Advanced Synthesis of Montelukast Sodium Intermediate for Commercial Scale-up
Advanced Synthesis of Montelukast Sodium Intermediate for Commercial Scale-up
The global demand for high-quality asthma medications continues to drive innovation in the synthesis of key active pharmaceutical ingredients (APIs), particularly Montelukast Sodium. As a selective leukotriene receptor antagonist, Montelukast Sodium remains a cornerstone therapy for respiratory conditions, necessitating robust and cost-effective supply chains for its precursors. Patent CN111848511A introduces a transformative synthesis method for a critical Montelukast sodium intermediate, specifically compound (I), which serves as a pivotal building block in the final drug assembly. This technical insight report analyzes the proprietary methodology disclosed in the patent, highlighting its potential to redefine manufacturing standards for reliable pharmaceutical intermediate suppliers. By shifting away from lengthy, multi-step traditional routes, this novel approach offers a streamlined pathway that addresses long-standing challenges in yield optimization and process safety.
The chemical architecture of Montelukast Sodium is complex, featuring a quinoline ring, a vinyl bridge, and a chiral side chain, all of which must be assembled with precise stereochemical control. The intermediate discussed herein, ((E)-2-[3-[3-[2-(7-chloroquinolyl)ethenyl]phenyl]-3-oxopropyl]benzoic acid methyl ester, represents a advanced stage in this assembly where the core carbon skeleton is largely established. 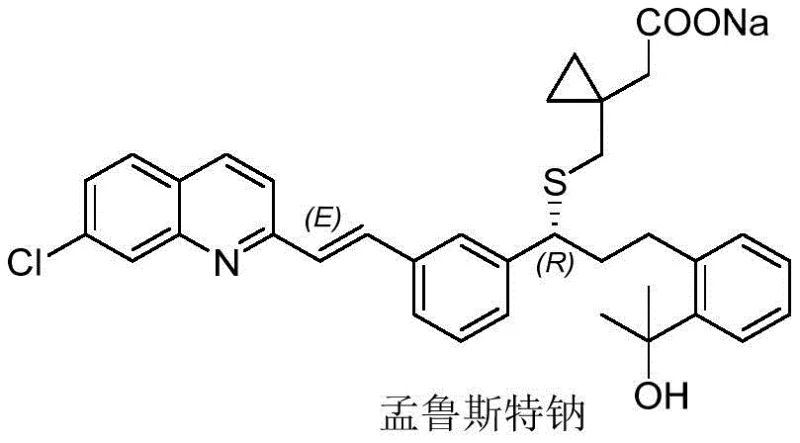 Efficient access to this specific intermediate is crucial for downstream processing, as any inefficiencies at this stage propagate through the entire production line, affecting final API costs and availability. The patent provides a detailed roadmap for achieving this synthesis with remarkable efficiency, leveraging well-understood organic transformations in a novel sequence.
Efficient access to this specific intermediate is crucial for downstream processing, as any inefficiencies at this stage propagate through the entire production line, affecting final API costs and availability. The patent provides a detailed roadmap for achieving this synthesis with remarkable efficiency, leveraging well-understood organic transformations in a novel sequence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry relied heavily on synthesis routes such as the one disclosed in WO2008058118, which utilized isophthalaldehyde and 7-chloro-2-methylquinoline as starting materials. While chemically feasible, this legacy approach suffers from significant drawbacks that hinder modern manufacturing efficiency. The most glaring issue is the excessive number of reaction steps; the conventional route requires a total of seven distinct synthetic operations to reach the desired intermediate. Each additional step introduces opportunities for yield loss, impurity accumulation, and increased consumption of raw materials and solvents. Furthermore, the reaction conditions in these older methods are often harsh, requiring stringent temperature controls or hazardous reagents that complicate safety protocols and increase operational expenditures.
The cumulative effect of these limitations is a production process that is both time-consuming and economically burdensome. Long production cycles lead to extended lead times for procurement teams, while low overall yields necessitate larger batch sizes to meet demand, straining reactor capacity and utility resources. Additionally, the generation of substantial chemical waste from seven steps poses environmental compliance challenges, forcing manufacturers to invest heavily in waste treatment infrastructure. For a procurement manager focused on cost reduction in API manufacturing, these factors make the conventional route increasingly unviable in a competitive market where margin compression is a constant reality.
