Advanced Manufacturing Process for High-Purity P300/CBP Modulator Intermediates
Advanced Manufacturing Process for High-Purity P300/CBP Modulator Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes that can transition seamlessly from laboratory discovery to commercial manufacturing. Patent CN112351982A discloses a highly optimized process for the preparation of Compound (I), a potent modulator of p300 and/or CBP activity with significant potential in treating various cancers including prostate, hematological, bladder, and lung cancers. This technical insight report analyzes the strategic shifts in synthetic design that enhance economic viability and supply chain reliability. The core innovation lies in the reordering of synthetic steps to introduce high-value functional groups at a later stage, thereby minimizing waste and maximizing overall yield. For R&D directors and procurement managers, understanding these mechanistic nuances is critical for evaluating the long-term feasibility of sourcing this complex pharmaceutical intermediate.
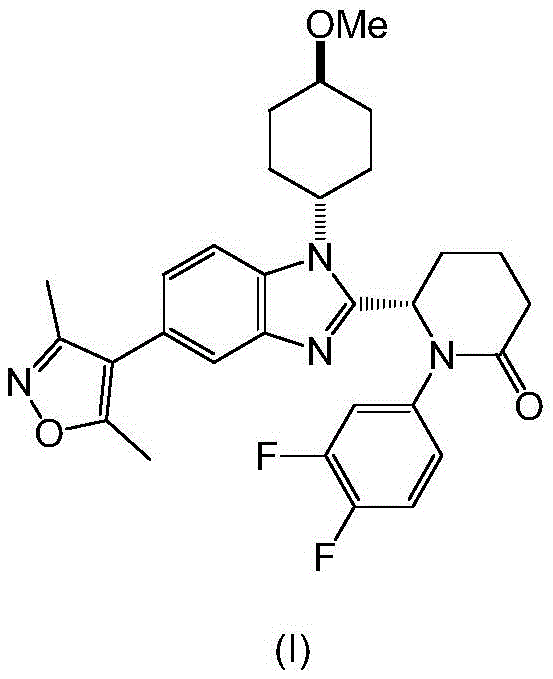
Compound (I), chemically defined as (S)-6-(5-(3,5-dimethylisoxazol-4-yl)-1-((1R,4S)-4-methoxycyclohexyl)-1H-benzo[d]imidazol-2-yl)-1-(3,4-difluorophenyl)piperidin-2-one, represents a challenging synthetic target due to its multiple stereocenters and sensitive heterocyclic systems. The patent highlights that while early laboratory routes were successful in generating milligram quantities for biological testing, they failed to meet the rigorous demands of industrial production. The disclosed method addresses these limitations by implementing a convergent strategy that prioritizes atom economy and operational simplicity. By focusing on the thermodynamic stability of the final crystalline form, the process ensures consistent quality essential for regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The initial laboratory synthesis, referred to as Scheme A in the patent documentation, suffered from several critical inefficiencies that render it unsuitable for commercial scale-up. A primary drawback was the early introduction of the expensive 3,5-dimethylisoxazol-4-substituted boronic acid ester reagent in the very first step. Since this valuable building block was present throughout all five subsequent conversion steps, any yield losses in downstream operations resulted in a disproportionately high consumption of this costly starting material. Furthermore, the reliance on HATU (O-(7-azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate) for amide coupling introduced significant operational hazards. The use of HATU necessitated chromatographic purification to isolate the desired product, a technique that is notoriously difficult to scale and often leads to glassware corrosion due to the generation of corrosive byproducts. These factors collectively inflated the cost of goods sold (COGS) and complicated the supply chain logistics.
