Advanced Cefdinir Manufacturing: High-Purity Synthesis via Mesyl Chloride Activation for Global Pharma Supply
Introduction to Advanced Cefdinir Synthesis Technology
The global demand for third-generation cephalosporins continues to drive innovation in antibiotic manufacturing, with Cefdinir standing out as a critical active pharmaceutical ingredient (API) due to its broad spectrum and favorable safety profile. Patent CN102617601A introduces a transformative methodology for preparing Cefdinir that addresses long-standing inefficiencies in traditional synthetic routes. By leveraging a novel activation strategy using mesyl chloride and a precise gradient pH crystallization technique, this technology delivers a product with exceptional purity and stable quality. The structural integrity of the final molecule, characterized by its vinyl group and aminothiazole side chain, is preserved through mild yet effective reaction conditions. 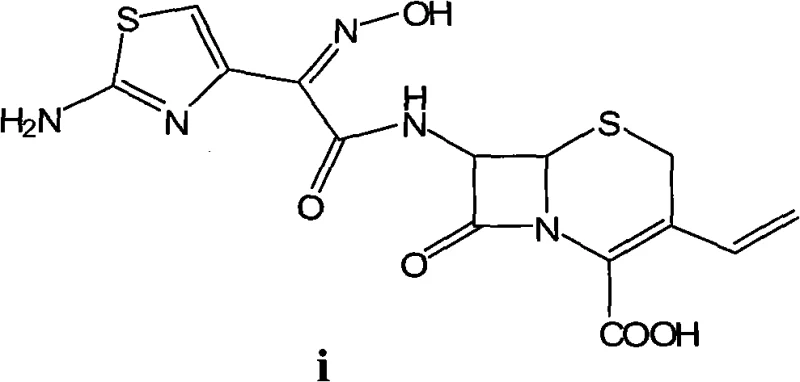
This technical breakthrough is particularly significant for supply chain stakeholders who require reliable sources of high-quality intermediates. The method replaces costly and hard-to-source activating agents with common industrial chemicals, thereby streamlining the procurement process. Furthermore, the elimination of complex intermediate isolation steps reduces the overall processing time and waste generation. For R&D directors and process chemists, this patent offers a robust framework for optimizing yield and minimizing impurity profiles, ensuring that the final API meets stringent regulatory standards for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefdinir has been plagued by the reliance on expensive and chemically complex activating groups. As illustrated in prior art such as WO9724358, traditional routes often utilize trityl aminothiazolyloximate active esters which require intricate preparation and purification steps. 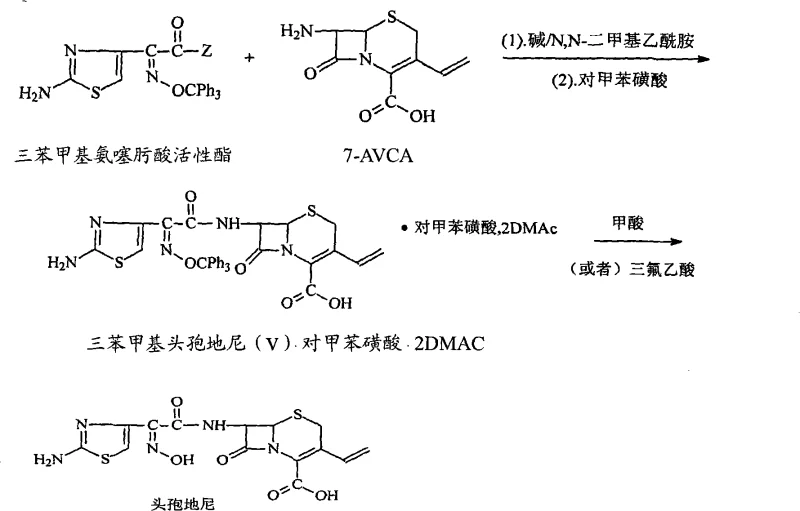
Another common approach, disclosed in patents like CN101817835, employs pentafluorophenol as an activating group. While effective, pentafluorophenol is a high-cost reagent that significantly inflates the raw material bill for large-scale production. Additionally, these conventional methods often suffer from rigid purification protocols where rapid pH adjustments lead to fast crystallization. This rapid precipitation tends to occlude impurities within the crystal lattice, resulting in products with variable color and purity levels typically hovering around 99%, which may require additional recrystallization steps to meet pharmacopeial standards. 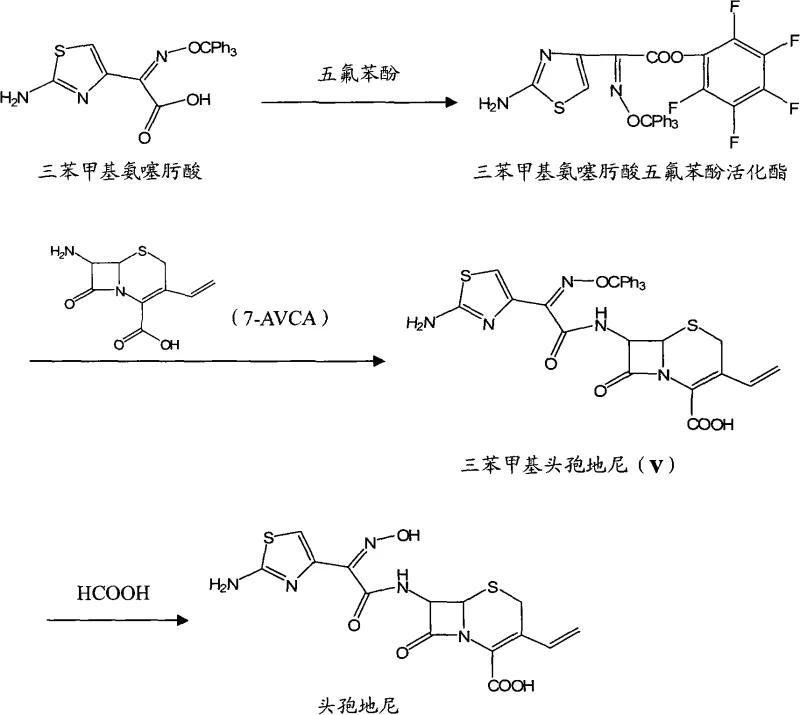
The Novel Approach
