Revolutionizing Indole-3-Carbaldehyde Production: A Scalable, High-Yield Synthetic Route for Global Pharma Supply Chains
The global demand for high-purity indole derivatives continues to surge, driven by their critical role as scaffolds in oncology, neuroscience, and cardiovascular drug development. Patent CN108329249B represents a significant technological breakthrough in the synthesis of indole-3-carbaldehyde compounds, addressing long-standing inefficiencies in traditional formylation methods. This innovation shifts the paradigm from hazardous, waste-intensive processes to a streamlined, Lewis acid-catalyzed protocol utilizing hexamethylenetetramine (HMTA) as a stable carbon source. For R&D directors and procurement strategists, this patent offers a viable pathway to secure a reliable indole-3-carbaldehyde supplier capable of delivering consistent quality while mitigating regulatory and environmental risks associated with legacy chemistries.
Historically, the functionalization of the indole C-3 position has relied heavily on the Vilsmeier-Haack reaction, Reimer-Tiemann reaction, or Duff reaction. These conventional methodologies are plagued by severe operational drawbacks, including the necessity for toxic phosphoryl chloride (POCl3), extreme pH conditions during workup, and poor functional group tolerance. Furthermore, more recent catalytic oxidation approaches, while avoiding POCl3, often introduce new complexities such as the requirement for unstable peroxide oxidants, expensive transition metal catalysts like Ruthenium or Copper, and prolonged reaction times under oxygen atmospheres. 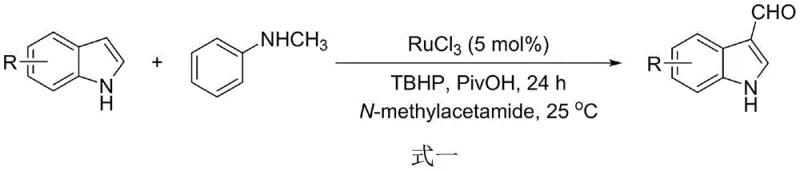
The novel approach detailed in CN108329249B elegantly circumvents these bottlenecks by employing crystalline aluminum trichloride (AlCl3·6H2O) as a robust, inexpensive catalyst and HMTA as a safe, solid formylating agent. This system operates in N,N-dimethylformamide (DMF) at a moderate temperature of 120°C, eliminating the need for hazardous peroxides or gaseous oxygen handling. The reaction demonstrates exceptional substrate scope, accommodating various substituents on the indole ring without compromising yield, thereby offering a versatile solution for cost reduction in pharmaceutical intermediate manufacturing where diverse analog libraries are required.
Mechanistic Insights into AlCl3-Catalyzed Electrophilic Formylation
The core of this synthetic advancement lies in the unique activation mechanism facilitated by the Lewis acidic nature of aluminum trichloride. In this system, AlCl3 coordinates with the nitrogen atoms of the hexamethylenetetramine cage, generating a highly electrophilic iminium-like intermediate with the carbon atom acting as the active center. This activated species undergoes a selective electrophilic aromatic substitution at the electron-rich C-3 position of the indole nucleus, a regioselectivity that is intrinsic to the indole structure but enhanced by the mild conditions of this protocol. Following the substitution, the intermediate undergoes intramolecular electron transfer and subsequent hydrolysis of the imine moiety to release the final aldehyde product. 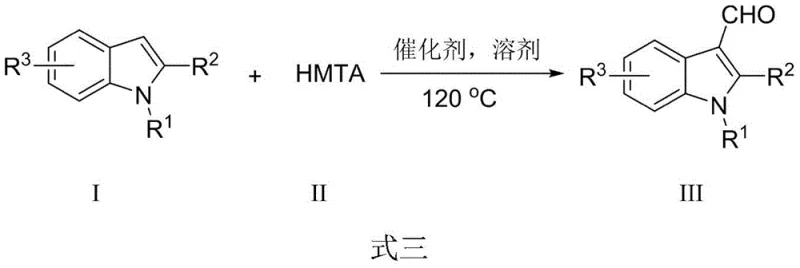
From an impurity control perspective, this mechanism offers distinct advantages over oxidative methods. By avoiding radical pathways associated with peroxide oxidants, the formation of over-oxidized byproducts such as carboxylic acids or ring-opened degradation products is minimized. The use of DMF as a polar aprotic solvent further stabilizes the charged intermediates, ensuring a clean reaction profile that simplifies downstream purification. For quality assurance teams, this translates to a simpler impurity谱 (impurity profile) and higher confidence in meeting stringent purity specifications required for GMP-grade API intermediates.
