Scalable Production of High-Purity Cefuroxime Acid via Advanced Ternary Solvent Extraction
Scalable Production of High-Purity Cefuroxime Acid via Advanced Ternary Solvent Extraction
The pharmaceutical landscape for second-generation cephalosporins demands intermediates of exceptional purity to ensure the safety and efficacy of final antibiotic formulations. Patent CN102134252A introduces a transformative preparation method for high-purity cefuroxime acid, addressing critical bottlenecks in the synthesis of cefuroxime sodium and cefuroxime axetil. This technology leverages a sophisticated purification strategy centered on a ternary solvent extraction system, effectively eliminating persistent lactone by-products that have historically plagued conventional manufacturing routes. By shifting from aqueous crystallization to a multi-phase solvent partitioning approach, this method achieves product purity levels exceeding 99%, setting a new benchmark for quality in the production of beta-lactam intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cefuroxime acid has been constrained by the inefficiencies inherent in aqueous phase crystallization techniques. Traditional protocols typically involve the formation of the acid in an aqueous environment, followed by direct crystallization, which presents severe thermodynamic limitations regarding impurity segregation. Specifically, these methods struggle to effectively remove side-reaction products such as lactones, which often persist in the final crystal lattice at concentrations ranging from 3.2% to 3.8%. Furthermore, the hygroscopic nature of the filter cake in aqueous systems results in moisture content surpassing 50%, necessitating prolonged drying cycles at elevated temperatures. These harsh drying conditions not only increase energy consumption but also degrade the thermal stability of the beta-lactam structure, leading to products with inferior color profiles and reduced shelf-life stability.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally restructures the downstream processing workflow by introducing a precise ternary solvent extraction system. Instead of relying on simple precipitation, the process converts the cefuroxime acid into its water-soluble sodium salt using sodium bicarbonate, allowing for a strategic liquid-liquid extraction. A carefully calibrated mixture of tetrahydrofuran, dichloromethane, and ethyl acetate is employed to selectively dissolve organic unsaponifiable impurities and lactone by-products into the organic phase, leaving the desired product in the aqueous layer. Following phase separation, the aqueous phase is acidified, and the pure cefuroxime acid is re-extracted into the organic solvent system. This dual-extraction mechanism ensures that water-soluble impurities are left behind in the aqueous waste stream while organic contaminants are removed in the initial wash, resulting in a white crystalline powder with exceptional stability.
Mechanistic Insights into N-Acylation and Solvent Partitioning
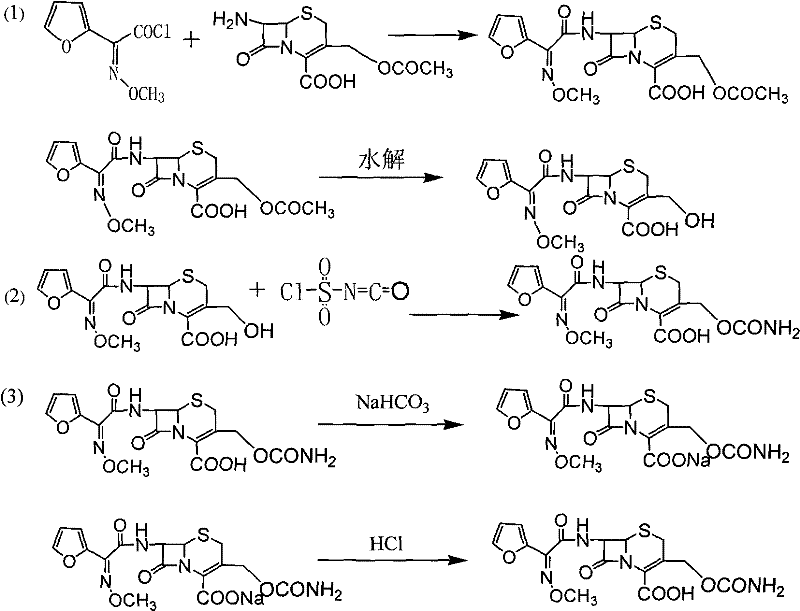
The synthetic route begins with the N-acylation of 7-aminocephalosporanic acid (7-ACA) using furoyl acetyl chloride, a reaction that must be meticulously controlled at low temperatures between -20°C and -60°C to prevent degradation of the sensitive beta-lactam ring. Following acylation, the 3-acetyl group is hydrolyzed using sodium hydroxide to yield the key intermediate, 3-deformamido cefuroxime acid (DCC). The subsequent nucleophilic addition of chlorosulfonyl isocyanate (CSI) to the DCC intermediate in tetrahydrofuran is the critical step that constructs the carbamate side chain essential for biological activity. This reaction is highly exothermic and requires cryogenic conditions (-30°C to -80°C) to maintain regioselectivity and prevent the formation of polymeric by-products or ring-opening degradation.
