Optimizing Cefdinir Production: A Technical Analysis of High-Yield Deprotection Methodologies
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibiotic intermediates, particularly for third-generation cephalosporins like Cefdinir. Patent CN1366527A introduces a transformative methodology for the preparation of 3-vinyl-cephalosporin compounds, addressing long-standing challenges in the final deprotection stages of synthesis. This technology shifts the paradigm from hazardous Lewis acid catalysis to a safer, more efficient system utilizing perhalic acids in combination with organic protic acids. By fundamentally altering the reaction environment, this process mitigates the degradation of the sensitive beta-lactam core while simultaneously enhancing overall reaction yields. For R&D directors and process chemists, this represents a significant opportunity to refine impurity profiles and streamline manufacturing protocols for high-value oral antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deprotection of protected 3-vinyl-cephalosporin derivatives has been plagued by inefficient methodologies that compromise both safety and economic viability. Prior art, such as the methods disclosed in Japanese Patent Publication No. 1-49273, relied heavily on boron trifluoride etherate in anisole and acetic acid mixtures. These conventional approaches suffered from critically low reaction yields, often stagnating around 35%, necessitating extensive recycling of starting materials and driving up production costs. Furthermore, the reliance on large quantities of dangerous Lewis acids introduced significant occupational health hazards and required specialized containment infrastructure. Alternative methods utilizing trifluoroacetic acid (TFA) in the presence of anisole, as seen in JP-A-62-294687, fared even worse with yields dropping to approximately 28%. The volatility of TFA, combined with its high cost and the complexity of handling large volumes of strong acids, rendered these processes unsuitable for modern, green chemistry-compliant industrial manufacturing.
The Novel Approach
The innovative process detailed in the patent data overcomes these historical bottlenecks by employing a dual-acid system comprising a perhalic acid and an organic protic acid within an organic solvent matrix. Instead of relying on harsh Lewis acids that attack the molecular skeleton, this method leverages the unique ability of organic protic acids, such as formic acid or acetic acid, to form stabilizing hydrogen bonds with the amide and amino groups of the substrate. This stabilization allows the use of only catalytic or minimal stoichiometric amounts of strong perhalic acids, such as perchloric acid, to drive the deprotection efficiently. The result is a dramatic improvement in process performance, with experimental data indicating yields soaring to between 90% and 96%. This approach not only safeguards the integrity of the acid-labile beta-lactam ring but also simplifies the downstream processing, as the reaction mixture can be neutralized and extracted using standard aqueous bicarbonate solutions without the need for complex distillation or hazardous waste treatment associated with excess TFA.
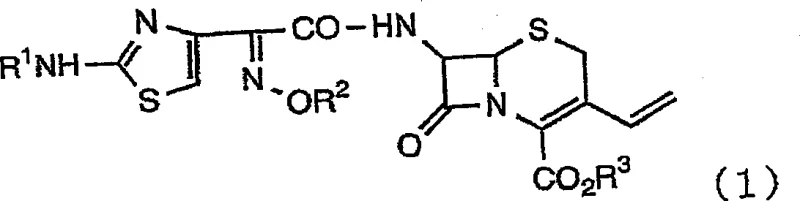
Mechanistic Insights into Perhalic Acid-Catalyzed Deprotection
The core mechanistic advantage of this synthesis lies in the synergistic interaction between the organic protic acid and the perhalic acid catalyst. In traditional strong acid hydrolysis, the high concentration of protons often leads to indiscriminate protonation of various functional groups, including the oxime ether and the beta-lactam nitrogen, triggering decomposition pathways or unwanted cis/trans isomerization of the oxime moiety. In contrast, the novel method utilizes organic acids with a pKa range of 3 to 5, such as formic acid, which selectively interact with the basic sites on the molecule through hydrogen bonding. This interaction effectively shields the sensitive regions of the molecule from the harsh effects of the strong perhalic acid. The perhalic acid, present in minimal amounts (0.1 to 5 molar equivalents), acts primarily to activate the protecting groups for cleavage without overwhelming the system with free protons that would degrade the product. This delicate balance ensures that the deprotection occurs rapidly at mild temperatures, typically between 0°C and 50°C, preserving the stereochemical integrity of the (6R, 7R) configuration essential for biological activity.
