reduces utility consumption for heating and cooling cycles. By eliminating the isolation and purification of unstable intermediates, the process minimizes material loss and labor hours, resulting in a significantly improved cost structure compared to multi-step linear syntheses. The use of inexpensive, commercially available starting materials like crotonaldehyde and ethyl 3-nitroacrylate further anchors the production cost at a competitive level, enabling substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Dependence on a synthetic route utilizing bulk chemicals mitigates the risk of supply disruptions that frequently plague natural product supply chains. The robustness of the chemistry, characterized by high-yielding transformations and tolerant reaction conditions, ensures consistent batch-to-batch reproducibility, which is essential for maintaining regulatory compliance and meeting just-in-time delivery schedules. This reliability empowers supply chain heads to reduce safety stock levels and optimize inventory turnover, knowing that the production of high-purity antiviral intermediates can be ramped up quickly in response to market demand surges without the lead time delays inherent in agricultural sourcing.
- Scalability and Environmental Compliance: The process design inherently supports scalability, as the one-pot operations reduce the number of unit operations and transfer steps, thereby lowering the potential for human error and contamination during scale-up. The reduction in 'three wastes' emission—waste water, waste gas, and solid residue—aligns with increasingly stringent global environmental regulations, reducing the liability and permitting hurdles associated with chemical manufacturing. This environmentally friendly profile facilitates smoother regulatory approvals and enhances the corporate social responsibility standing of the manufacturing entity, making it a preferred partner for eco-conscious global health initiatives.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and quality assurance of this synthetic pathway. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing transparency for technical evaluators assessing the feasibility of this route for their specific production needs. Understanding these nuances is critical for aligning R&D capabilities with commercial manufacturing goals.
Q: How does this synthetic route improve upon traditional extraction from star anise?
A: Traditional extraction relies on seasonal agricultural sources with limited content and high fractional extraction costs. This synthetic method uses common chemical raw materials like crotonaldehyde, ensuring consistent supply continuity不受 seasonality, shorter reaction steps (4 steps), and a total yield of approximately 22% with purity exceeding 98% ee.
Q: What represents the key cost-saving mechanism in this patent technology?
A: The process utilizes 'one-pot' reaction strategies, specifically combining Diels-Alder cycloaddition, deprotection, and nitro elimination in a single vessel. This eliminates intermediate isolation steps, significantly reducing solvent consumption, labor time, and 'three wastes' emission, leading to substantial operational cost reductions.
Q: Is the optical purity sufficient for antiviral drug manufacturing?
A: Yes. By employing Sharpless asymmetric epoxidation with kinetic resolution (stopping at 58% conversion) and a final recrystallization step, the process achieves an optical purity of greater than 98% ee, meeting the stringent quality specifications required for active pharmaceutical ingredient (API) synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Shikimic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral therapeutics depends on a secure and high-quality supply of key intermediates like Shikimic Acid. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical >98% ee optical purity required for API synthesis. We are committed to delivering reliable shikimic acid supplier services that combine technical excellence with supply chain security, helping our partners navigate the complexities of global pharmaceutical manufacturing with confidence.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this synthetic route. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your production goals are met with the highest standards of quality and reliability.
