Advanced Manufacturing of Substituted Indole-3-Carbaldehyde Intermediates via Optimized Leimgruber-Batcho Synthesis
Introduction to High-Efficiency Indole Intermediate Manufacturing
The pharmaceutical and agrochemical industries rely heavily on the availability of high-purity heterocyclic building blocks, with substituted indole-3-carbaldehyde compounds serving as pivotal precursors for a vast array of bioactive molecules. As detailed in patent CN102746211A, a revolutionary manufacturing protocol has been established that addresses the longstanding inefficiencies of traditional indole synthesis. This proprietary methodology leverages an optimized Leimgruber-Batcho sequence coupled with a refined Vilsmeier-Haack formylation, achieving an unprecedented overall yield exceeding 80% with individual step conversions consistently surpassing 90%. The significance of this technological leap cannot be overstated for R&D directors seeking reliable supply chains for complex API intermediates.
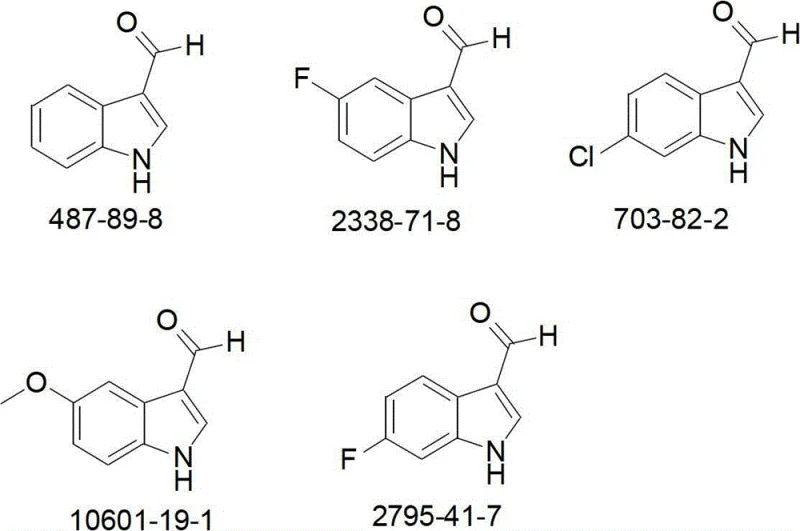
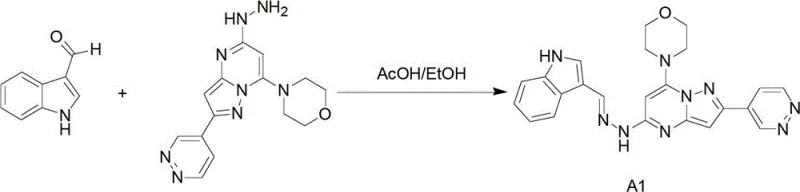
The versatility of this synthetic platform is demonstrated by its ability to accommodate diverse electronic environments on the indole ring, including halogens and alkoxy groups, without compromising reaction efficiency. For instance, the resulting intermediates are directly applicable in the synthesis of potent immunomodulators, as evidenced by downstream transformations into compounds like A1 shown in the reaction scheme below. This capability positions the process as a critical enabler for the rapid development of next-generation therapeutics targeting immune system disorders and cardiovascular diseases.
Furthermore, the strategic integration of this synthesis into commercial workflows allows for the seamless production of key scaffolds required for tryptophan analogues and plant growth regulators. By shifting away from archaic high-temperature protocols, manufacturers can now access these valuable chemical entities with drastically reduced energy consumption and improved safety profiles. The following analysis dissects the mechanistic advantages and commercial implications of adopting this state-of-the-art production route for global supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole nucleus has been dominated by classical methodologies such as the Fischer Indole Synthesis and the Reisert Indole Synthesis, both of which suffer from inherent thermodynamic and operational constraints that hinder large-scale manufacturing. A primary drawback of these traditional routes is the frequent necessity to install a carboxyl group at the 2-position of the indole ring, which subsequently requires a decarboxylation step to yield the unsubstituted or specifically substituted target. This decarboxylation process typically demands extreme thermal conditions, often exceeding 200°C, which not only imposes severe energy costs but also introduces significant risks of thermal decomposition and polymerization of sensitive intermediates.