The true mechanistic breakthrough, however, lies in the physicochemical principles governing the purification stage. The utilization of a ternary solvent system exploits the differential solubility profiles of the cefuroxime sodium salt versus the lactone impurities. By adjusting the volume ratios of tetrahydrofuran, dichloromethane, and ethyl acetate, the polarity of the organic phase is tuned to maximize the partition coefficient of the lactone by-products into the organic layer while retaining the ionic sodium salt in the aqueous phase. Upon acidification with hydrochloric acid to a pH of 1.0 to 4.0, the cefuroxime acid precipitates or partitions back into the fresh organic solvent, effectively leaving behind inorganic salts and highly polar water-soluble impurities. This precise manipulation of solubility parameters is what enables the removal of trace impurities that co-crystallize in simpler solvent systems.
How to Synthesize Cefuroxime Acid Efficiently
The synthesis of high-purity cefuroxime acid requires strict adherence to temperature protocols and solvent ratios to ensure the successful removal of lactone impurities. The process integrates low-temperature acylation, controlled nucleophilic substitution, and a unique two-stage extraction protocol. For detailed operational parameters, including specific solvent volumes and stirring rates, refer to the standardized guide below.
- Perform N-acylation on 7-ACA with furoyl acetyl chloride followed by low-temperature hydrolysis to obtain the intermediate DCC.
- React DCC with chlorosulfonyl isocyanate in tetrahydrofuran at cryogenic temperatures to generate the cefuroxime acid precursor.
- Execute a dual-stage purification using a ternary solvent system (THF, dichloromethane, ethyl acetate) to remove lactone impurities and crystallize the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere technical specifications. The elimination of persistent lactone impurities reduces the risk of batch rejection during quality control testing, thereby enhancing overall supply reliability. Furthermore, the process design inherently supports cost reduction initiatives by optimizing raw material utilization and minimizing the need for extensive reprocessing or recrystallization steps that typically erode profit margins in antibiotic intermediate manufacturing.
- Cost Reduction in Manufacturing: The implementation of the ternary solvent extraction system significantly lowers production costs by streamlining the purification workflow. Unlike traditional methods that require multiple recrystallizations to achieve acceptable purity, this single-pass extraction protocol efficiently removes impurities, reducing solvent consumption and labor hours. Additionally, the solvents used—tetrahydrofuran, dichloromethane, and ethyl acetate—are commodity chemicals that can be recovered and recycled through distillation, further driving down the variable cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route directly translates to improved supply chain continuity. By achieving yields exceeding 91% consistently, manufacturers can better forecast output volumes and meet tight delivery schedules without the buffer stock typically required to account for low-yield batches. The use of stable, commercially available starting materials like 7-ACA and chlorosulfonyl isocyanate ensures that raw material sourcing remains resilient against market fluctuations, securing a steady flow of high-quality intermediates for downstream drug formulation.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is exceptionally well-suited for commercial scale-up. The reduction in drying time and temperature not only saves energy but also minimizes the thermal degradation of the product, ensuring consistent quality across large batch sizes. Moreover, the ability to recycle the organic solvent mixture aligns with modern green chemistry principles, reducing the volume of hazardous waste generated and simplifying compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of cefuroxime acid using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners and technical stakeholders evaluating this supply chain solution.
Q: How does the ternary solvent system improve cefuroxime acid purity compared to traditional methods?
A: Traditional aqueous crystallization often traps lactone by-products and results in high moisture content. The patented ternary solvent system (THF/dichloromethane/ethyl acetate) selectively extracts organic unsaponifiable impurities and lactones into the organic phase while keeping the sodium salt in the aqueous phase, enabling a purity level exceeding 99%.
Q: What are the critical temperature controls required for the nucleophilic addition step?
A: The reaction between the DCC intermediate and chlorosulfonyl isocyanate is highly exothermic and sensitive. The process requires maintaining cryogenic conditions between -30°C and -80°C during the dropwise addition to prevent side reactions and ensure the structural integrity of the beta-lactam ring.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It utilizes common industrial solvents that can be recycled, avoids expensive transition metal catalysts, and achieves high yields (>91%) with simplified drying steps due to low moisture content in the final filter cake.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the production of high-purity cefuroxime acid requires not just chemical expertise but a deep understanding of scalable process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest international pharmacopoeia standards.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their antibiotic supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product quality while reducing overall procurement costs.