How to Synthesize Indole-3-Carbaldehyde Efficiently
The operational simplicity of this method makes it particularly attractive for process chemistry teams looking to transfer technology from the lab to the pilot plant. The procedure involves a straightforward one-pot mixing of substrates followed by thermal activation, removing the need for slow addition protocols or cryogenic conditions often seen in Vilsmeier reactions. Detailed standardized synthesis steps are provided below to guide process implementation.
- Mix indole substrate, hexamethylenetetramine (HMTA), and DMF solvent in a reaction vessel with stirring until dissolved.
- Add crystalline aluminum trichloride catalyst and heat the mixture to 120°C under reflux for 1 to 20 hours.
- Cool to room temperature, filter, wash with ethyl acetate, extract, dry, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this HMTA-based methodology represents a strategic opportunity to optimize total cost of ownership and enhance supply resilience. Traditional routes often rely on reagents with volatile pricing or complex logistics, such as compressed gases or moisture-sensitive liquids. In contrast, the key reagents here—HMTA and crystalline AlCl3—are commodity chemicals with stable global supply chains, ensuring consistent availability and shielding production schedules from raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts (like Ru or Cu complexes) and hazardous oxidants directly lowers the bill of materials. Furthermore, the avoidance of toxic POCl3 removes the substantial costs associated with specialized corrosion-resistant equipment and hazardous waste disposal. The process also allows for the recycling of the DMF solvent, contributing to substantial cost savings in solvent procurement and waste treatment operations over the lifecycle of the product.
- Enhanced Supply Chain Reliability: By utilizing solid, shelf-stable reagents that do not require special storage conditions (such as refrigeration or inert atmosphere), the logistical burden is significantly reduced. This stability minimizes the risk of supply disruptions caused by reagent degradation during transport. Additionally, the robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without the need for highly specialized operator training or complex safety infrastructure.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal industrial "three wastes" compared to acid-heavy traditional methods. The simple workup involving filtration and extraction avoids the generation of large volumes of acidic or basic aqueous waste. This aligns perfectly with modern environmental regulations and corporate sustainability goals, facilitating easier permitting for commercial scale-up of complex heterocyclic intermediates from 100 kgs to multi-ton annual capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers.
Q: What are the advantages of using HMTA over Vilsmeier-Haack reagents?
A: Unlike the Vilsmeier-Haack reaction which requires toxic POCl3 and harsh acidic workups, the HMTA method utilizes stable, solid reagents and avoids large doses of strong acids or bases, significantly reducing safety risks and waste treatment costs.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process operates at moderate temperatures (120°C) with simple equipment requirements and allows for solvent recycling, making it highly amenable to commercial scale-up from kilograms to metric tons.
Q: What is the typical yield range for this synthesis?
A: According to patent data, the method achieves high yields, with specific examples demonstrating isolated yields up to 94% for unsubstituted indole-3-carbaldehyde.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-3-Carbaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of life-saving medicines. Our technical team has thoroughly analyzed the potential of the HMTA-mediated formylation strategy and is well-positioned to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of indole-3-carbaldehyde meets the highest international standards.
We invite you to collaborate with us to evaluate the feasibility of this advanced synthesis for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to obtain specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply chain for your next-generation pharmaceutical intermediates.