The methodology described in CN102617601A fundamentally shifts the paradigm by utilizing mesyl chloride (methanesulfonyl chloride) as the activating agent for the cefdinir side-chain acid. This approach allows for the in situ formation of the reactive intermediate without the need for isolation, creating a seamless one-pot condensation with the 7-AVCA nucleus. 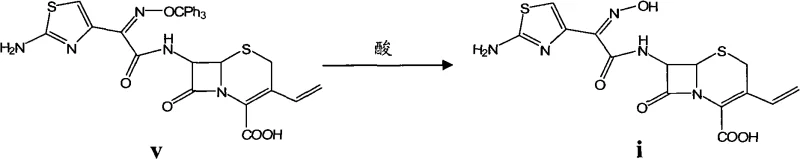
By avoiding the use of exotic activators like pentafluorophenol, the process drastically simplifies the supply chain requirements. The reaction conditions are optimized to proceed rapidly at low temperatures, ensuring high conversion rates. Moreover, the subsequent deprotection and purification stages utilize a gradient temperature and pH control system. This gentle manipulation of the solution environment allows for the selective precipitation of pure Cefdinir while leaving by-products and impurities in the mother liquor, achieving purity levels greater than 99.5% directly from the crystallization step.
Mechanistic Insights into Mesyl Chloride Activation and Gradient Crystallization
The core chemical innovation lies in the efficient activation of the carboxylic acid group on the side chain using mesyl chloride in the presence of an organic base. At temperatures ranging from -25°C to 0°C, the mesyl chloride reacts with the trityl aminothiazolyloximate to form a highly reactive mixed anhydride or activated ester species. This species is sufficiently electrophilic to attack the amino group of the 7-AVCA beta-lactam nucleus, forming the amide bond essential for the cephalosporin structure. The use of bases such as triethylamine, diisopropylethylamine, or tetramethylguanidine facilitates this coupling by scavenging the generated acid, driving the equilibrium towards product formation without degrading the sensitive beta-lactam ring.
Following the condensation, the removal of the trityl protecting group is achieved under acidic conditions, preferably using phosphoric acid or hydrochloric acid. The critical mechanistic advantage of this patent is the post-reaction workup. Instead of a binary pH switch, the process employs a multi-stage pH regulation. Initially, the pH is adjusted to a neutral range (6.0-7.0) to extract impurities into the aqueous or organic phase depending on their solubility. Subsequently, the pH is gradually lowered to the isoelectric point of Cefdinir (around pH 2.5-3.0) while carefully controlling the temperature from 15°C up to 40°C and then cooling back down. This thermodynamic control ensures that nucleation occurs slowly and orderly, promoting the growth of large, pure crystals and excluding solvent inclusions or structural impurities.