Moreover, the operational complexity associated with maintaining such high temperatures in large-scale reactors creates substantial engineering challenges, including the need for specialized heating media and robust pressure containment systems. From a purity perspective, the harsh conditions often lead to the formation of complex tarry byproducts and difficult-to-remove impurities, necessitating extensive and yield-eroding purification steps. These factors collectively result in a manufacturing process that is economically inefficient and environmentally burdensome, making it ill-suited for the rigorous demands of modern GMP-compliant pharmaceutical production where consistency and cost-control are paramount.
The Novel Approach
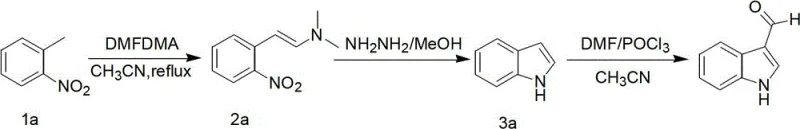
In stark contrast, the novel approach delineated in patent CN102746211A utilizes the Leimgruber-Batcho indole synthesis strategy, which fundamentally reimagines the bond construction logic to operate under significantly milder and more controllable conditions. This method exploits the strong electron-withdrawing nature of the nitro group in substituted o-nitrotoluene starting materials to activate the adjacent methyl group, allowing for a smooth condensation with N,N-dimethylformamide dimethyl acetal (DMFDMA) in a polar solvent system. The resulting beta-enamino nitro compound serves as a highly reactive precursor that undergoes facile reductive cyclization, completely bypassing the need for high-temperature decarboxylation and eliminating the associated thermal degradation pathways.
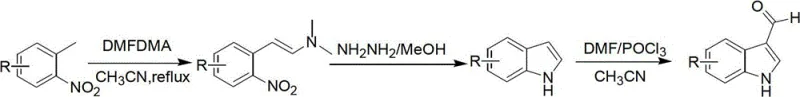
The transition to this modern synthetic route offers a paradigm shift in process safety and scalability, as the entire sequence can be executed at temperatures ranging from ambient to roughly 95°C, well within the standard operating range of conventional glass-lined steel reactors. This reduction in thermal severity not only lowers the capital expenditure required for specialized equipment but also dramatically enhances the safety profile of the operation by minimizing the risk of runaway exotherms. Furthermore, the modularity of this approach allows for the easy introduction of various substituents at the 5- or 6-positions, providing a versatile platform for generating diverse libraries of indole derivatives essential for medicinal chemistry campaigns without the need for process re-optimization.
Mechanistic Insights into Leimgruber-Batcho Cyclization and Vilsmeier Formylation
The core of this manufacturing breakthrough lies in the precise orchestration of three distinct chemical transformations, beginning with the formation of the enamino nitro intermediate through a condensation reaction that is kinetically controlled by solvent polarity and temperature. By utilizing acetonitrile as the reaction medium and maintaining a reflux temperature between 80°C and 95°C, the process ensures optimal solubility of the reactants while facilitating the elimination of methanol or ethanol byproducts, driving the equilibrium towards the desired beta-dimethylamino-2-nitrostyrene with exceptional conversion rates. This step is critical as it sets the stage for the subsequent cyclization, and the use of a slight excess of DMFDMA (molar ratio 4-6:1) guarantees complete consumption of the valuable nitrotoluene starting material, minimizing raw material waste.
Following the condensation, the reductive cyclization step represents a major departure from traditional metal-mediated reductions, employing a hydrazine and methanol mixture to effect the closure of the five-membered pyrrole ring. This homogeneous reduction mechanism avoids the generation of inorganic sludge typical of iron/acetic acid reductions, thereby preventing the physical adsorption of the product onto solid waste matrices which historically plagued isolation yields. The reaction proceeds smoothly at moderate temperatures of 45°C to 60°C, where the hydrazine acts as both a reducing agent for the nitro group and a nucleophile that attacks the activated enamine carbon, triggering the intramolecular cyclization that forms the indole core with high regioselectivity and minimal byproduct formation.
The final installation of the aldehyde functionality at the 3-position is achieved via a modified Vilsmeier-Haack reaction, where the careful management of water content proves to be the decisive factor for success. The inclusion of 4A molecular sieves in the reaction mixture containing DMF and phosphorus oxychloride (POCl3) serves to sequester trace moisture that would otherwise hydrolyze the reactive Vilsmeier reagent or the intermediate iminium salt. This anhydrous environment, maintained initially at low temperatures (0-5°C) during reagent addition and subsequently warmed to room temperature, ensures that the electrophilic attack on the electron-rich indole ring occurs exclusively at the 3-position, yielding the target aldehyde after alkaline workup with impressive purity and yield metrics suitable for direct use in downstream coupling reactions.