The Novel Approach
In stark contrast, the method outlined in CN111848511A achieves the synthesis of the target intermediate in just two primary strategic steps, representing a paradigm shift in process design. The new route begins with a Wittig reaction to construct the essential vinyl linkage, followed immediately by a palladium-catalyzed coupling to attach the remaining side chain. This drastic reduction in step count simplifies the entire workflow, minimizing the need for intermediate isolations and purifications that typically erode yield. The reaction conditions are notably mild, operating at moderate temperatures and utilizing standard organic solvents, which enhances operational safety and ease of handling. 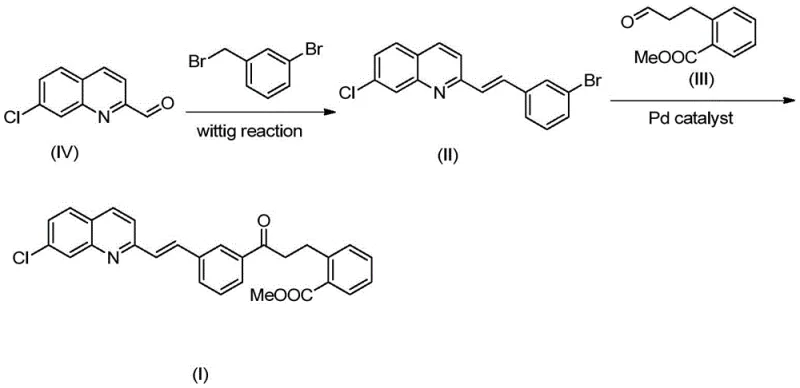
This streamlined approach directly translates to substantial process cost savings and improved throughput. By condensing the synthesis into fewer operations, manufacturers can significantly reduce the footprint of the production facility required and accelerate the turnover rate of batches. The simplicity of the operation also lowers the barrier for technical staff, reducing the likelihood of human error during execution. For supply chain heads, this means a more resilient production schedule capable of responding rapidly to market fluctuations. The ability to produce high-purity intermediates with fewer resources positions this novel approach as a superior alternative for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Wittig Olefination and Pd-Catalyzed Coupling
The success of this synthesis relies on the precise execution of two fundamental organic reactions: the Wittig olefination and the palladium-catalyzed carbonylative coupling. The first step involves the reaction of 7-chloro-2-quinolinecarboxaldehyde (Compound IV) with 3-bromobenzyl bromide. In this transformation, the benzyl bromide is first converted into a phosphonium salt using a tertiary phosphine reagent, such as triphenylphosphine or triethylphosphine. Subsequent treatment with a strong base generates a reactive ylide species in situ. This ylide then attacks the aldehyde group of the quinoline derivative, forming the desired (E)-alkene linkage with high stereoselectivity. 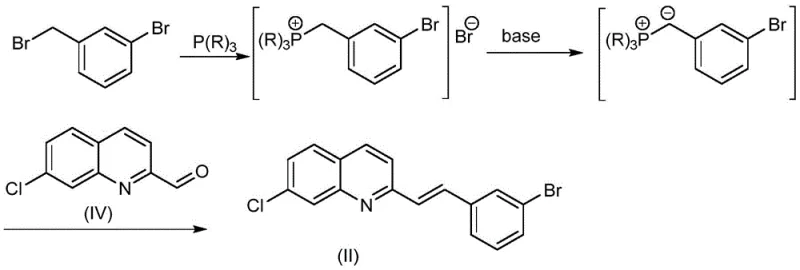 The patent highlights the versatility of this step, noting that various bases like sodium methoxide or potassium tert-butoxide can be employed effectively, allowing for flexibility in reagent sourcing.
The patent highlights the versatility of this step, noting that various bases like sodium methoxide or potassium tert-butoxide can be employed effectively, allowing for flexibility in reagent sourcing.
The second critical transformation is the coupling of the resulting bromide intermediate (Compound II) with the aldehyde ester (Compound III). This step utilizes a palladium catalyst system to forge the carbon-carbon bond connecting the aromatic core to the propyl side chain. The mechanism likely proceeds through an oxidative addition of the aryl bromide to the palladium center, followed by coordination and insertion of the alkene or aldehyde component, and finally reductive elimination to release the product. The choice of catalyst, such as palladium acetate or tris(dibenzylideneacetone)dipalladium, along with specific ligands, is crucial for maximizing turnover and minimizing side reactions. 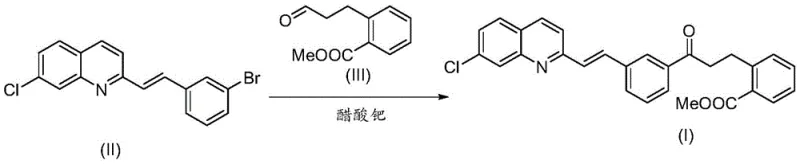 This catalytic cycle is highly efficient, enabling the reaction to proceed to completion within a few hours at temperatures around 120-140°C, demonstrating the robustness of the chemistry for industrial application.