How to Synthesize Cefdinir Efficiently
The synthesis of Cefdinir via this optimized route involves a sequence of activation, condensation, and controlled crystallization that maximizes yield and purity. The process begins with the activation of the side chain followed by immediate coupling with the cephalosporin nucleus, eliminating the need for intermediate storage. Detailed standardized operating procedures regarding stoichiometry, solvent volumes, and specific temperature ramps are critical for reproducibility. For a comprehensive guide on executing this synthesis with precision, refer to the step-by-step instructions below.
- Activate the cefdinir side-chain acid trityl aminothiazolyloximate with mesyl chloride in ethyl acetate at low temperatures (-25 to 0°C) to form the reactive intermediate in situ.
- Condense the activated intermediate with 7-AVCA (7-amino-3-vinyl-3-cephem-4-carboxylic acid) using an organic base such as triethylamine or diisopropylethylamine without isolating the intermediate.
- Perform detritylation and purification through a gradient pH and temperature control process, adjusting pH from neutral to acidic ranges while managing temperature to ensure high crystal purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits centered around cost stability and operational efficiency. The substitution of high-value specialty chemicals with commodity reagents like mesyl chloride creates a more resilient supply chain less susceptible to price volatility. The simplification of the process flow, particularly the elimination of intermediate isolation, reduces the number of unit operations required, which directly correlates to lower labor costs and reduced equipment occupancy time. This efficiency translates into a more competitive cost structure for the final API, allowing manufacturers to maintain healthy margins even in price-sensitive markets.
- Cost Reduction in Manufacturing: The primary driver for cost reduction is the replacement of expensive activating agents such as pentafluorophenol or pre-formed active esters with mesyl chloride, which is a widely available and inexpensive industrial chemical. By performing the activation and condensation in a single pot without isolating the intermediate, the process saves on solvent usage, filtration media, and drying energy. This streamlined approach significantly lowers the variable cost per kilogram of production, making the manufacturing of Cefdinir economically superior to legacy methods that rely on complex multi-step activations.
- Enhanced Supply Chain Reliability: The reliance on common raw materials such as 7-AVCA, trityl aminothiazolyloximate, and mesyl chloride ensures a robust supply chain. Unlike specialized reagents that may have limited suppliers or long lead times, these inputs are standard in the fine chemical industry. This availability reduces the risk of production stoppages due to material shortages. Furthermore, the high yield and purity achieved reduce the need for reprocessing or sourcing secondary suppliers for off-spec material, thereby stabilizing the overall supply continuity for downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable due to its straightforward reaction conditions and the use of common solvents like ethyl acetate and acetone. The gradient pH crystallization technique not only improves quality but also simplifies waste treatment by concentrating impurities in the mother liquor, which can be processed more efficiently. The reduction in reaction steps and the avoidance of heavy metal catalysts or toxic activators align with green chemistry principles, facilitating easier regulatory compliance and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefdinir synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: How does the mesyl chloride activation method improve cost efficiency compared to traditional active esters?
A: Traditional methods often rely on expensive activating agents like pentafluorophenol or complex trityl active esters which are difficult to source and costly. The patented method utilizes mesyl chloride, a common and inexpensive reagent, to activate the side chain in situ. This eliminates the need for pre-synthesizing and isolating expensive active esters, significantly reducing raw material costs and simplifying the operational workflow for large-scale manufacturing.
Q: What specific purification technique ensures the high purity of the final Cefdinir product?
A: The process employs a sophisticated gradient pH and temperature regulation strategy during the workup phase. Instead of a single sharp pH adjustment which can trap impurities, the method gradually lowers the pH from neutral (6.0-7.0) to acidic ranges (2.5-3.0) while simultaneously managing temperature shifts (0°C to 40°C). This controlled crystallization allows impurities to remain in the mother liquor while the Cefdinir precipitates as high-quality crystals with purity exceeding 99.5%.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the route is specifically designed for industrialization. It uses readily available raw materials like 7-AVCA and common solvents (ethyl acetate, acetone). The reaction times are short (10-30 minutes for activation), and the one-pot condensation avoids intermediate isolation, which enhances throughput. Furthermore, the robust purification protocol ensures consistent quality, making it highly reliable for commercial scale production ranging from hundreds of kilograms to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102617601A to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this mesyl chloride activation method are fully realized in a GMP-compliant environment. We are committed to delivering Cefdinir and its intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and high-yield technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of quality and efficiency in Cefdinir manufacturing.