How to Synthesize Indole-3-Carbaldehyde Efficiently
The practical execution of this synthesis requires strict adherence to the optimized parameters regarding reagent stoichiometry, temperature gradients, and workup procedures to replicate the high yields reported in the patent literature. The process is designed to be robust and scalable, moving from laboratory benchtop quantities to multi-tonne commercial production with minimal deviation in quality attributes. Below is a summary of the critical operational phases that define this efficient manufacturing route, highlighting the specific conditions required to maximize throughput and minimize impurity generation.
- Condense substituted 2-nitrotoluene with DMFDMA in acetonitrile at 80-95°C to form beta-dimethylamino-2-nitrostyrene.
- Perform reductive cyclization using a hydrazine/methanol mixture at 45-60°C to generate the substituted indole core.
- Execute Vilsmeier-Haack formylation using DMF and POCl3 with 4A molecular sieves, followed by alkaline hydrolysis to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this patented synthesis route translates into tangible economic benefits driven by simplified unit operations and enhanced material efficiency. The elimination of high-temperature decarboxylation steps removes the need for energy-intensive heating protocols, resulting in a drastic reduction in utility costs per kilogram of finished product. Additionally, the shift from heterogeneous iron powder reductions to homogeneous hydrazine reductions simplifies the filtration and waste disposal processes, significantly lowering the environmental compliance costs associated with the treatment of heavy metal-containing sludge.
- Cost Reduction in Manufacturing: The streamlined three-step sequence minimizes the number of isolation and purification stages required, which directly reduces solvent consumption and labor hours associated with batch processing. By avoiding the use of expensive transition metal catalysts or harsh mineral acids in excessive quantities, the raw material cost profile is optimized, allowing for substantial cost savings in the overall cost of goods sold (COGS). The high yield at each step (>90%) means that less starting material is required to produce the same amount of final API intermediate, further enhancing the economic viability of the process for large-scale contracts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as substituted nitrotoluenes, DMFDMA, and hydrazine ensures a stable and diversified supply base, mitigating the risk of shortages associated with exotic or single-source reagents. The robustness of the reaction conditions, particularly the tolerance for standard industrial solvents like acetonitrile and methanol, means that production can be easily transferred between different manufacturing sites without the need for specialized equipment modifications. This flexibility strengthens supply chain resilience, ensuring consistent delivery schedules even in the face of regional logistical disruptions or raw material volatility.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with exothermic events carefully managed through controlled dosing and moderate temperature ranges that are easily handled by standard jacketed reactors. The reduction in hazardous waste generation, specifically the absence of iron sludge and the minimization of acidic wastewater, aligns with increasingly stringent global environmental regulations, reducing the regulatory burden on manufacturing facilities. This 'green chemistry' aspect not only future-proofs the supply chain against tightening emissions standards but also enhances the corporate sustainability profile of the end-product, a key consideration for major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific improvements and data points outlined in the patent documentation. These insights are intended to clarify the operational advantages and quality assurances provided by this advanced manufacturing method for stakeholders evaluating supplier capabilities.
Q: Why is the Leimgruber-Batcho method preferred over Fischer Indole Synthesis for this intermediate?
A: The Fischer method typically introduces a carboxyl group at the 2-position requiring harsh decarboxylation temperatures above 200°C, which complicates industrial operations. The Leimgruber-Batcho route operates under much milder conditions (80-95°C) and avoids high-temperature decomposition risks.
Q: How does the new process improve impurity control compared to traditional iron powder reduction?
A: Traditional methods use iron powder/acetic acid which generates significant solid sludge that adsorbs product and creates filtration bottlenecks. The patented hydrazine/methanol reduction is homogeneous, minimizing solid waste and significantly improving isolation yields to over 90%.
Q: What is the role of 4A molecular sieves in the formylation step?
A: In the Vilsmeier-Haack reaction, water is a byproduct that can hydrolyze the reactive iminium intermediate. The addition of 4A molecular sieves effectively scavenges trace moisture, driving the equilibrium towards the desired aldehyde and ensuring yields exceed 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-3-Carbaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capacity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN102746211A are faithfully reproduced at an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch of substituted indole-3-carbaldehyde intermediates we supply.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective and environmentally superior synthesis route for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-quality indole intermediates that accelerate your drug development timelines while optimizing your manufacturing budget.