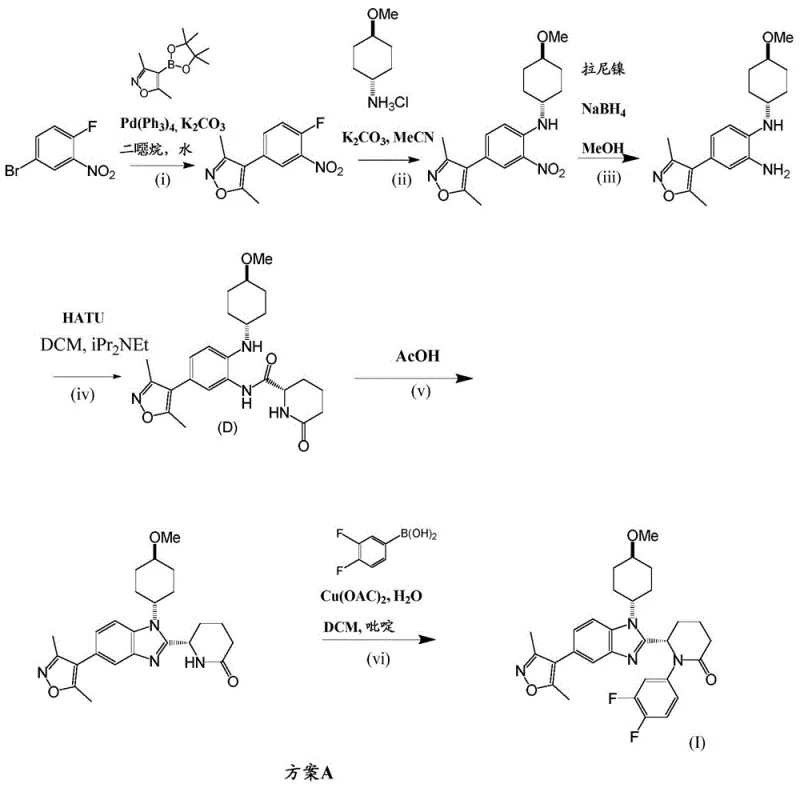
The Novel Approach
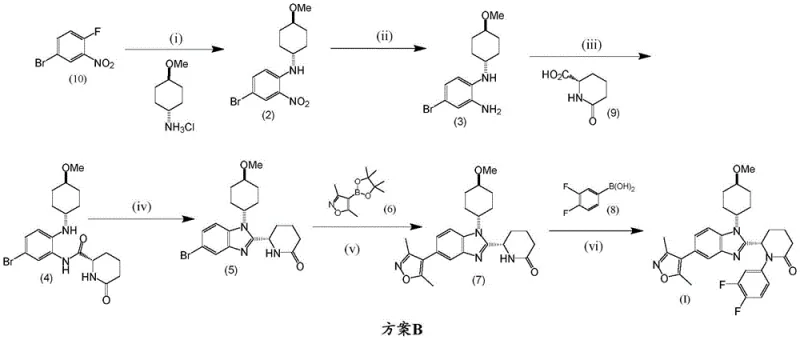
In stark contrast, the novel process outlined in the invention, designated as Scheme B, fundamentally restructures the synthetic timeline to optimize resource utilization. The most significant improvement is the deferral of the 3,5-dimethylisoxazole moiety installation until the penultimate step. By constructing the benzimidazole core and the delta-lactam ring system first, the process ensures that the expensive boronic ester is only subjected to a single transformation step before the final product is obtained. This strategic delay drastically reduces the amount of reagent required to produce a fixed quantity of Compound (I). Additionally, the replacement of HATU with 1-propylphosphonic acid cyclic anhydride (T3P®) for the initial amide coupling eliminates the need for chromatography. T3P® facilitates the formation of the amide bond under milder conditions, allowing for isolation via simple concentration or crystallization, which is far more amenable to large-scale processing.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling and Cyclization
The success of the novel route hinges on two pivotal catalytic transformations: the palladium-catalyzed Suzuki-Miyaura coupling and the copper-mediated Chan-Lam type coupling. In step (v) of Scheme B, the bromo-benzimidazole intermediate (5) reacts with the isoxazole boronic ester (6) in the presence of tetrakis(triphenylphosphine)palladium and potassium carbonate. This reaction is typically conducted in a biphasic system of 1,4-dioxane and water at elevated temperatures (85-95°C). The mechanistic efficiency here is crucial; the electron-deficient nature of the benzimidazole ring facilitates the oxidative addition of the palladium catalyst, while the base activates the boron species for transmetallation. Careful control of the reaction atmosphere, often involving nitrogen purging, prevents catalyst deactivation and ensures high conversion rates. Following this, the intermediate (7) undergoes purification, potentially involving chelating agents like diethanolamine to scavenge residual boron impurities, ensuring the purity required for the final step.
The final assembly of the molecule involves a copper-mediated coupling between intermediate (7) and 3,4-difluorophenylboronic acid (8). This step, performed in dichloromethane with pyridine and copper(II) acetate under an air atmosphere, installs the difluorophenyl group onto the lactam nitrogen. The mechanism likely involves the formation of a copper-amine complex which then reacts with the organoboron species. A key aspect of the process control described in the patent is the management of polymorphism. Compound (I) can exist in different crystalline forms, but the process favors the thermodynamically stable Form 1. This is achieved through precise control of recrystallization parameters, such as cooling rates (e.g., 0.1°C/min) and solvent selection (e.g., ethyl acetate/n-heptane mixtures). Maintaining chiral integrity during the acetic acid-mediated cyclization (step iv) is also paramount, achieved by keeping temperatures moderate (40-50°C) to prevent racemization of the cyclohexyl stereocenter.