Furthermore, the choice of solvent plays a pivotal role in the success of this mechanistic pathway. The patent highlights the efficacy of halogenated hydrocarbons like dichloromethane and chloroform, as well as aromatic solvents like toluene and benzene. These solvents provide an optimal dielectric environment that supports the formation of the necessary ion pairs and hydrogen-bonded complexes without participating in side reactions. The stability of the intermediate species in these solvents allows for extended reaction times if necessary, although high conversion is typically achieved within one hour. This robustness against solvent variation provides process engineers with flexibility in selecting solvents based on availability and environmental regulations, ensuring that the reaction remains high-yielding even if supply chain constraints affect the availability of a specific solvent grade. The minimization of acid usage also means that the final product is less contaminated with acid salts, reducing the burden on purification steps and enhancing the overall purity profile of the final API intermediate.
How to Synthesize 3-Vinyl-Cephalosporin Compounds Efficiently
The implementation of this deprotection strategy offers a straightforward path for laboratory and pilot-scale synthesis, requiring standard equipment and readily available reagents. The process begins by dissolving the protected precursor in a chosen organic solvent, followed by the controlled addition of the acid mixture. The reaction progress is easily monitored, and the workup involves a simple liquid-liquid extraction, avoiding the need for chromatographic purification in many cases.
- Dissolve the protected 3-vinyl-cephalosporin derivative (Formula 1) in a suitable organic solvent such as dichloromethane or toluene.
- Add an organic protic acid (e.g., formic acid) and a catalytic amount of perhalic acid (e.g., perchloric acid) to the reaction mixture.
- Stir the reaction at 0-50°C, followed by extraction with aqueous bicarbonate and acidification to isolate the pure Cefdinir compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route presents compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of expensive and hazardous reagents like boron trifluoride etherate and large volumes of trifluoroacetic acid directly translates to substantial cost reductions in raw material procurement. Additionally, the simplified workup procedure reduces the consumption of neutralizing agents and minimizes the generation of hazardous acidic waste, leading to lower disposal costs and a reduced environmental footprint. The robustness of the reaction conditions also implies a more reliable supply chain, as the process is less sensitive to minor fluctuations in reagent quality or temperature control, thereby reducing the risk of batch failures and ensuring consistent delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The shift away from high-cost Lewis acids and volatile fluorinated reagents significantly lowers the direct material costs associated with the deprotection step. By utilizing catalytic amounts of perchloric acid and commodity organic acids like formic acid, the process reduces the dependency on specialty chemicals that are subject to price volatility and strict regulatory controls. Furthermore, the high reaction yield minimizes the loss of valuable intermediate materials, ensuring that a greater proportion of the input mass is converted into saleable product, which effectively lowers the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The use of common organic solvents such as dichloromethane, toluene, and ethyl acetate ensures that the supply chain is not bottlenecked by the availability of exotic or highly regulated solvents. These solvents are widely produced and available from multiple global suppliers, mitigating the risk of supply disruptions. Moreover, the mild reaction conditions reduce the wear and tear on reactor vessels and piping, extending the lifespan of manufacturing equipment and reducing maintenance downtime, which contributes to a more stable and predictable production schedule for meeting market demand.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable at mild temperatures and the absence of gas-evolving side reactions that complicate large-scale operations. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. The ability to perform simple extractions and crystallizations for purification means that the process can be easily transferred from pilot plants to multi-ton commercial reactors without the need for complex engineering modifications, accelerating the time to market for generic versions of the antibiotic.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this deprotection technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on reaction parameters and product quality.
Q: What are the primary advantages of using perhalic acid over boron trifluoride for Cefdinir synthesis?
A: The use of perhalic acid eliminates the need for hazardous and expensive Lewis acids like boron trifluoride etherate, which historically resulted in low yields (around 35%) and complex handling requirements. The new method achieves significantly higher yields (over 90%) while simplifying the workup procedure.
Q: How does this process improve impurity control compared to traditional trifluoroacetic acid methods?
A: Traditional methods using large excesses of trifluoroacetic acid often lead to cis/trans isomerization of the oxime group and degradation of the beta-lactam ring. The novel approach utilizes a hydrogen-bonding mechanism with organic protic acids that stabilizes the molecule, preventing unwanted isomerization and maintaining high stereochemical purity.
Q: Is this synthesis method scalable for industrial production of antibiotic intermediates?
A: Yes, the process is designed for industrial applicability. It uses common organic solvents like dichloromethane and toluene, operates at mild temperatures (0-50°C), and requires only simple extraction and crystallization steps for purification, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-quality antibiotic intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Cefdinir intermediate meets the highest global pharmacopoeia standards. Our facility is equipped to handle the specific solvent systems and acid handling requirements of this advanced deprotection process, providing a secure and compliant manufacturing environment for our partners.
We invite global pharmaceutical partners to collaborate with us to leverage this superior synthetic technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our optimized manufacturing capabilities can enhance your product portfolio and reduce your overall cost of goods sold.