This catalytic cycle is highly efficient, enabling the reaction to proceed to completion within a few hours at temperatures around 120-140°C, demonstrating the robustness of the chemistry for industrial application.
How to Synthesize Montelukast Sodium Intermediate Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and reaction monitoring to ensure optimal results. The patent provides specific embodiments that serve as a blueprint for laboratory and pilot-scale operations. The process begins with the preparation of the phosphonium salt, followed by the generation of the ylide and the subsequent Wittig reaction. Once the alkene intermediate is isolated, it is subjected to the coupling conditions with the appropriate aldehyde partner. Detailed standardized synthesis steps see the guide below.
- Perform a Wittig reaction between 7-chloro-2-quinolinecarboxaldehyde (IV) and 3-bromobenzyl bromide using a phosphine reagent and base to generate the alkene intermediate (II).
- Conduct a palladium-catalyzed coupling reaction between the resulting bromide (II) and the aldehyde ester (III) under mild heating conditions.
- Isolate the final product (I) through standard extraction, washing, and recrystallization procedures to ensure high purity suitable for downstream API synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers in the pharmaceutical supply chain, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple chemistry. The primary benefit lies in the structural simplification of the manufacturing process, which directly correlates to reduced operational complexity and lower capital expenditure requirements. By eliminating four to five synthetic steps compared to legacy methods, the new process drastically reduces the volume of solvents and reagents required per kilogram of product. This reduction in material intensity not only lowers direct material costs but also diminishes the logistical burden associated with storing and handling large quantities of hazardous chemicals.
- Cost Reduction in Manufacturing: The streamlined two-step process inherently drives down manufacturing costs by minimizing unit operations. Fewer steps mean less energy consumption for heating, cooling, and agitation, as well as reduced labor hours for monitoring and processing. Furthermore, the high yields reported in the patent examples suggest that raw material utilization is maximized, reducing the cost of goods sold (COGS). The elimination of expensive transition metal removal steps often required in other coupling strategies further contributes to significant cost savings, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as 3-bromobenzyl bromide and standard quinoline aldehydes, ensures a secure supply of inputs. Unlike routes that rely on bespoke or hard-to-source intermediates, this method leverages commodity chemicals that are readily accessible from multiple vendors. This diversification of the supply base mitigates the risk of shortages and price volatility. Additionally, the mild reaction conditions reduce the risk of batch failures due to thermal runaways or equipment incompatibility, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The simplicity of the workup procedures, involving standard extraction and crystallization techniques, facilitates easy scale-up from kilogram to tonne quantities. The process avoids the generation of complex waste streams associated with multi-step syntheses, simplifying effluent treatment and disposal. This alignment with green chemistry principles not only reduces environmental fees but also enhances the corporate sustainability profile of the manufacturer. The robustness of the palladium coupling step ensures that quality remains consistent even as batch sizes increase, supporting the commercial scale-up of complex pharmaceutical intermediates without compromising purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The novel route described in patent CN111848511A significantly reduces the number of synthetic steps from seven to just two major transformations. This reduction eliminates multiple isolation and purification stages, leading to lower solvent consumption, reduced waste generation, and a substantially shorter production cycle compared to prior art methods like WO2008058118.
Q: Which catalysts are preferred for the coupling step to ensure high yield?
A: The patent specifies several effective palladium catalysts, including palladium acetate and tris(dibenzylideneacetone)dipalladium. These catalysts, when used with appropriate ligands such as pyrrolidine derivatives, facilitate efficient coupling under relatively mild temperatures (120-140°C), ensuring robust conversion rates without requiring extreme pressure or hazardous conditions.
Q: How does this process impact the scalability of Montelukast production?
A: By utilizing common organic solvents like DMF, THF, or toluene and avoiding cryogenic conditions or highly unstable intermediates, this method is inherently designed for industrial scale-up. The operational simplicity allows for easier temperature control and safer handling of reagents, making it an ideal candidate for reliable large-scale manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful production of life-saving medications like Montelukast Sodium. Our technical team has thoroughly analyzed the innovative route described in CN111848511A and is fully prepared to implement this advanced chemistry at an industrial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for pharmaceutical applications.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce costs in pharmaceutical intermediate manufacturing through our commitment to innovation and quality excellence.