How to Synthesize Compound (I) Efficiently
The synthesis of Compound (I) via the disclosed process offers a streamlined pathway that balances chemical complexity with operational practicality. The route begins with the condensation of a bromo-nitro-aniline derivative with a chiral lactam acid, followed by cyclization to form the benzimidazole core. Subsequent cross-coupling reactions build the molecular complexity in a convergent manner. The detailed procedural steps involve specific solvent swaps, temperature gradients, and workup protocols designed to maximize recovery and purity. For process chemists looking to replicate or adapt this chemistry, adherence to the specified stoichiometry and purification techniques is essential to avoid the pitfalls of the older methodology. The following guide summarizes the critical operational phases derived from the patent examples.
- Perform amide coupling between compound (3) and compound (9) using T3P and DIPEA in DCM, followed by acetic acid mediated cyclization at 40-50°C to form compound (5).
- Execute a Suzuki-Miyaura coupling reaction between compound (5) and the boronic ester (6) using Pd(Ph3)4 and K2CO3 in dioxane/water to yield intermediate (7).
- Conduct a copper-mediated coupling of intermediate (7) with 3,4-difluorophenylboronic acid (8) using Cu(OAc)2 and pyridine in DCM to afford the final Compound (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from Scheme A to Scheme B represents a substantial opportunity for cost reduction in pharmaceutical manufacturing. The elimination of chromatographic purification steps not only reduces solvent consumption and waste disposal costs but also significantly shortens the production cycle time. By avoiding the use of HATU, the process mitigates the risk of equipment corrosion, thereby extending the lifespan of reactor vessels and reducing maintenance downtime. Furthermore, the late-stage introduction of the expensive isoxazole boronic ester means that inventory holding costs for this high-value raw material are minimized, as it is only required for the final stages of production. This lean approach to material usage enhances the overall economic viability of the project, making the final API more competitive in the marketplace.
- Cost Reduction in Manufacturing: The substitution of HATU with T3P® and the avoidance of column chromatography lead to significant savings in reagent and solvent costs. The process relies on crystallization for purification, which is inherently cheaper and more scalable than chromatography. Additionally, by delaying the addition of the costly boronic ester until the end of the synthesis, the impact of yield losses in earlier steps on the total material cost is drastically reduced. This structural change in the synthetic route ensures that the most expensive components are not wasted on intermediates that might be discarded due to downstream failures.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route contributes to a more reliable supply chain. The use of standard reagents like T3P® and common solvents like dichloromethane and ethyl acetate ensures that raw material availability is not a bottleneck. The process conditions, such as the moderate temperatures for cyclization and the use of air in the final coupling step, are easier to control in a multipurpose plant compared to the stringent requirements of the previous route. This operational flexibility allows manufacturers to respond more quickly to demand fluctuations without compromising product quality or safety standards.
- Scalability and Environmental Compliance: From an environmental perspective, the new process generates less hazardous waste. The removal of HATU byproducts and the reduction in solvent volume associated with chromatography lower the environmental footprint of the manufacturing process. The ability to recycle solvents like toluene and ethyl acetate further aligns with green chemistry principles. Scalability is enhanced by the fact that the key purification steps rely on crystallization, a unit operation that scales linearly and predictably from the laboratory to the pilot plant and finally to commercial production scales, ensuring consistent batch-to-batch quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Compound (I). These answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their broader drug development pipelines. The focus is on practical implementation, quality control, and the strategic advantages of the disclosed methodology.
Q: How does the new process improve cost efficiency compared to Scheme A?
A: The new process delays the introduction of the expensive 3,5-dimethylisoxazol-4-substituted boronic acid ester until the penultimate step. This minimizes material loss associated with low yields in earlier steps. Additionally, replacing HATU with T3P eliminates the need for chromatographic purification and reduces glassware corrosion.
Q: What are the critical quality attributes controlled during the synthesis?
A: Critical attributes include the chiral integrity of the methoxycyclohexyl group, which is maintained by performing the cyclization at mild temperatures (40-50°C). Furthermore, boron residues are effectively removed using chelating agents like diethanolamine or silica treatment to ensure high purity.
Q: Is the process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for commercial scale-up. It utilizes robust reaction conditions, avoids hazardous reagents where possible, and employs crystallization rather than chromatography for purification, making it highly viable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Compound (I) Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, offering unparalleled expertise in the production of complex oncology intermediates like Compound (I). Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical sector, and our facilities are equipped to deliver materials that meet the highest international standards. By leveraging our advanced process chemistry capabilities, we can help you optimize your supply chain and reduce time-to-market for your therapeutic candidates.
We invite you to engage with our technical procurement team to discuss your specific requirements for this p300/CBP modulator intermediate. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new projects, we are ready to provide comprehensive support. Partnering with us ensures access to a reliable source of high-quality intermediates, backed by a commitment to innovation and customer success in the competitive landscape of drug development.
